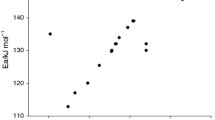
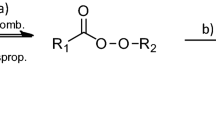
Abstract
The exothermic decomposition of cumene hydroperoxide (CHP) in cumene liquid was characterized by isothermal microcalorimetry, involving the thermal activity monitor (TAM). Unlike the exothermic behaviors previously determined from an adiabatic calorimeter, such as the vent sizing package 2 (VSP2), or differential scanning calorimetry (DSC), thermal curves revealed that CHP undergoes an autocatalytic decomposition detectable between 75 and 90°C. Previous studies have shown that the CHP in a temperature range higher than 100°C conformed to an n th order reaction rate model. CHP heat of decomposition and autocatalytic kinetics behavior were measured and compared with previous reports, and the methodology and the advantages of using the TAM to obtain an autocatalytic model by curve fitting are reported. With various autocatalytic models, such as the Prout-Tompkins equation and the Avrami-Erofeev rate law, the best curve fit among models was also investigated and proposed.
Similar content being viewed by others
References
NFPA 432, Code for the Storage of Organic Peroxide Formulations, National Fire Protection Association, Quincy, MA, USA 2000.
Y. S. Duh, C. C. Hsu, C. S. Kao and S. W. Yu, Thermochim. Acta, 285 (1997) 67.
Y. W. Wang, C. M. Shu, Y. S. Duh and C. S. Kao, Ind. Eng. Chem. Res., 40 (2001) 1125.
H. Y. Hou, Y. S. Duh, W. H. Lin and C. M. Shu, J. Therm. Anal. Cal., 85 (2006) 145.
C. M. Shu, Y. W. Wang, Y. S. Duh and C. S. Kao, The 1st International Conference on the Methodology of Reaction Hazards Investigation and Vent Sizing, St. Petersburg, Russia 1999, p. 15.
S. H. Wu, T. L. Tsai, H. Y. Hou, J. M. Tseng and C. M. Shu, 2006 Japan/Taiwan/Korea Chemical Engineering Conference, Japan 2006.
H. Y. Hou, C. M. Shu and Y. S. Duh, AIChE J., 47 (2001) 1893.
C. C. Chen, C. M. Shu, C. A. Yeh, S. C. Chen and M. L. Shyu, The 17th Annual Conference of Asia Pacific Occupational Safety and Health Organization, Taipei, Taiwan, ROC 2001, p. 295.
H. D. Ferguson, D. I. Towsend, T. C. Hofelich and P. M. Russel, J. Hazard. Mater., 37 (1994) 285.
STARe Software with Solaris Operating System, Operating Instructions; Mettler Toledo: Sweden 2004.
A. Miyake, A. Kimura, T. Ogawa, Y. Satoh and M. Inano, J. Therm. Anal. Cal., 80 (2005) 515.
E. Marti, E. Kaisersberger and W.-D. Emmerich, J. Therm. Anal. Cal., 77 (2004) 905.
S. P. Sivapirakasam, M. Surianarayanan, F. Chandrasekaran and G. Swaminathan, J. Therm. Anal. Cal., 78 (2004) 799.
D. Fessas, M. Signorelli and A. Schiraldi, J. Therm. Anal. Cal., 82 (2005) 691.
C. C. Liao, S. H. Wu, T. S. Su, M. L. Shyu and C. M. Shu, J. Therm. Anal. Cal., 85 (2006) 65.
Isothermal Calorimetric Manual for Thermometric AB, Jarfalla, Sweden 2007.
Guidelines for Chemical Reactivity Evaluation and Application to Process Design, AIChE, New York, USA 1995.
S. Chervin and G. T. Bodman, The 1st International Conference Methodology of Reaction Hazards Investigation and Vent Sizing, Saint Petersburg, Russia 1999, p. 1.
J. A. Barton and R. L. Rogers, Chemical Reaction Hazards-A Guide, 2nd Ed. UK 1995, p. 56.
Y. S. Duh, C. S. Kao, C. Lee and S. W. Yu, Trans. IChemE., 75 (1997) 73.
F. Brogli, P. Grimm, M. Meyer and H. Zubler, The 3rd International Symposium of Loss Prevention, Swiss Soc. Chem. Ind., Basel, Switzerland 1980, p. 655.
J. W. Beckmann, J. S. Wilwes and R. R. McGuire, Thermochim. Acta, 19 (1997) 111.
D. A. Frank-Kamenetskii, Diffusion and Heat Transfer in Chemical Kinetics, 2nd Ed., Moscow, USSR 1969, p. 301.
S. Chervin and G. T. Bodman, Process Saf. Prog., 16 (1997) 94.
I. D. Clark, L. G. Harrison, V. N. Kondratiev, Z. G. Szabo and R. P. Wayne, The Theory of Kinetics, Comprehensive Chemical Kinetics, Vol. 2, New York, USA 1969.
R. H. Chang, J. M. Tseng, J. M. Jehng, C. M. Shu and H. Y. Hou, J. Therm. Anal. Cal., 83 (2006) 57.
J. M. Tseng, R. H. Chang, J. J. Horng, M. K. Chang and C. M. Shu, J. Therm. Anal. Cal., 85 (2006) 189.
J. M. Tseng, Y. Y. Chang, T. S. Su and C. M. Shu, J. Hazard. Mater., 142 (2007) 765.
K. Y. Chen, S. H. Wu, Y. W. Wang and C. M. Shu, J. Loss Prev. Process Ind., 21 (2008) 101.
J. M. Tseng, R. H. Chang, J. J. Horng, M. K. Chang and C. M. Shu, J. Therm. Anal. Cal., 85 (2006) 189.
K. W. Wu, H. Y. Hou and C. M. Shu, J. Therm. Anal. Cal., 83 (2006) 57.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, J.R., Wu, S.H., Lin, S.Y. et al. Utilization of microcalorimetry for an assessment of the potential for a runaway decomposition of cumene hydroperoxide at low temperatures. J Therm Anal Calorim 93, 127–133 (2008). https://doi.org/10.1007/s10973-007-8834-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-007-8834-9