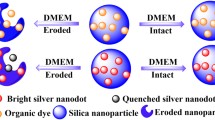
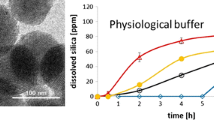
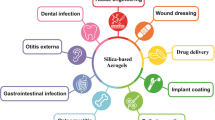
Abstract
This review shows the most common and promising strategies to generate colloidally stable silica nanoparticles (NPs) in simulated biological fluids and sheds light on the latest advances in producing degradable silica-based structures. Silica NPs can be synthesized in a wide variety of morphologies, porosity levels, and sizes. This versatility makes silica NPs one of the most promising nano-platforms for imaging and disease treatment. Nonetheless, biological barriers can decrease the success of translating them for therapeutic applications since the media composition can induce their colloidal stability loss. It can, consequently, lead to the NPs aggregation and affect their degradation profile. The interplay between NPs aggregation and degradation has been scarcely explored in the literature when biological fluids are seriously taken into account. Herein we discuss the theory behind the colloidal stability of silica NPs, the processes leading to their aggregation, and some strategies to overcome this issue (mainly focused on NPs surface functionalization). Furthermore, we addressed the main issues that affect the degradability of NPs in biological fluids, and explored some strategies, such as chemical surface modification, which are able to tune these degradation-driven profiles. Thus, the understanding of the silica NPs behavior in body fluids is essential for the approval of nanomedicines and, therefore, more investigations concerning the dynamics, thermodynamics, biological response, and structural parameters of silica-based NPs are of utmost importance.

Highlights
-
We discuss the challenges faced by silica-based nanoparticles in biological media.
-
We address relevant strategies to avoid aggregation of silica nanoparticles in biological fluids.
-
We present the impact of media and particle characteristics on silica nanoparticles degradation.
-
We discuss strategies used to tune silica nanoparticle degradability in the biological environment.








Similar content being viewed by others
References
Huang P, Chen Y, Lin H et al. (2017) Molecularly organic/inorganic hybrid hollow mesoporous organosilica nanocapsules with tumor-specific biodegradability and enhanced chemotherapeutic functionality. Biomaterials 125:23–37. https://doi.org/10.1016/j.biomaterials.2017.02.018
Croissant JG, Fatieiev Y, Almalik A, Khashab NM (2018) Mesoporous silica and organosilica nanoparticles: physical chemistry, biosafety, delivery strategies, and biomedical applications. Adv Health Mater 7:1700831. https://doi.org/10.1002/adhm.201700831
Maggini L, Cabrera I, Ruiz-Carretero A et al. (2016) Breakable mesoporous silica nanoparticles for targeted drug delivery. Nanoscale 8:7240–7247. https://doi.org/10.1039/C5NR09112H
Tayama M, Inose T, Yamauchi N et al. (2019) Fabrication and dual-modal imaging properties of quantum dot/silica core-shell particles with immobilized single-nanometer-sized gold nanoparticles. Colloids Surf A Physicochem Eng Asp 574:162–170. https://doi.org/10.1016/j.colsurfa.2019.04.055
Lei Q, Guo J, Noureddine A et al. (2020) Sol–gel‐based advanced porous silica materials for biomedical applications. Adv Funct Mater 30:1909539. https://doi.org/10.1002/adfm.201909539
Stöber W, Fink A, Bohn E (1968) Controlled growth of monodisperse silica spheres in the micron size range. J Colloid Interface Sci 26:62–69. https://doi.org/10.1016/0021-9797(68)90272-5
