Abstract
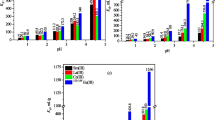
Commercial bentonite (BFN) and organoclay (WS35), as well as iron oxide/clay composite (Mag_BFN) and iron/oxide organoclay composite (Mag_S35) were prepared for toluene and naphthalene sorption. Mag_BFN and Mag_S35 were obtained, respectively, by the precipitation of iron oxide hydrates onto sodium BFN and S35 clay particles. The materials were characterized by powder X-ray diffraction (XRD), X-ray Fluorescence (XRF), and TG and DTA. From XRF results and TG data on calcined mass basis, a quantitative method was developed to estimate the iron compound contents of the composites, as well as the organic matter content present in WS35 and Mag_S35.







Similar content being viewed by others
Abbreviations
- BFN:
-
Commercial bentonite
- BTEX:
-
Benzene, toluene, ethylbenzene, and xylenes
- d 001 :
-
Basal spacing
- DTA:
-
Differential thermal analysis
- Mag_BFN:
-
Iron oxide/clay composite
- Mag_S35:
-
Iron/oxide organoclay composite
- M cS :
-
Calcined mass of sample S
- M iS :
-
Initial mass of sample S
- NOC:
-
Nonpolar organic compounds
- PAH:
-
Polycyclic aromatic hydrocarbons
- S35:
-
Unwashed organoclays
- TG:
-
Thermogravimetry
- WS35:
-
Washed organoclay
- XRD:
-
Powder X-ray diffraction (XRD)
- XRF:
-
X-ray fluorescence
- γ-Fe2O3 :
-
Maghemite
References
Jaynes WF, Boyd SA. Trimethylphenylamonium -smectite as an effective adsorbent of water soluble aromatic hydrocarbons. J Air Waste Manag Assoc. 1990;40:1649–53.
Yariv S, Lapides I. The use of thermo-xrd-analysis in the study of organo-smectite complexes. J Therm Anal Calorim. 2005;80:11–26.
Vianna MMG, Dweck J, Kozievitch VJ, Valenzuela-Diaz FR, Büchler PM. Characterization and study of sorptive properties of differently prepared organoclays from a Brazilian natural bentonite. J Therm Anal Calorim. 2005;82:595–602.
Boyd SA, Mortland MM, Chiou CT. Sorption characteristics of organic compounds on hexadecyltrimethylammonium-smectite. Soil Sci Soc Am J. 1988;52:652–7.
Sora NI, Pelosato R, Zampori L, Botta D, Dotelli G, Vitelli M. Matrix optimization for hazardous organic waste sorption. Appl Clay Sci. 2005;28:43–54.
Dekany I, Turi L, Tombacz E, Fendler JH. Preparation of size-quantized CdS and ZnS particles in nanophase reactors provided by binary liquids adsorbed at layered silicates. Langmuir. 1995;11:2285–92.
Dekany I, Turi L, Kiraly Z. CdS, TiO and Pd8 nanoparticles growing in the 2 interlamellar space of montmorillonite in binary liquids. Appl Clay Sci. 1999;15:221–39.
Oliveira LCA, Rios RVRA, Fabris JD, Lago RM. A simple preparation of magnetic composites for the adsorption of water contaminants. J Chem Educ. 2004;81:248–50.
Bourlinos AB, Karakassides MA, Simopoulos A, Petridis D. Synthesis and characterization of magnetically modified clay composites. Chem Mater. 2000;12:2640–5.
Bourlinos AB, Devlin E, Boukos N, Simopoulos A, Petridis D. Magnetite and Co ferrite-based clay composites. Clay Miner. 2002;37:135–41.
Mastalir A, Notheisz F, Kiraly Z, Bartok M, Dekany I. Novel clay intercalated metal catalysts: a study of the hydrogenation of styrene and 1-octene on clay intercalated Pd catalysts. Stud Surf Sci Catal. 1997;108:477–84.
Penchev IP, Hristov JY. Behaviour of fluidized beds of ferromagnetic particles in an axial magnetic field. Powder Technol. 1990;61:103–18.
Yariv S. The role of charcoal on DTA curves of organo-clay complexes: an overview. Appl Clay Sci. 2004;24:225–36.
Morais LC, Valenzuela-Diaz FR, Dweck J, Büchler PM. Thermal analysis characterization of lactose and derived products in dairy product industry effluents processing. J Therm Anal Calorim. 2005;82:315–8.
Dweck J. Qualitative and quantitative characterization of Brazilian natural and organophilic clays by thermal analysis. J Therm Anal Calorim. 2008;92(1):129–35.
Lagaly G. Layer charge heterogeneity in vermiculites. Clays Clay Miner. 1982;30:215–22.
Jaynes WF, Boyd SA. Clay mineral type and organic compound sorption by hexadecyltrimethylammonium-exchanged clays. Soil Sci Soc Am J. 1991;55:43–8.
Cornell RM, Schwertmann U. The iron oxides. structure, properties, reactions, occurences and uses. 1st ed. Weinheim: VCH; 1996. p. 127–73.
Tabak A, Afsin B, Aygun SF, Koksal E. Structural characteristics of organo-modified bentonites of different origin. J Therm Anal Calorim. 2007;87:375–81.
Chen D, Zhu JX, Yuan P, Yang SJ, Chen T-H, He HP. Preparation and characterization of anion-cation surfactants modified montmorillonite. J Therm Anal Calorim. 2008;94:841–8.
Acknowledgements
To the National Institute of Science and Technology for Environmental Studies (INCT-EMA), State of São Paulo Research Foundation (FAPESP), BF Clay Especialidades Lta, X-ray Laboratory, and X-ray Fluorescence Laboratory of the Geosciences Institute of the University of São Paulo.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vianna, M.M.G.R., Dweck, J., Quina, F.H. et al. Toluene and naphthalene sorption by iron oxide/clay composites. J Therm Anal Calorim 100, 889–896 (2010). https://doi.org/10.1007/s10973-009-0427-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0427-3