Abstract
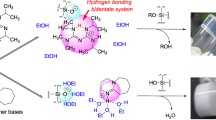
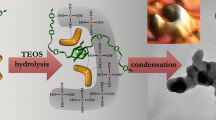
Hybrid organic–inorganic materials, silica–diol, were synthesized by the sol–gel process from mixtures of tetraethylorthosilicate (TEOS) and diols: ethylene glycol (HO–CH2–CH2–OH) and 1,3 propane diol (HO–CH2–CH2–CH2–OH), in acid catalysis. The gels have been synthesized for a molar ratio H2O:TEOS = 4:1 and different molar ratios diol/TEOS: 0.25; 0.5; 0.75; 1.0; 1.25 and 1.5. The resulting gels were studied by thermal analysis and FT-IR spectroscopy, in order to evidence the interaction of diols with silica matrix. Thermal analysis indicated that the condensation degree increases with the molar ratio diol/TEOS until a certain value. The thermal decomposition of the organic chains bonded within the silica network in the temperature range 250–320 °C, leaded to a silica matrix with modified morphology. The adsorption–desorption isotherms type is different for the samples with and without diol. Thus, the specific surface areas have values <11 m2/g for the samples without diol and >200 m2/g for the samples with diols, depending on the annealing temperature.







Similar content being viewed by others
References
Guerrero H, Rosa G, Morales MP, del Monte F, Moreno EM, Levy D, et al. Faraday rotation in magnetic gamma-Fe2O3/SiO2 nanocomposites. Appl Phys Lett. 1997;71:2698–700.
Lund CRF, Dumesic JA. Strong oxide-oxide interactions in silica-supported magnetite catalysts. 2. The core/shell nature of the interaction. J Phys Chem. 1982;86:130–5.
Atarashi T, Kim YS, Fujita T, Nakatsuka K. Synthesis of ethylene-glycol-based magnetic fluid using silica-coated iron particle. J Magn Magn Mater. 1999;201:7–10.
Li P, Miser DE, Rabiei S, Yadav RT, Hajaligol MR. The removal of carbon monoxide by iron oxide nanoparticles. Appl Catal B Environ. 2003;43:151–62.
Taylor JI, Hurst CD, Davis MJ, Sachsinger N, Bruce IJ. Application of magnetite and silica–magnetite composites to the isolation of genomic DNA. J Chrom A. 2000;890:159–66.
Brinker CJ, Scherrer GW. Sol-gel science: the physics and chemistry of sol-gel processing. New York: Academic Press; 1990.
Kursawe M, Anselmann R, Hilarius V, Pfaff G. Nano-particles by wet chemical processing in commercial applications. J Sol-Gel Sci Technol. 2005;33:71–4.
Laridjani M, Lafontaine E, Bayyl JP, Judeinstein P. Structural studies of ideal organic-inorganic nanocomposites by high-resolution diffractometry and NMR spectroscopy. J Mater Sci. 1999;34:5945–53.
Ågren P, Rosenhohn JB. Phase behavior and structural changes in tetraethylorthosilicate-derived gels in the presence of polyethylene glycol, studied by rheological techniques and visual observations. J Colloid Interface Sci. 1998;204:45–52.
Siuzdak D, Start PR, Mauritz KA. Surlyn (R)/silicate hybrid materials. I. Polymer in situ sol-gel process and structure characterization. J Appl Polym Sci. 2000;77:2832–44.
Lin JM, Ma CCM, Wang FY, Wu HD, Kuang SC. Thermal, mechanical, and morphological properties of phenolic resin/silica hybrid creamers. J Polym Sci B Polym Phys. 2000;38:1699–706.
Stefanescu M, Stoia M, Stefanescu O, Popa A, Simon M, Ionescu C. The interaction between TEOS and some polyols. J Therm Anal Cal. 2007;88(1):19–26.
Stefanescu M, Stoia M, Stefanescu O. Thermal and FT-IR study of the hybrid ethylene-glycol–silica matrix. J Sol-Gel Sci Tech. 2007;41:71–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ştefănescu, O., Stoia, M., Ştefănescu, M. et al. Study on the influence of teos–diol molar ratio on their chemical interaction during the gelation process. J Therm Anal Calorim 97, 251 (2009). https://doi.org/10.1007/s10973-009-0247-5
Published:
DOI: https://doi.org/10.1007/s10973-009-0247-5