Bogush GH, Tracy MA, Zukoski CF (1988) Preparation of monodisperse silica particles: control of size and mass fraction. J Non Cryst Solids 104:95–106. https://doi.org/10.1016/0022-3093(88)90187-1
Liberman A, Mendez N, Trogler WC, Kummel AC (2014) Synthesis and surface functionalization of silica nanoparticles for nanomedicine. Surf Sci Rep 69:132–158. https://doi.org/10.1016/j.surfrep.2014.07.001
Beck JS, Vartuli JC, Roth WJ et al. (1992) A new family of mesoporous molecular sieves prepared with liquid crystal templates. J Am Chem Soc 114:10834–10843. https://doi.org/10.1021/ja00053a020
Kresge CT, Leonowicz ME, Roth WJ et al. (1992) Ordered mesoporous molecular sieves synthesized by a liquid-crystal template mechanism. Nature 359:710–712. https://doi.org/10.1038/359710a0
Tarn D, Ashley CE, Xue M et al. (2013) Mesoporous silica nanoparticle nanocarriers: biofunctionality and biocompatibility. Acc Chem Res 46:792–801. https://doi.org/10.1021/ar3000986
Tissot I, Reymond JP, Lefebvre F, Bourgeat-Lami E (2002) SiOH-functionalized polystyrene latexes. A step toward the synthesis of hollow silica nanoparticles. Chem Mater 14:1325–1331. https://doi.org/10.1021/cm0112441
Chen JF, Ding HM, Wang JX, Shao L (2004) Preparation and characterization of porous hollow silica nanoparticles for drug delivery application. Biomaterials 25:723–727. https://doi.org/10.1016/S0142-9612(03)00566-0
Iler RK (1979) The chemistry of silica: solubility, polymerization, colloid and surface properties, and biochemistry. John Wiley & Sons
Brinker CJ, Scherer GW (1990) Sol-gel science. Elsevier
Bergna HE, Roberts WO (2006) Colloidal silica: fundamentals and applications. surfactant science series. CRC Press, Taylor and Francis Group, LLC
Zou Y, Huang B, Cao L et al. (2021) Tailored mesoporous inorganic biomaterials: assembly, functionalization, and drug delivery engineering. Adv Mater 33:2005215. https://doi.org/10.1002/adma.202005215
Li Z, Mu Y, Peng C et al. (2021) Understanding the mechanisms of silica nanoparticles for nanomedicine. Wiley Interdiscip Rev Nanomed Nanobiotechnol 13:1–23. https://doi.org/10.1002/wnan.1658
Jiang S, Prozeller D, Pereira J et al. (2020) Controlling protein interactions in blood for effective liver immunosuppressive therapy by silica nanocapsules. Nanoscale 12:2626–2637. https://doi.org/10.1039/c9nr09879h
Hadipour Moghaddam SP, Mohammadpour R, Ghandehari H (2019) In vitro and in vivo evaluation of degradation, toxicity, biodistribution, and clearance of silica nanoparticles as a function of size, porosity, density, and composition. J Control Release 311–312:1–15. https://doi.org/10.1016/j.jconrel.2019.08.028
Zhu X, Vo C, Taylor M, Smith BR (2019) Non-spherical micro- and nanoparticles in nanomedicine. Mater Horiz 6:1094–1121. https://doi.org/10.1039/C8MH01527A
Picco AS, Mondo GB, Ferreira LF et al. (2021) Protein corona meets freeze-drying: overcoming the challenges of colloidal stability, toxicity, and opsonin adsorption. Nanoscale 13:753–762. https://doi.org/10.1039/d0nr06040b
Capeletti LB, Oliveira JFA, Loiola LMD et al. (2019) Gram‐negative bacteria targeting mediated by carbohydrate–carbohydrate interactions induced by surface‐modified nanoparticles. Adv Funct Mater 29:1904216. https://doi.org/10.1002/adfm.201904216
Lee JE, Lee N, Kim T et al. (2011) Multifunctional mesoporous silica nanocomposite nanoparticles for theranostic applications. Acc Chem Res 44:893–902. https://doi.org/10.1021/ar2000259
Guerrero-Martínez A, Pérez-Juste J, Liz-Marzán LM (2010) Recent progress on silica coating of nanoparticles and related nanomaterials. Adv Mater 22:1182–1195. https://doi.org/10.1002/adma.200901263
