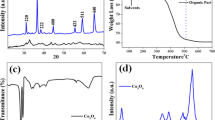
Abstract
Platinum (Pt) has been used in a wide range of catalytic applications such as oxygen-reduction reaction (ORR) in fuel cells. Key requirements for the applications are high dispersion and good anchorage of Pt nanoparticles on support materials. Metal ex-solution on perovskite oxide has been considered as a highly promising way to meet these demands, but Pt is extremely difficult to incorporate perovskite lattices due to its thermal unstability (oxygen loss). Here, we introduce a facile synthesis method mediated by NaNO3 to prepare Pt-doped CaTiO3. Use of amorphous TiO2 allows the particles to obtain spherical morphology (300 nm) and the high surface area (16 m2 g−1). The Pt ex-solved on CaTiO3 upon reducing atmosphere exhibit comparable catalytic activities for ORR in acidic media to conventional Pt/C catalysts. With good stability of titanate perovskites to acidic conditions, it would be highly applicable to various catalytic applications in redox environments.
Graphical Abstract

Highlights
-
Synthesis of Pt-doped CaTiO3 through a NaNO3-mediated fusion method.
-
Pt ions incorporated in the B-sites of the perovskite-structured CaTiO3.
-
Pt nanoparticles ex-solved from the perovskite-structured CaTiO3 upon reduction.
-
Compatible activity of 1 mol% Pt-ex-solved CaTiO3 catalysts to a commercial 40 wt.% Pt/C for oxygen-reduction reactions in acidic media.




Similar content being viewed by others
Data availability
The data and materials of this study are available from the corresponding author, [JGL], upon reasonable request.
References
Meille V(2006) Review on methods to deposit catalysts on structured surfaces. Appl Catal A Gen 315:1–17
Haruta M (1997) Size- and support-dependency in the catalysis of gold. Catal Today 36:153–166
Enache DI, Edwards JK, Landon P, Solsona-Espriu B, Carley AF, Herzing AA, Watanabe M, Kiely CJ, Knight DW, Hutchings GJ (2006) Solvent-free oxidation of primary alcohols to aldehydes using Au-Pd/ TiO2 catalysts. Science 311:362–365
Shchukin DG, Caruso RA (2004) Photocatalytic properties of porous metal oxide networks formed by nanoparticle infiltration in a polymer gel template. Chem Mater 16:2287–2292
Rolison DR, Long JW, Lytle JC, Fischer AE, Rhodes CP, McEvoy TM, Bourga ME, Lubers AM (2009) Multifunctional 3D nanoarchitectures for energy storage and conversion. Chem Soc Rev 38:226–252
Arico AS, Bruce P, Scrosati B, Tarascon JM, Van Schalkwijk W (2005) Nanostructured materials for advanced energy conversion and storage devices. Nat Mater 4:366–377
Rolison DR (2003) Catalytic nanoarchitectures-the importance of nothing and the unimportance of periodicity. Science 299:1698–1701
Irvine JTS, Neagu D, Verbraeken MC, Chatzichristodoulou C, Graves C, Mogensen MB (2016) Evolution of the electrochemical interface in high-temperature fuel cells and electrolysers. Nat Energy 1:15014
Starz KA, Auer E, Lehmann TH, Zuber R (1999) Characteristics of platinum-based electrocatalysts for mobile PEMFC. Appl J Power Sources 84:167–172
Chatterjee D, Deutschmann O, Warnatz J (2002) Detailed surface reaction mechanism in a three-way catalyst. Faraday Discuss 119:371–384
Horn R, Williams KA, Degenstein NJ, Bitsch-Larsen A, Nogare DD, Tupy SA, Schmidt LD (2007) Methane catalytic partial oxidation on autothermal Rh and Pt foam catalysts: oxidation and reforming zones, transport effects, and approach to thermodynamic equilibrium. J Catal 249:380–393
Roen LM, Paik CH, Jarvi TD (2004) Electrocatalytic corrosion of carbon support in PEMFC cathodes. Electrochem Solid State Lett A19:7
Wang J, Yin G, Shao Y, Zhang S, Wang Z, Gao Y (2007) Effect of carbon black support corrosion on the durability of Pt/C catalyst. J Power Sources 171:331–339
