Abstract
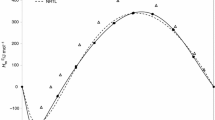
We have determined the excess molar enthalpies \( H_{\text{m}}^{\text{E}} \) at 298.15 K and normal atmospheric pressure for the binary mixtures containing dichloropropane and n-alkane [{xCH2ClCHClCH3 + (1−x) C n H2n+2 (n = 6, 8, 10, 12)} and {xCH2ClCH2CH2Cl + (1−x) C n H2n+2 (n = 8, 10)}] using a Calvet microcalorimeter. The \( H_{\text{m}}^{\text{E}} \) values for all the mixtures show endothermic behaviour for the whole composition range. The Redlich–Kister equation was used to correlated the experimental values. The experimental excess molar enthalpies were examined on basis of the DISQUAC group-contribution model and the UNIFAC group-contribution method using the version considered by Larsen et al. The experimental and calculated results are discussed in terms of molecular interactions and the proximity effect.




Similar content being viewed by others
References
Kehiaian HV, Maronguiu B. A comparative study of thermodynamic properties and molecular interactions in mono- and polychloroalkane + n-alkane or + cyclohexane mixtures. Fluid Phase Equilib. 1988;40:23–78.
Larsen BL, Rasmussen P, Fredenslund A. A modified UNIFAC group-contribution model for prediction of phase equilibria and heats of mixing. Ind Eng Chem Res. 1987;26:2274–86.
Ortega J, Plácido J. Excess enthalpy. 1, 3- Dichloropropane—pentane system. Int DATA Ser Sel Data Mixtures Ser A. 1993;21(1):10.
Ortega J, Plácido J. Excess enthalpy. 1, 3- Dichloropropane—heptane system. Int DATA Ser Sel Data Mixtures Ser A. 1993;21(1):11.
Ortega J, Plácido J. Excess enthalpy. 1, 3- Dichloropropane—nonane system. Int DATA Ser Sel Data Mixtures Ser A. 1993;21(1):12.
Ortega J, Plácido J. Excess enthalpy. 1, 3- Dichloropropane—undecane system. Int DATA Ser Sel Data Mixtures Ser A. 1993;21(1):13.
Baños I, Valero J, Pérez P, Gracia M, Gutiérrez Losa C. Excess molar enthalpies of mixtures containing 1, 3-dichloropropane. J Chem Thermodyn. 1990;22:67–72.
Polo C, Gutiérrez Losa C, Kechavarz MR, Kehiaian HV. Excess enthalpies of liquid α, ω -dichloroalkane + n-hexane mixtures. A group contribution study of the Cl–Cl proximity effect. Ber Bunsen Ges Phys Chem. 1980;84:525–9.
Grolier JPE, Kehiaian HV. Enthalpies of mixing of some α, ω-dichloroalkane + hydrocarbon systems. J Chim Phys Phys Chim Biol. 1973;70:807–10.
Baños I, Valero J, Pérez P, Gracia M, Gutiérrez Losa C. Excess molar enthalpies of mixtures containing 1, 2-dichloropropane. J Chem Thermodyn. 1989;21:709–12.
Mc Glashan ML, Stoeckli HF. Flow calorimeter for enthalpies of mixing. Enthalpy of mixing of n-hexane + cyclohexane at 298.15.deg.K. J Chem Thermodyn. 1969;1:589–94.
Paz Andrade MI. Les développements récents de la microcalorimétrie et de la thermogenèse. 1st ed. Paris: Centre national de la recherche scientifique-CRNS; 1967.
Paz Andrade MI, Castromil S, Baluja MC. Enthalpies of mixing: n-hexane + o-xylene at 25, 35, and 50°C. J Chem Thermodyn. 1970;2:775–7.
IUPAC (International Union of Pure and Applied Chemistry). Atomic weights of the elements 1985. Pure Appl Chem. 1986;58:1677–92.
Redlich O, Kister AT. Thermodynamics of nonelectrolytic solutions. Algebraic representation of thermodynamic properties and the classification of solutions. J Ind Eng Chem. 1948;40:345–8.
Marquardt DW. An algorithm for least squares estimation of nonlinear parameters. J Soc Ind Appl Math. 1963;11(2):431–41.
Bevington P. Data reduction and error analysis for the physical sciences. 1st ed. New York: McGraw-Hill; 1969.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mato, M.M., Fernández, J., Legido, J.L. et al. Excess molar enthalpies of dichloropropane + n-alkane mixtures. J Therm Anal Calorim 101, 1121–1125 (2010). https://doi.org/10.1007/s10973-010-0732-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-010-0732-x