Rosen JE, Gu FX (2011) Surface functionalization of silica nanoparticles with cysteine: a low-fouling zwitterionic surface. Langmuir 27:10507–10513. https://doi.org/10.1021/la201940r
Croissant JG, Fatieiev Y, Khashab NM (2017) Degradability and clearance of silicon, organosilica, silsesquioxane, silica mixed oxide, and mesoporous silica nanoparticles. Adv Mater 29:1–51. https://doi.org/10.1002/adma.201604634
Ehrlich H, Demadis KD, Pokrovsky OS, Koutsoukos PG (2010) Modern views on desilicification: biosilica and abiotic silica dissolution in natural and artificial environments. Chem Rev 110:4656–4689. https://doi.org/10.1021/cr900334y
Bobo D, Robinson KJ, Islam J et al. (2016) Nanoparticle-based medicines: a review of FDA-approved materials and clinical trials to date. Pharm Res 33:2373–2387. https://doi.org/10.1007/s11095-016-1958-5
Lo TH, Wu ZY, Chen SY et al. (2021) Curcumin-loaded mesoporous silica nanoparticles with dual-imaging and temperature control inhibits the infection of Zika virus. Microporous Mesoporous Mater 314:110886. https://doi.org/10.1016/j.micromeso.2021.110886
Carvalho GC, Sábio RM, de Cássia Ribeiro T, et al. (2020) Highlights in mesoporous silica nanoparticles as a multifunctional controlled drug delivery nanoplatform for infectious diseases treatment. Pharm Res 37:191. https://doi.org/10.1007/s11095-020-02917-6
Ni D, Bu W, Ehlerding EB et al. (2017) Engineering of inorganic nanoparticles as magnetic resonance imaging contrast agents. Chem Soc Rev 46:7438–7468. https://doi.org/10.1039/c7cs00316a
Anselmo AC, Mitragotri S (2015) A review of clinical translation of inorganic nanoparticles. AAPS J 17:1041–1054. https://doi.org/10.1208/s12248-015-9780-2
Giri K, Kuschnerus I, Ruan J, Garcia‐Bennett AE (2019) Influence of a protein corona on the oral pharmacokinetics of testosterone released from mesoporous silica. Adv Ther 3:1900110. https://doi.org/10.1002/adtp.201900110
Moore TL, Rodriguez-Lorenzo L, Hirsch V et al. (2015) Nanoparticle colloidal stability in cell culture media and impact on cellular interactions. Chem Soc Rev 44:6287–6305. https://doi.org/10.1039/c4cs00487f
Tenzer S, Docter D, Kuharev J et al. (2013) Rapid formation of plasma protein corona critically affects nanoparticle pathophysiology. Nat Nanotechnol 8:772–781. https://doi.org/10.1038/nnano.2013.181
Graf C, Gao Q, Schütz I et al. (2012) Surface functionalization of silica nanoparticles supports colloidal stability in physiological media and facilitates internalization in cells. Langmuir 28:7598–613. https://doi.org/10.1021/la204913t
Hao N, Liu H, Li L et al. (2012) In vitro degradation behavior of silica nanoparticles under physiological conditions. J Nanosci Nanotechnol 12:6346–6354. https://doi.org/10.1166/jnn.2012.6199
Mortensen NP, Hurst GB, Wang W et al. (2013) Dynamic development of the protein corona on silica nanoparticles: composition and role in toxicity. Nanoscale 5:6372–6380. https://doi.org/10.1039/c3nr33280b
Cauda V, Argyo C, Bein T (2010) Impact of different PEGylation patterns on the long-term bio-stability of colloidal mesoporous silica nanoparticles. J Mater Chem 20:8693–8699. https://doi.org/10.1039/c0jm01390k
Khung YL, Narducci D (2015) Surface modification strategies on mesoporous silica nanoparticles for anti-biofouling zwitterionic film grafting. Adv Colloid Interface Sci 226:166–186. https://doi.org/10.1016/j.cis.2015.10.009
Tiemblo P, García N, Hoyos M et al. (2015) Organic modification of hydroxylated nanoparticles: silica, sepiolite, and polysaccharides. In: Aliofkhazraei M (ed) Handbook of nanoparticles. Springer International Publishing, Cham, p 1–35
