Summary
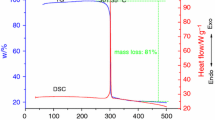
There has been recent interest in the hazard properties of mixtures of ammonium nitrate (AN) and the sodium salt of dichloroisocyanuric acid (SDIC) due to the possible involvement of such mixtures in the tragic accident in Toulouse, France, in September 2001. The thermal hazards of the mixtures were investigated using differential scanning calorimetry (DSC), accelerating rate calorimetry (ARC), thermogravimetry (TG), simultaneous TG-DTA-FTIR-MS, heat flow calorimetry (HFC), and isothermal nanocalorimetry (INC). The sensitivity of the mixtures to impact, friction, and electrostatic discharge was also investigated. ARC experiments on a 2 g mass of mixture in humid air revealed an onset temperature for thermal decomposition as low as 37°C. INC experiments revealed three overlapping exothermic peaks that resulted in a total energy release of 0.4 kJ g-1 over the course of thirteen days at 25°C. The reaction products were determined using simultaneous TG-DTA with FTIR and MS detectors, and they included CO2, HCl, N2, N2O, NO2 and Cl2. The results from this study suggest that accidental mixing of bulk quantities of these materials would pose a considerable hazard and should be avoided.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Badeen, C., Kwok, Q., Vachon, M. et al. Hazard characterization of mixtures of ammonium nitrate with the sodium salt of dichloroisocyanuric acid. J Therm Anal Calorim 81, 225–233 (2005). https://doi.org/10.1007/s10973-005-0771-x
Issue Date:
DOI: https://doi.org/10.1007/s10973-005-0771-x