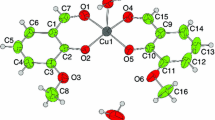
Abstract
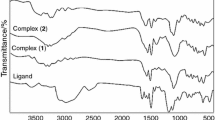
The thermal decomposition kinetics of sterically hindered salen type ligand (L) and its metal complexes [M=Co(II), Ni(II), Cu(II)] were investigated by thermogravimetric analysis. A direct insertion probe-mass spectrometer (DIP-MS) was used for the characterization of metal complexes of L and all fragmentations and stable ions were characterized. The thermogravimetry and differential thermogravimetry (TG-DTG) plots of salen type salicylaldimine ligand and complexes showed a single step.
The kinetic analysis of thermogravimetric data was performed by using the invariant kinetic parameter method (IKP). The values of the invariant activation energy, E inv and the invariant pre-exponential factor, A inv, were calculated by using Coats-Redfern (CR) method. The thermal stabilities and activation energies of metal complexes of sterically hindered salen type ligand (L) were found as Co(II)>Cu(II)>Ni(II)>L and E Cu>E Ni>E Co>L. Also, the probabilities of decomposition functions were investigated. The diffusion functions (D n) are most probable for the thermal decomposition of all complexes.
Similar content being viewed by others
References
W. Zhang, J. L. Loebach, S. R. Wilson and E. N. Jacobsen, J. Am. Chem. Soc., 112 (1990) 2801.
T. Matsuura, Tetrahedron, 33 (1977) 2869.
J. L. Serrano and L. Oriol, Adv. Mater., 7 (1995) 365.
L. Mao, K. Yamamato, W. Zhou and L. Jin, Electroanalysis, 12 (2000) 72.
J. F. Larrow, E. N. Jacobsen, Y. Gao, Y. Hong, X. Nie and C. M. Zepp, J. Org. Chem., 59 (1994) 1939.
J. Madarász, G. Pokol and S. Gál, J. Thermal Anal., 42 (1994) 539.
I. L. Lapides, J. Thermal Anal., 50 (1997) 269.
F. Carrasco, Thermochim. Acta, 213 (1993) 115.
M. V. Kök and N. Acar, J. Therm. Anal. Cal., 83 (2006) 445.
M. V. Kök, J. Therm. Anal. Cal., 88 (2007) 663.
M. V. Kök and A. G. Iscan, J. Therm. Anal. Cal., 88 (2007) 657.
J. Zsakó, J. Thermal Anal., 46 (1996) 1854.
A. A. Soliman, S. M. El-Medani and O. A. M. Ali, J. Therm. Anal. Cal., 83 (2006) 385.
S. Durmuş, Ü. Ergün, J. C. Jaud, K. C Emregül, H. Fues and O Atakol, J. Therm. Anal. Cal., 86 (2006) 337.
O. Z. Yeşilel, H. Ölmez and H. İçbudak, J. Therm. Anal. Cal., OnlineFirst DOI: 10.1007/s10973-005-7479-9.
G. G. Mohamed and H. Z. El-Wahab, J. Therm. Anal. Cal., 73 (2003) 347.
F. Doğan, S. Gülcemal, M. Yürekli and B. Çetinkaya, J. Therm. Anal. Cal., 91 (2008) 395.
V. T. Kasumov and F. Koksal, Spectrochim. Acta, Part A, 61A(1–2) (2004) 225.
V. T. Kasumov, S. O.-Yaman and E. Tas, Spectrochim. Acta, Part A, 62A(1–3) (2005) 716.
S. V Levchik, G. F. Levchik and A. L. Lesnikovich, Thermochim. Acta, 92 (1985) 157.
M. Arshad, Saeed-ur-Rehman, S. Ali Khan, K. Masud, N. Arshad and A. Ghani, Thermochim. Acta, 364 (2002) 143.
G. G. Mohamed, F. A. Nour-El Dien and E. A. El-Gamel, J. Therm. Anal. Cal., 67 (2002) 135.
F. Doğan, M. Ulusoy, Ö. F. Öztürk, I. Kaya and B. Salih, J. Therm. Anal. Cal., (2009), in print.
V. Mamleev, S. Bourbigot, M. Le Bras, S. Duquesne and J. Sešták, Phys. Chem. Chem. Phys., 2 (2000) 4708.
A. J. Lesnickovick and S. V Levchick, J. Thermal Anal., 30 (1985) 667.
A. W. Coats and J. P. Redfern, Nature, 201 (1964) 68.
A. J. Lesnickovick and S. V Levchick, J. Thermal Anal., 27 (1983) 83.
S. Bourbigot, R. Delobel, M. Le Bras and D. Normand, J. Chim. Phys., 90 (1993) 1909.
S. Bourbigot, X. Flambard and S. Duquesne, Polym. Int., 50 (2001) 157.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Doğan, F., Ulusoy, M., Öztürk, Ö.F. et al. Thermal studies of Co(II), Ni(II) and Cu(II) complexes of N,N′-bis(3,5-Di-t-butylsalicylidene)ethylenediamine. J Therm Anal Calorim 96, 267–276 (2009). https://doi.org/10.1007/s10973-008-8980-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-008-8980-8