Estephan ZG, Schlenoff PS, Schlenoff JB (2011) Zwitteration as an alternative to PEGylation. Langmuir 27:6794–6800. https://doi.org/10.1021/la200227b
Li Y, Hu X, Ding D et al. (2017) In situ targeted MRI detection of Helicobacter pylori with stable magnetic graphitic nanocapsules. Nat Commun 8:1–10. https://doi.org/10.1038/ncomms15653
de Oliveira JFA, da Silva RF, Ribeiro IRS et al. (2020) Selective targeting of lymphoma cells by monoclonal antibody grafted onto Zwitterionic-functionalized nanoparticles. Part Part Syst Charact 37:1–5. https://doi.org/10.1002/ppsc.201900446
Cheng W, Nie J, Xu L et al. (2017) pH-sensitive delivery vehicle based on folic acid-conjugated polydopamine-modified mesoporous silica nanoparticles for targeted cancer therapy. ACS Appl Mater Interfaces 9:18462–18473. https://doi.org/10.1021/acsami.7b02457
de Oliveira LF, Bouchmella K, Gonçalves KDA et al. (2016) Functionalized silica nanoparticles as an alternative platform for targeted drug-delivery of water insoluble drugs. Langmuir 32:3217–3225. https://doi.org/10.1021/acs.langmuir.6b00214
Del Pino P, Pelaz B, Zhang Q et al. (2014) Protein corona formation around nanoparticles – from the past to the future. Mater Horiz 1:301–313. https://doi.org/10.1039/C3MH00106G
Nel AE, Mädler L, Velegol D et al. (2009) Understanding biophysicochemical interactions at the nano–bio interface. Nat Mater 8:543–557. https://doi.org/10.1038/nmat2442
Schneid AC, Silveira CP, Galdino FE et al. (2020) Colloidal stability and redispersibility of mesoporous silica nanoparticles in biological media. Langmuir 36:11442–11449. https://doi.org/10.1021/acs.langmuir.0c01571
Braun K, Pochert A, Beck M et al. (2016) Dissolution kinetics of mesoporous silica nanoparticles in different simulated body fluids. J Sol-Gel Sci Technol 79:319–327. https://doi.org/10.1007/s10971-016-4053-9
Wohlleben W, Driessen MD, Raesch S et al. (2016) Influence of agglomeration and specific lung lining lipid/protein interaction on short-term inhalation toxicity. Nanotoxicology 10:970–980. https://doi.org/10.3109/17435390.2016.1155671
Francia V, Yang K, Deville S et al. (2019) Corona composition can affect the mechanisms cells use to internalize nanoparticles. ACS Nano 13:11107–11121. https://doi.org/10.1021/acsnano.9b03824
Auría-Soro C, Nesma T, Juanes-Velasco P, et al. (2019) Interactions of nanoparticles and biosystems: Microenvironment of nanoparticles and biomolecules in nanomedicine. Nanomaterials 9:1365. https://doi.org/10.3390/nano9101365
Israelachvili J (2013) Intermolecular and surface forces, 3rd edn. Academic Press
Boström M, Deniz V, Franks GV, Ninham BW (2006) Extended DLVO theory: electrostatic and non-electrostatic forces in oxide suspensions. Adv Colloid Interface Sci 123–126:5–15. https://doi.org/10.1016/j.cis.2006.05.001
Tadros T (2011) General principles of colloid stability and the role of surface forces. In: Tadros T (ed.) Colloid stability. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany, p 1–22
Parsegian VA (2005) Van Der Waals forces: a handbook for biologists, chemists, engineers, and physicists. Cambridge University Press
Bergström L (1997) Hamaker constants of inorganic materials. Adv Colloid Interface Sci 70:125–169. https://doi.org/10.1007/978-3-642-72624-8_11
Kontogeorgis GM, Kiil S (2016) Introduction to applied colloid and surface chemistry. John Wiley & Sons, Ltd, Chichester, UK
Parks GA, de Bruyn PL (1962) The zero point of charge of oxides. J Phys Chem 66:967–973. https://doi.org/10.1021/j100812a002
Adamson A, Gast A (1997) Physical chemistry of surfaces, 6th edn. John Wiley & Sons, Inc.
