Summary
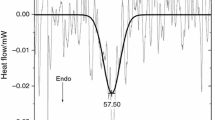
The effect of pH was characterised on the thermal stability of magnesium saturated skeletal and cardiac α-actin isoforms with differential scanning calorimetry (DSC) at pH 7.0 and 8.0. The calorimetric curves were further analysed to calculate the enthalpy and transition entropy changes. The activation energy was also determined to describe the energy consumption of the initiation of the thermal denaturation process. Although the difference in T mvalues is too small to interpret the difference between the a-actin isoforms, the values of the activation energy indicated that the α-skeletal actin is probably more stable compared to the α-cardiac actin. The difference in the activation energies indicated that lowering the pH can produce a more stable protein matrix in both cases of the isoforms. The larger range of the difference in the values of the activation energies suggested that the α-cardiac actin is probably more sensitive to the change of the pH compared to the α -skeletal actin.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Papp, G., Bugyi, B., Ujfalusi, Z. et al. The effect of pH ont he thermal stability of<span style='font-size:10.0pt;font-family:"SymbolProp BT";mso-bidi-font-family:"SymbolProp BT"'>a-actin isoforms. J Therm Anal Calorim 82, 281–285 (2005). https://doi.org/10.1007/s10973-005-0881-5
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-0881-5