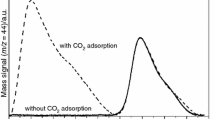
Abstract
The effect of Cu/Al molar ratio on the high-temperature adsorption characteristics of CO2 on the mixed oxides of Cu–Al hydrotalcite skeletal structure has been studied by thermogravimetry. The Cu/Al molar ratio of the hydrotalcites synthesized was varied between 1.0 and 3.0, and the adsorption temperature ranged from ambient to 600 °C. The hydrotalcite with Cu/Al molar ratio of 2.0 was found to be the most suitable adsorbent for high-temperature CO2 adsorption, in both the capacity and the rate of adsorption. The activation energy values suggested that the physical adsorption dominates at low temperatures (<400 °C) and the chemisorption dominates at high temperatures (>400 °C).





Similar content being viewed by others
References
Burchell TD, Judkins RR, Rogers MR, Williams AM. A novel process and material for the separation of carbon dioxide and hydrogen sulfide gas mixtures. Carbon. 1997;35:1279–94.
McKernan IW, Holder MJ. Purification of hydrocarbons. GB Patent 2267096; 1993.
Wieland PO. Living together in space: the design and operation of the life support systems on the International Space Station. NASA TM-206956, XI-267; 1998.
Ding Y, Elpay E. Adsorption-enhanced steam–methane reforming. Chem Eng Sci. 2000;55:3929–40.
Hufton JR, Mayorga S, Sircar S. Sorption-enhanced reaction process for the hydrogen production. AIChE J. 1999;45:248–56.
Hufton J, Mayorga S, Gaffney T, Nataraj S, Sircar S. Sorption enhanced reaction process (SERP). USDOE Hydrogen Program Rev. 1997;1:179–94.
Lee KB, Beaver MG, Caram HS, Sircar S. Reversible chemisorption of carbon dioxide: simultaneous production of fuel-cell grade H2 and compressed CO2 from synthesis gas. Adsorption. 2007;13:385–97.
Gaffney TR, Golden TC, Mayorga SG, Brzozowski JR, Tayler FW. Carbon dioxide pressure swing adsorption process using modified alumina adsorbents. US Patent 5917136; 1999.
Sakadjian BB, Iyer MV, Gupta H, Fan L-S. Kinetics and structural characterization of calcium-based sorbents calcined under subatmospheric conditions for the high-temperature CO2 capture process. Ind Eng Chem Res. 2007;46:35–42.
Auroux A, Gervasini A. Microcalorimetric study of the acidity and basicity of metal oxides surface. J Phys Chem. 1990;94:6371–9.
Cho YK, Han KW, Lee KH. Separation of CO2 by modified γ-Al2O3 membranes at high temperature. J Membr Sci. 1995;104:219–30.
Lee KB, Beaver MG, Caram HS, Sircar S. Chemisorption of carbon dioxide on sodium oxide promoted alumina. AIChE J. 2007;53:2824–31.
Yong Z, Mata VG, Rodrigues AE. Adsorption of carbon dioxide on chemically modified high surface area carbon-based adsorbents at high temperature. Adsorption. 2001;7:41–50.
Plaza MG, Pevida C, Arias B, Fermoso J, Arenillas A, Rubiera F, et al. Application of thermogravimetric analysis to the evaluation of aminated solid sorbents for CO2 capture. J Therm Anal Cal. 2008;92:601–6.
Ding Y, Elpay E. Equilibria and kinetics of CO2 adsorption on hydrotalcite adsorbent. Chem Eng Sci. 2000;55:3461–74.
Yong Z, Mata V, Rodrigues AE. Adsorption of carbon dioxide at high temperature – a review. Sep Purif Technol. 2002;26:195–205.
Hutson ND, Attwood BC.High temperature adsorption of CO2 on various hydrotalcite-like compounds. Adsorption. 2008;14:781–9.
Reddy MKR, Xu ZP, Lu GQ, Diniz da Costa JC. Layered double hydroxides for CO2 capture: structure evolution and regeneration. Ind Eng Chem Res. 2006;45:7504–9.
Yong Z, Mata V, Rodrigues AE. Adsorption of carbon dioxide onto hydrotalcite-like compounds (HTlcs) at high temperatures. Ind Eng Chem Res. 2001;40:204–9.
Reichle WT. Anionic clay minerals. CHEMTECH. 1986;16:58–63.
Vaccari A. Preparation and catalytic properties of cationic and anionic clays. Catal Today. 1998;41:53–71.
Lwin Y, Yarmo MA, Yaakob Z, Mohamad AB, Daud WRW. Synthesis and characterization of Cu–Al layered double hydroxides. Mater Res Bull. 2001;36:193–8.
Voyer N, Soisnard A, Palmer SJ, Martens WN, Frost RL. Thermal decomposition of the layered double hydroxides of formula Cu6Al2(OH)16CO3 and Zn6Al2(OH)16CO3. J Therm Anal Cal. 2009;96:481–5. doi: 10.1007/s10973-008-9169-x.
Lwin Y, Mohamad AB, Yaakob Z, Daud WRW. XRD and TPR studies of Cu–Al hydrotalcite derived highly dispersed mixed metal oxides. React Kinet Catal Lett. 2000;70:303–10.
Kannan S, Rives V, Knozinger H. High-temperature transformations of Cu-rich hydrotalcites. J Solid State Chem. 2004;177:319–31.
Mao G, Tsuji M, Tamaura Y. Synthesis and CO2 adsorption features of a hydrotalcite-like compound of the Mg2+–Al3+–Fe(CN)6 4-. Clays Clay Miner. 1993;41:731–7.
Tsuji M, Mao G, Tamaura Y. Hydrotalcites with an extended Al3+-substitution: synthesis, simultaneous TG-DTA-MS study, and their CO2 adsorption behaviors. J Mater Res. 1993;8:1137–42.
Palmer SJ, Spratt HJ, Frost RL. Thermal decomposition of hydrotalcites with variable cationic ratios. J Therm Anal Cal. 2009;95:123–9.
Kloprogge JT, Frost RL. Infrared emission spectroscopic study of the thermal transformation of Mg-, Ni- and Co-hydrotalcite catalysts. Appl Catal A. 1999;184:61–71.
Acknowledgments
Much of this work was performed in the Universiti Sains Malaysia, which supported financially through the Short-term Grant FPP 052-2003. The first author would also like to acknowledge the support provided by the Universiti Malaysia Perlis and the Universiti Teknologi PETRONAS in preparing this document.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lwin, Y., Abdullah, F. High temperature adsorption of carbon dioxide on Cu–Al hydrotalcite-derived mixed oxides: kinetics and equilibria by thermogravimetry. J Therm Anal Calorim 97, 885–889 (2009). https://doi.org/10.1007/s10973-009-0156-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-009-0156-7