Kosmulski M (2009) Compilation of PZC and IEP of sparingly soluble metal oxides and hydroxides from literature. Adv Colloid Interface Sci 152:14–25. https://doi.org/10.1016/j.cis.2009.08.003
Parks GA (1965) The isoelectric points of solid oxides, solid hydroxides, and aqueous hydroxo complex systems. Chem Rev 65:177–198. https://doi.org/10.1021/cr60234a002
Kosmulski M (2016) Isoelectric points and points of zero charge of metal (hydr) oxides: 50 years after Parks’ review. Adv Colloid Interface Sci 238:1–61. https://doi.org/10.1016/j.cis.2016.10.005
Currie EPK, Norde W, Cohen Stuart MAC (2003) Tethered polymer chains: surface chemistry and their impact on colloidal and surface properties. Adv Colloid Interface Sci 100–102:205–265. https://doi.org/10.1016/S0001-8686(02)00061-1
Bharti B, Meissner J, Klapp SHL, Findenegg GH (2014) Bridging interactions of proteins with silica nanoparticles: the influence of pH, ionic strength and protein concentration. Soft Matter 10:718–28. https://doi.org/10.1039/c3sm52401a
Grasso D, Subramaniam K, Butkus M et al. (2002) A review of non-DLVO interactions in environmental colloidal systems. Rev Environ Sci Bio/Technol 1:17–38. https://doi.org/10.1023/A:1015146710500
Ninham BW (1999) On progress in forces since the DLVO theory. Adv Colloid Interface Sci 83:1–17. https://doi.org/10.1016/S0001-8686(99)00008-1
Sonnefeld J, Löbbus M, Vogelsberger W (2001) Determination of electric double layer parameters for spherical silica particles under application of the triple layer model using surface charge density data and results of electrokinetic sonic amplitude measurements. Colloids Surf A Physicochem Eng Asp 195:215–225. https://doi.org/10.1016/S0927-7757(01)00845-7
Hiemstra T, De Wit JC, Van Riemsdijk W (1989) Multisite proton adsorption modeling at the solid/solution interface of (hydr)oxides: a new approach. J Colloid Interface Sci 133:105–117. https://doi.org/10.1016/0021-9797(89)90285-3
Sverjensky DA (2005) Prediction of surface charge on oxides in salt solutions: revisions for 1:1 (M+L-) electrolytes. Geochim Cosmochim Acta 69:225–257. https://doi.org/10.1016/j.gca.2004.05.040
Leroy P, Devau N, Revil A, Bizi M (2013) Influence of surface conductivity on the apparent zeta potential of amorphous silica nanoparticles. J Colloid Interface Sci 410:81–93. https://doi.org/10.1016/j.jcis.2013.08.012
Allen LH, Matijevíc E, Meites L (1971) Exchange of Na+ for the silanolic protons of silica. J Inorg Nucl Chem 33:1293–1299. https://doi.org/10.1016/0022-1902(71)80423-2
Ong S, Zhao X, Eisenthal KB (1992) Polarization of water molecules at a charged interface: second harmonic studies of the silica/water interface. Chem Phys Lett 191:327–335. https://doi.org/10.1016/0009-2614(92)85309-X
Pfeiffer-Laplaud M, Costa D, Tielens F et al. (2015) Bimodal acidity at the amorphous silica/water interface. J Phys Chem C 119:27354–27362. https://doi.org/10.1021/acs.jpcc.5b02854
I-Ssuer C, Maciel GE (1996) Probing hydrogen bonding and the local environment of silanols on silica surfaces via nuclear spin cross polarization dynamics. J Am Chem Soc 118:401–406. https://doi.org/10.1021/ja951550d
West R, Whatley LS, Lake KJ (1961) Hydrogen bonding studies. V. The relative basicities of ethers, alkoxysilanes and siloxanes and the nature of the silicon-oxygen bond. J Am Chem Soc 83:761–764. https://doi.org/10.1021/ja01465a001
Depasse J, Watillon A (1970) The stability of amorphous colloidal silica. J Colloid Interface Sci 33:430–438. https://doi.org/10.1016/0021-9797(70)90235-3
Allen LH, Matijević E (1969) Stability of colloidal silica. J Colloid Interface Sci 31:287–296. https://doi.org/10.1016/0021-9797(69)90172-6
Vigil G, Xu Z, Steinberg S, Israelachvili J (1994) Interactions of silica surfaces. J Colloid Interface Sci 165:367–385. https://doi.org/10.1006/jcis.1994.1242
