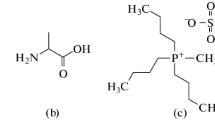
Abstract
Low-temperature heat capacity of unstable β-glycine was measured in a temperature range 5.5 to 295 K, and thermodynamic functions were calculated. At very low temperatures, heat capacity fits a sum of cubic (Debye) and linear terms: Cp=aT+bT 3. The linear contribution increases with temperature and disappears at the second-order phase transition near 252 K which was observed for the first time.
Similar content being viewed by others
References
V. A. Drebushchak, Yu. A. Kovalevskaya, I. E. Paukov and E. V. Boldyreva, J. Therm. Anal. Cal., 74 (2003) 109.
E. V. Boldyreva, V. A. Drebushchak, T. N. Drebushchak, I. E. Paukov, Y. A. Kovalevskaya and E. S. Shutova, J. Therm. Anal. Cal., 73 (2003) 409.
E. V. Boldyreva, V. A. Drebushchak, T. N. Drebushchak, I. E. Paukov, Y. A. Kovalevskaya and E. S. Shutova, J. Therm. Anal. Cal., 73 (2003) 419.
V. A. Drebushchak, E. V. Boldyreva, T. N. Drebushchak and E. S. Shutova, J. Crystal Growth, 241 (2002) 266.
E. V. Boldyreva, T. N. Drebushchak and E. S. Shutova, Z. Kristallogr., 218 (2003) 366.
G. L. Perlovich, L. K. Hansen and A. Bauer-Brandl, J. Therm. Anal. Cal., 66 (2001) 699.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Drebushchak, V.A., Boldyreva, E.V., Kovalevskaya, Y.A. et al. Low-temperature heat capacity of β-glycine and a phase transition at 252 K. J Therm Anal Calorim 79, 65–70 (2005). https://doi.org/10.1007/s10973-004-0563-8
Issue Date:
DOI: https://doi.org/10.1007/s10973-004-0563-8