Abstract
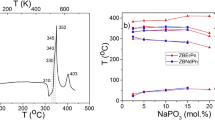
Glasses have been synthesized in the system SiO2–Al2O3–Na2O–AlF3–LaF3–Er2O3. A base glass (in mol% 67SiO2–9Al2O3–20Na2O–Al2F6–3La2F6) was modified by 0.5, 0.75, 1, 1.25, 1.5, 2 and 5 mol% Er2O3, respectively. Glasses were prepared by conventional fusion method from 20 g batches. The glass transition temperature (T g), the jump-like changes of the specific heat (ΔC p) accompanying the glass transition and the enthalpy of crystallization (ΔH) were calculated. DTA measurements clearly reveal that the increase of the Er2O3 content in the glass changes the effects of crystallization and diminishes the thermal stability of the glassy network. In the same time the changes in the transition temperature are observed. The formation of NaLaF4 and Na1.45La9.31(SiO4)6(F0.9O1.1) as a main phase was confirmed. The diminishing of the thermal stability was connected with erbium which incorporated into Na1.45La9.31(SiO4)6(F0.9O1.1) structure.





Similar content being viewed by others
References
Mortier M, Bensalah A, Dantelle G, Patriarche G, Vivien D. Rare-earth doped oxyfluoride glass-ceramics and fluoride ceramics: synthesis and optical properties. Opt Mater. 2007;29:1263–70.
Kam CH, Buddhudu S. Photoluminescence properties of Eu3+:ZrF4-BaF2-LaF3-YF3-AlF3-NaF glasses. Phys B: Phys Condens Matter. 2004;344(1–4):182–9.
Becker PC, Olsson NA, Simpson JR. Erbium-doped fiber amplifier: fundamentals and technology. San Diego, CA: Academic Press; 1999.
Miniscalco WJ. Erbium-doped glasses for fibre amplifiers at 1500 nm. J Lightwave Technol. 1991;9:234–50.
Marcus MA, Polman A. Local structure around Er in silica and sodium-silicate glasses. J Non-Cryst Solids. 1991;136:260–5.
Neeves AE, Bruce AJ, Reed WA, Rabinovich EM, Grodkiewicz WH, Kopylov NA, et al. Effect of concentration, host glass composition, and processing on the absorption and emission of Er+3 ions in sodium silicate glasses. In: Bruce AJ, Hiremath BV, editors. Solid state optical materials, vol. 28. Westerville, OH: American Ceramic Society; 1992. p. 353–67.
Du J, Cormack AN. The structure of erbium doped sodium silicate glasses. J Non-Cryst Solids. 2005;351:2263–76.
Delben RJ, Chaves DR, Candelorio PD, Delben AAST. Glass forming ability and thermal stability in the system ZrF46BaF26PrF3. J Therm Anal Calorim. 2006;83:411–4.
Reben M, Wacławska I, Paluszkiewicz C, Środa M. Thermal and structural studies of nanocrystallization of oxyfluoride glasses. J Therm Anal Calorim. 2007;88:285–9.
Dejneka MJ, Transparent oxyfluoride glass ceramics. MRS Bulletin, November 1998, 57–62.
Tikhomirov K, Furniss D, Seddon AB, Reaney IM, Beggiora M, Ferrari M, et al. Fabrication and characterization of nanoscale, Er3+-doped, ultratransparent oxyfluoride glass-ceramics. Appl Phys Lett. 2002;81(11):1937–9.
Środa M. Effect of LaF3 admixture on thermal stability of borosilicate glasses. J Therm Anal Calorim. 2007;88:245–9.
Volf MB. Chemical approach to glass, glass science and technology, vol. 7. Elsevier, Amsterdam; 1984.
Mortier M, Patriarche G. Structural characterisation of transparent oxyfluoride glass-ceramics. J Mater Sci. 2000;35:4849–56.
Dantelle G, Mortier M, Vivien D, Patriarche G. Nucleation efficiency of erbium and ytterbium fluorides in transparent oxyfluoride glass-ceramics. J Mater Res. 2005;20(2):472–81.
Handke M, Mozgawa W. Vibrational spectroscopy of the amorphous silicates. Vibrat Spect. 1993;5:75–84.
Tarte P. Infrared spectra of inorganic aluminates and characteristic vibrational frequencies of AlO4 tetrahedra and AlO6 octahedra. Spectrochim Acta. 1967;A23:2127–43.
Handke M, Mozgawa W. Model quasi-molecule Si2O as an approach in the IR spectra description glassy and crystalline framework silicates. J Mol Struct. 1995;348:341–4.
Acknowledgements
The work was supported by the Polish Ministry of Education and Science, grant No. 3T08D04829.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Środa, M. Effect of Er2O3 on thermal stability of oxyfluoride glass. J Therm Anal Calorim 97, 239–243 (2009). https://doi.org/10.1007/s10973-009-0257-3
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0257-3