Adler JJ, Rabinovich YI, Moudgil BM (2001) Origins of the Non-DLVO force between glass surfaces in aqueous solution. J Colloid Interface Sci 237:249–258. https://doi.org/10.1006/jcis.2001.7466
Zhmud BV, Meurk A, Bergström L (1998) Evaluation of surface ionization parameters from AFM data. J Colloid Interface Sci 207:332–343. https://doi.org/10.1006/jcis.1998.5783
Schrader AM, Monroe JI, Sheil R et al. (2018) Surface chemical heterogeneity modulates silica surface hydration. Proc Natl Acad Sci USA 115:2890–2895. https://doi.org/10.1073/pnas.1722263115
He L, Hu Y, Wang M, Yin Y (2012) Determination of solvation layer thickness by a magnetophotonic approach. ACS Nano 6:4196–4202. https://doi.org/10.1021/nn3007288
Kobayashi M, Skarba M, Galletto P et al. (2005) Effects of heat treatment on the aggregation and charging of Stöber-type silica. J Colloid Interface Sci 292:139–147. https://doi.org/10.1016/j.jcis.2005.05.093
Harding RD (1971) Stability of silica dispersions. J Colloid Interface Sci 35:172–174. https://doi.org/10.1016/0021-9797(71)90203-7
Drescher D, Orts-Gil G, Laube G et al. (2011) Toxicity of amorphous silica nanoparticles on eukaryotic cell model is determined by particle agglomeration and serum protein adsorption effects. Anal Bioanal Chem 400:1367–1373. https://doi.org/10.1007/s00216-011-4893-7
Lesniak A, Fenaroli F, Monopoli MP et al. (2012) Effects of the presence or absence of a protein corona on silica nanoparticle uptake and impact on cells. ACS Nano 6:5845–5857. https://doi.org/10.1021/nn300223w
Izak-Nau E, Voetz M, Eiden S et al. (2013) Altered characteristics of silica nanoparticles in bovine and human serum: the importance of nanomaterial characterization prior to its toxicological evaluation. Part Fibre Toxicol 10:56. https://doi.org/10.1186/1743-8977-10-56
Docter D, Bantz C, Westmeier D et al. (2014) The protein corona protects against size- and dose-dependent toxicity of amorphous silica nanoparticles. Beilstein J Nanotechnol 5:1380–1392. https://doi.org/10.3762/bjnano.5.151
Halamoda-kenzaoui B, Ceridono M, Colpo P et al. (2015) Dispersion behaviour of silica nanoparticles in biological media and its influence on cellular uptake. PLoS One 10:e0141593. https://doi.org/10.1371/journal.pone.0141593
Saikia J, Yazdimamaghani M, Hadipour Moghaddam SP, Ghandehari H (2016) Differential protein adsorption and cellular uptake of silica nanoparticles based on size and porosity. ACS Appl Mater Interfaces 8:34820–34832. https://doi.org/10.1021/acsami.6b09950
Gollwitzer C, Bartczak D, Goenaga-Infante H et al. (2016) A comparison of techniques for size measurement of nanoparticles in cell culture medium. Anal Methods 8:5272–5282. https://doi.org/10.1039/C6AY00419A
Bharti B, Meissner J, Findenegg GH (2011) Aggregation of silica nanoparticles directed by adsorption of lysozyme. Langmuir 27:9823–9833. https://doi.org/10.1021/la201898v
Kumar S, Aswal VK, Kohlbrecher J (2011) SANS and UV-vis spectroscopy studies of resultant structure from lysozyme adsorption on silica nanoparticles. Langmuir 27:10167–10173. https://doi.org/10.1021/la201291k
Orts-Gil G, Natte K, Thiermann R et al. (2013) On the role of surface composition and curvature on biointerface formation and colloidal stability of nanoparticles in a protein-rich model system. Colloids Surf B Biointerfaces 108:110–119. https://doi.org/10.1016/j.colsurfb.2013.02.027
Galdino FE, Picco AS, Sforca ML et al. (2020) Effect of particle functionalization and solution properties on the adsorption of bovine serum albumin and lysozyme onto silica nanoparticles. Colloids Surf B Biointerfaces 186:110677. https://doi.org/10.1016/j.colsurfb.2019.110677
Monopoli MP, Walczyk D, Campbell A et al. (2011) Physical-chemical aspects of protein corona: relevance to in vitro and in vivo biological impacts of nanoparticles. J Am Chem Soc 133:2525–2534. https://doi.org/10.1021/ja107583h
