Abstract
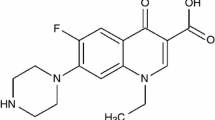
Some metal complexes of norfloxacin (NOR) with the formula [M(NOR)2]X2·nH2O [M = Zn(II), (X = Cl−, AcO−, Br− and I−), Cd(II), (X = Cl−, NO3 − and SO4 2−) and Hg(II) (X = Cl−, NO3 − and AcO−)] have been synthesised and studied using elemental analysis (CHN), electronic (UV–vis, mid infrared, mass, and 1H-NMR spectra), TG and DTA. The thermal decomposition processes of these complexes were discussed. The Correlation coefficient, the activation energies, E*, the pre-exponential factor, A, the entropies, S*, enthalpies, H* and Gibbs free energies, G*, of the thermal decomposition reactions have been derived from thermogravimetric (TG) and differential thermogravimetric (DTG) curves. The characterization of the final products of the decomposition was achieved by IR spectra and X-ray powder diffraction (XRD). Using the Coats–Redfern and Horowitz–Metzeger methods, kinetic analysis of the thermogravimetric data is performed.






Similar content being viewed by others
References
Zaki A, Schreiber EC, Weliky I, Knill JR, Hubsher JA. Clinical pharmacology of oral cephradine. J Clin Pharmacol. 1974;14:118–26.
Klastersky J, Daneau D, Weerts D. Cephradine. Chemotherapy. 1973;18:191–204.
Anacona JR. Synthesis and antibacterial activity of some metal complexes of beta-lactamic antibiotics. J Coord Chem. 2001;54:355–65.
Lozano MJ, Borrás J. Antibiotic as ligand. Coordinating behavior of the cephalexin towards Zn(II) and Cd(II) ions. J Inorg Biochem. 1987;31:187–95.
Zhao A, Carraher CE, Barone G, Pellerito C, Scopelliti M, Pellerito L. Mössbauer investigation on organotin polyester amines containing ciprofloxacin. Polym Mater: Sci Eng. 2005;93:414–6.
Iqbal MS, Ahmad AR, Sabir M, Asad SM. Preparation, characterization and biological evaluation of copper(II) and zinc(II) complexes with cephalexin. J Pharm Pharmacol. 1999;51:371–5.
Sorenson JRJ. Copper chelates as possible active forms of the antiarthritic agents. J Med Chem. 1976;19:135–48.
Brown DH, Smith WE, Teape JW, Lewis AJ. Antiinflammatory effects of some copper complexes. J Med Chem. 1980;23:729–34.
Williams DR. The metals of life. London: Van Nostrand Reinhold; 1971.
Ruiz M, Perelló L, Ortiz R, Castiñeiras A, Maichle-Mössmer C, Cantón E. Synthesis, characterization, and crystal structure of [Cu(cinoxacinate)2]·2H2O complex: a square-planar CuO4 chromophore. Antibacterial studies. J Inorg Biochem. 1995;59:801–10.
Castillo-Blum SE, Barba-Behrens N. Coordination chemistry of some biologically active ligands. Coord Chem Rev. 2000;196:3–30.
Turel I, Leban I, Bukovec N. Crystal structure and characterization of the bismuth(III) compound with quinolone family member (ciprofloxacin). Antibacterial study. J Inorg Biochem. 1997;66:241–5.
Turel I, Golič L, Bukovec P, Gubina M. Antibacterial tests of bismuth(III)–quinolone (ciprofloxacin, cf) compounds against Helicobacter pylori and some other bacteria. Crystal structure of (cfH2)2[Bi2Cl10]·4H2O. J Inorg Biochem. 1998;71:53–60.
Yang P, Li JB, Tian YN, Yu KB. Synthesis and crystal structure of rare earth complex with Ciprofloxacin. Chin Chem Lett. 1999;10:879–80.
Wu G, Wang G, Fu X, Zhu L. Synthesis, crystal structure, stacking effect and antibacterial studies of a novel quaternary copper(II) complex with quinolone. Molecules. 2003;8(2):287–96.
Turel I, Leban I, Klintschar G, Bukovec N, Zalar S. Synthesis, crystal structure, and characterization of two metal-quinolone compounds. J Inorg Biochem. 1997;66:77–82.
Turel I, Gruber K, Leban I, Bukovec N. Synthesis, crystal structure, and characterization of three novel compounds of the quinolone family member (norfloxacin). J Inorg Biochem. 1996;61:197–212.
Turel I, Leban I, Zupancic M, Bukovec N, Gruber K. An adduct of magnesium sulfate with a member of the quinolone family (ciprofloxacin). Acta Crystallogr C. 1996;52:2443–5.
Chen Z-F, Xiong R-G, Zuo J-L, Guo Z, You X-Z, Fun H-K. X-Ray crystal structures of Mg2+ and Ca2+ dimers of the antibacterial drug norfloxacin. J Chem Soc Dalton Trans. 2000;4013–4014.
Al-Mustafa J. Magnesium, calcium and barium perchlorate complexes of ciprofloxacin and norfloxacin. Acta Chim Slov. 2002;49:457–66.
Ruíz M, Perelló L, Server-Carrió J, Ortiz R, García-Granda S, Díaz MR, et al. Cinoxacin complexes with divalent metal ions. Spectroscopic characterization. Crystal structure of a new dinuclear Cd(II) complex having two chelate-bridging carboxylate groups. Antibacterial studies. J Inorg Biochem. 1998;69:231–9.
Gupta R, Saxena RK, Chaturvedi P, Virdi JS. Chitinase production by Streptomyces viridificans: its potential in fungal cell wall lysis. J Appl Bacteriol. 1995;78:378–83.
Geary WJ. The use of conductivity measurements in organic solvents for the characterisation of coordination compounds. Coord Chem Rev. 1971;7:81–122.
Coats W, Redfern JP. Kinetic parameters from thermogravimetric data. Nature. 1964;201:68–9.
Omar MM. Spectral, thermal and biological activity studies on ruthenium(II) complexes with some pyridylamines. J Therm Anal Calorim. 2009;96:607–15.
Rotaru A, Goşa M, Rotaru P. Computational thermal and kinetic analysis. Software for non-isothermal kinetics by standard procedure. J Therm Anal Calorim. 2008;94:367–71.
Verma RK, Verma L, Bhushan A, Verma BP. Thermal decomposition of complexes of cadmium(II) and mercury(II) with triphenylphosphanes. J Therm Anal Calorim. 2007;90:725–9.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Refat, M.S., Mohamed, G.G., de Farias, R.F. et al. Spectroscopic, thermal and kinetic studies of coordination compounds of Zn(II), Cd(II) and Hg(II) with norfloxacin. J Therm Anal Calorim 102, 225–232 (2010). https://doi.org/10.1007/s10973-009-0404-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0404-x