Maass S, Finsterwalder F, Frank G, Hartmann R, Merten C(2008) Carbon support oxidation in PEM fuel cell cathodes J Power Sources 176:444–451
Huang S, Ganesan P, Park S, Popov BN (2009) Development of a titanium dioxide-supported platinum catalyst with ultrahigh stability for polymer electrolyte membrane fuel cell applications. J Am Chem Soc 131(39):13898–13899
Goodenough JB, Cushing BL (2010). Oxide-based ORR catalysts. In Handbook of fuel cells. John Wiley & Sons, Ltd; 2010. https://doi.org/10.1002/9780470974001.f205040
Tanaka H, Taniguchi M, Kajita N, Uenishi M, Tan I, Sato N, Narita K, Kimura M(2004) Design of the intelligent catalyst for Japan ULEV standard Top Catal 30:389–396
Tanaka H (2005) An intelligent catalyst: the self-regenerative Palladium–Perovskite catalyst for automotive emissions control. Catal Surv Asia 9:63–74
Neagu D, Oh T–S, Miller DN, Ménard H, Bukhari SM, Gamble SR, Gorte RJ, Vohs JM, Irvine JTS (2015) Nano-socketed nickel particles with enhanced coking resistance grown in situ by redox exsolution. Nat Commun 6:8120
Neagu D, Tsekouras G, Miller DN, Ménard H, Irvine JTS (2013) In situ growth of nanoparticles through control of non-stoichiometry. Nat Chem 5(11):916–923
Neagu D, Papaioannou EI, Ramli WKW, Miller DN, Murdoch BJ, Ménard H, Umar A, Barlow AJ, Cumpson PJ, Irvine JTS, Metcalfe IS (2017) Demonstration of chemistry at a point through restructuring and catalytic activation at anchored nanoparticles. Nat Commun 8(1):1–8
Kothari M, Jeon Y, Miller DN, Pascui AE, Kilmartin J, Wails D, Ramos S, Chadwick A, Irvine JTS (2021) Platinum incorporation into titanate perovskites to deliver emergent active and stable platinum nanoparticles. Nat Chem 13:677–682
Seriani N, Pompe W, Ciacchi LC(2006) Catalytic oxidation activity of Pt3O4 surfaces and thin films J Phys Chem B 110(30):14860–14869
Turrillas X, Laviron C, Vincent H, Pannetier J, Joubert JC (1987) Synthesis and characterization of a new calcium platinum oxide: Ca2Pt3O8. J Solid State Chem 67:297–307
Abe Y, Kawamura M, Sasaki K (1999) Preparation of PtO and α-PtO2 thin films by reactive sputtering and their electrical properties. Jpn J Appl Phys 38:2092
Zhang S, Katz MB, Dai S, Zhang K, Du X, Graham GW, Pan X (2017) New atomic-scale insight into self-regeneration of Pt-CaTiO3 catalysts: incipient redox-induced structures revealed by a small-angle tilting STEM technique. J Phys Chem C 121(32):17348–17353
Lin C, Foucher AC, Ji Y, Curran CD, Stach EA, Mc Intosh S, Gorte RJ (2019) “Intelligent” Pt catalysts studied on high-surface-area CaTiO3 films. ACS Catal 9(8):7318–7327
Voorhees V, Adams R (1922) The use of the oxide of platinum for the catalytic reduction of organic compounds. I J Am Chem Soc 44(6):1397–1405
Lee JG, Myung J-H, Naden AB, Jeon OS, Shul YG, Irvine JTS (2020) Replacement of Ca by Ni in a perovskite titanate to yield a novel perovskite exsolution architecture for oxygen-evolution reactions. Adv Energy Mater 10:1903693
Acknowledgements
This work was supported by National Research Foundation of Korea (NRF) grant funded by Korean government (MSIT) (No. 2021R1A2C2092130).
Author information
Authors and Affiliations
Contributions
JGL outlined the research and conducted the experiments. JGL prepared the samples and analyzed materials characteristics. JGL wrote the manuscript and all authors have given approval to the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethics approval
This paper meets the ethical standards of this journal.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lee, J.G. NaNO3-mediated synthesis of Pt-doped titanate perovskites for oxygen-reduction reactions in acidic media. J Sol-Gel Sci Technol 108, 120–126 (2023). https://doi.org/10.1007/s10971-023-06188-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-023-06188-6