Lara S, Perez-Potti A, Herda LM et al. (2018) Differential recognition of nanoparticle protein corona and modified low-density lipoprotein by macrophage receptor with collagenous structure. ACS Nano 12:4930–4937. https://doi.org/10.1021/acsnano.8b02014
Yamamoto E, Kuroda K (2016) Colloidal mesoporous silica nanoparticles. Bull Chem Soc Jpn 89:501–539. https://doi.org/10.1246/bcsj.20150420
Lin Y, Abadeer N, Haynes CL (2011) Stability of small mesoporous silica nanoparticles in biological media. Chem Commun 47:532–534. https://doi.org/10.1039/C0CC02923H
Rascol E, Daurat M, Da Silva A et al. (2017) Biological fate of Fe3O4 core-shell mesoporous silica nanoparticles depending on particle surface chemistry. Nanomaterials 7:162. https://doi.org/10.3390/nano7070162
Lin Y, Abadeer N, Hurley KR, Haynes CL (2011) Ultrastable, redispersible, small, and highly organomodified mesoporous silica nanotherapeutics. J Am Chem Soc 133:20444–20457
Cauda V, Schlossbauer A, Bein T (2010) Bio-degradation study of colloidal mesoporous silica nanoparticles: effect of surface functionalization with organo-silanes and poly(ethylene glycol). Microporous Mesoporous Mater 132:60–71. https://doi.org/10.1016/j.micromeso.2009.11.015
Monopoli MP, Åberg C, Salvati A, Dawson KA (2012) Biomolecular coronas provide the biological identity of nanosized materials. Nat Nanotechnol 7:779–786. https://doi.org/10.1038/nnano.2012.207
Walkey CD, Chan WCW (2012) Understanding and controlling the interaction of nanomaterials with proteins in a physiological environment. Chem Soc Rev 41:2780–2799. https://doi.org/10.1039/c1cs15233e
Vroman L, Adams A, Fischer G, Munoz P (1980) Interaction of high molecular weight kininogen, factor XII, and fibrinogen in plasma at interfaces. Blood 55:156–159. https://doi.org/10.1182/blood.V55.1.156.156
Wang L, Wang K, Santra S et al. (2006) Watching silica nanoparticles glow in the biological world. Anal Chem 78:646–654. https://doi.org/10.1021/ac0693619
Nakamura M (2010) Approaches to the biofunctionalization of spherical silica nanomaterials. In: Kumar CSSR (ed) Nanotechnologies for the life sciences. Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim, Germany
Bagwe RP, Hilliard LR, Tan W (2006) Surface modification of silica nanoparticles to reduce aggregation and nonspecific binding. Langmuir 22:4357–4362. https://doi.org/10.1021/la052797j
Jokerst JV, Lobovkina T, Zare RN, Gambhir SS (2011) Nanoparticle PEGylation for imaging and therapy. Nanomedicine 6:715–728. https://doi.org/10.2217/nnm.11.19
Xu H, Yan F, Monson EE, Kopelman R (2003) Room-temperature preparation and characterization of poly (ethylene glycol)-coated silica nanoparticles for biomedical applications. J Biomed Mater Res A 66:870–879. https://doi.org/10.1002/jbm.a.10057
Thierry B, Zimmer L, McNiven S et al. (2008) Electrostatic self-assembly of PEG copolymers onto porous silica nanoparticles. Langmuir 24:8143–8150. https://doi.org/10.1021/la8007206
Rio-Echevarria IM, Selvestrel F, Segat D et al. (2010) Highly PEGylated silica nanoparticles: “ready to use” stealth functional nanocarriers. J Mater Chem 20:2780–2787. https://doi.org/10.1039/b921735e
Díaz B, Sánchez-Espinel C, Arruebo M et al. (2008) Assessing methods for blood cell cytotoxic responses to inorganic nanoparticles and nanoparticle aggregates. Small 4:2025–2034. https://doi.org/10.1002/smll.200800199
Zhu Y, Fang Y, Borchardt L, Kaskel S (2011) PEGylated hollow mesoporous silica nanoparticles as potential drug delivery vehicles. Microporous Mesoporous Mater 141:199–206. https://doi.org/10.1016/j.micromeso.2010.11.013
Mosquera J, García I, Henriksen-Lacey M et al. (2019) Reducing protein corona formation and enhancing colloidal stability of gold nanoparticles by capping with silica monolayers. Chem Mater 31:57–61. https://doi.org/10.1021/acs.chemmater.8b04647
