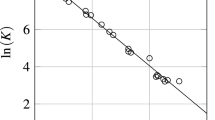
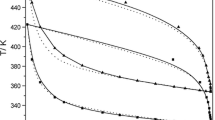
Abstract
A detailed thermodynamic study of the systems LnSe2–LnSe1.5 (Ln = La, Nd) was performed by static method of vapour pressure measurement using quartz membrane-gauge manometers within the temperature range 713–1,395 K. The p Se–T–x dependences obtained in this study have shown that the phase regions in composition intervals studied consist of discrete phases: LnSe1.95 LnSe1.90, LnSe1.85, LnSe1.80 (Ln = La, Nd). The enthalpies and the entropies for the stepwise dissociation process were calculated from the experimental data. The standard enthalpies of formation and the absolute entropies were estimated for the compounds investigated using literature data.


Similar content being viewed by others
References
Bootcher P, Doert Th, Arnold H, Tamazyan R. The compounds with layer structures LnX2. Z Kristallogr. 2000;215:246–52.
Jarembash EI, Eliseev AA. Chalkogenidi Redkosemelnich Elementov. Moscow: Nauka; 1975. (in Russian).
Eliseev AA, Grizik AA. Redkozemelnie Poluprovodniki. Leningrad: Nauka; 1977. (in Russian).
Michioka Ch, Fukushima K, Suzuki K, Yoshimira K. Physical properties of RSe2-x (R = Ce, Nd, Sm and Gd) and Rb x GdSe3-y . J Phys Chem Solids. 2005;66:1579–82.
Doert Th, Tsinde BPF, Lidin S, Garcia FJ. Site occupancy wave and charge density wave in the modulated structure of Nd0.6Gd0.4Se1.85. J Solid State Chem. 2004;177:1598–606.
Chusova TP, Zelenina LN, Vasilyeva IG, Graf C, Doert Th. Thermodynamic study of the systems PrSe2–PrSe1.5 and GdSe1.875–GdSe1.5. J Alloys Compd. 2008;452:94–8.
Doert Th, Graf C, Schmidt P, Vasilyeva IG, Simon P, Carrillo-Caberera W. The Pr2Se3–PrSe1.5 system: studies of the phase relationships and the modulated crystal structure of PrSe1.85. J Solid State Chem. 2007;180:496–509.
Dashjav E, Oeckler O, Doert Th, Mattausch H, Bottcher P. Gd8Se1.5–A 24-fold superstructure of the ZrSSi type. Angew Chem Int Ed. 2000;39:1987–8.
Vasilyeva IG, Belyaeva EI. Thermodynamic study of the SmS2–SmS1.5 system. J Solid State Chem. 1999;142:261–5.
Zelenina LN, Chusova TP, Stenin YuG, Bakovets VV. Fluorophenyl derivatives of elements II–VI groups: phase transitions thermodynamics and growing films by CVD. J Therm Anal Calorim. 2008;92:723–7.
Titov VA, Chusova TP, Kokovin GA. Improved variant of the state method for gas equilibria investigation at the high temperatures. Izv Akad Nauk SSSR ser chim. 1979;6:3–7. (in Russian).
Zelenina LN, Titov VA, Chusova TP, Stenin YuG, Titov AA. On the thermodynamic properties of Germanium-iodide compounds. J Chem Thermodyn. 2003;35:1601–12.
Belov GV, Iorish VS, Yungman VS. IVTANTHERMO for database on thermodynamic properties and related software. CALPHAD 1999; 23:173–180.
Zelenina LN, Titov AA, Chusova TP. Chemical equilibria in the gas phase of the Ge–Br system. Russ J Phys Chem. 2005;79:43–6.
Kopun A. Abstract “Heat capacity, entropy, effective Gibbs energy and enthalpy of rare earth metal selenides under standard conditions”. Kiev; 2005 (in Russian).
Gordienko SP, Fenochka BV, Wiksman GSh, editors. Thermodynamic of lanthanum compounds. Kiev: Naukova dumka; 1979. (in Russian).
Acknowledgements
This study was financially supported by the “Program in Support of Leading Scientific Schools”, project RF NSh no. 636.2008.3.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zelenina, L.N., Chusova, T.P. & Vasilyeva, I.G. Thermodynamic investigation of the systems LnSe2–LnSe1.5 (Ln = La, Nd). J Therm Anal Calorim 101, 59–62 (2010). https://doi.org/10.1007/s10973-010-0843-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-010-0843-4