Perdoor SS, Dubois F, Barbara A et al. (2020) Ultrabright silica-coated organic nanocrystals for two-photon in vivo imaging. ACS Appl Nano Mater 3:11933–11944. https://doi.org/10.1021/acsanm.0c02499
Keefe AJ, Jiang S (2012) Poly(zwitterionic)protein conjugates offer increased stability without sacrificing binding affinity or bioactivity. Nat Chem 4:59–63. https://doi.org/10.1038/nchem.1213
García KP, Zarschler K, Barbaro L et al. (2014) Zwitterionic-coated “stealth” nanoparticles for biomedical applications: Recent advances in countering biomolecular corona formation and uptake by the mononuclear phagocyte system. Small 10:2516–2529. https://doi.org/10.1002/smll.201303540
Schlenoff JB (2014) Zwitteration: coating surfaces with zwitterionic functionality to reduce non specific adsorption. Langmuir 30:9625–9636. https://doi.org/10.1021/la500057j
Knop K, Hoogenboom R, Fischer D, Schubert US (2010) Poly(ethylene glycol) in drug delivery: pros and cons as well as potential alternatives. Angew Chem Int Ed 49:6288–6308. https://doi.org/10.1002/anie.200902672
Panja P, Das P, Mandal K, Jana NR (2017) Hyperbranched polyglycerol grafting on the surface of silica-coated nanoparticles for high colloidal stability and low nonspecific interaction. ACS Sustain Chem Engeneering 5:4879–4889. https://doi.org/10.1021/acssuschemeng.7b00292
Liu J, Pelton R, Hrymak AN (2000) Properties of poly(N-isopropylacrylamide)-grafted colloidal silica. J Colloid Interface Sci 227:408–411. https://doi.org/10.1006/jcis.2000.6871
Schild HG (1992) Poly(N-isopropylacrylamide): experiment, theory and application. Prog Polym Sci 17:163–249. https://doi.org/10.1016/0079-6700(92)90023-R
Muller P (1994) Glossary of terms used in physical organic chemistry (IUPAC Recommendations 1994). Pure Appl Chem 66:1077–1184. https://doi.org/10.1351/pac199466051077
Estephan ZG, Jaber JA, Schlenoff JB (2010) Zwitterion-stabilized silica nanoparticles: toward nonstick nano. Langmuir 26:16884–16889. https://doi.org/10.1021/la103095d
Hayward JA, Chapman D (1984) Biomembrane surfaces as models for polymer design: the potential for haemocompatibility. Biomaterials 5:135–142. https://doi.org/10.1016/0142-9612(84)90047-4
Ishihara K, Aragaki R, Ueda T et al. (1990) Reduced thrombogenicity of polymers having phospholipid polar groups. J Biomed Mater Res 24:1069–1077. https://doi.org/10.1002/jbm.820240809
Ishihara K, Ziats NP, Tierney BP et al. (1991) Protein adsorption from human plasma is reduced on phospholipid polymers. J Biomed Mater Res 25:1397–1407. https://doi.org/10.1002/jbm.820251107
Knowles BR, Wagner P, Maclaughlin S et al. (2017) Silica nanoparticles functionalized with Zwitterionic sulfobetaine siloxane for application as a versatile antifouling coating system. ACS Appl Mater Interfaces 9:18584–18594. https://doi.org/10.1021/acsami.7b04840
Knowles BR, Yang D, Wagner P et al. (2019) Zwitterion functionalized silica nanoparticle coatings: the effect of particle size on protein, bacteria, and fungal spore adhesion. Langmuir 35:1335–1345. https://doi.org/10.1021/acs.langmuir.8b01550
Loiola LMD, Batista M, Capeletti LB et al. (2019) Shielding and stealth effects of zwitterion moieties in double-functionalized silica nanoparticles. J Colloid Interface Sci 553:540–548. https://doi.org/10.1016/j.jcis.2019.06.044
Hu F, Chen K, Xu H, Gu H (2015) Functional short-chain zwitterion coated silica nanoparticles with antifouling property in protein solutions. Colloids Surf B Biointerfaces 126:251–256. https://doi.org/10.1016/j.colsurfb.2014.12.036
Knowles BR, Wagner P, Maclaughlin S et al. (2020) Carboxybetaine functionalized nanosilicas as protein resistant surface coatings. Biointerphases 15:011001. https://doi.org/10.1063/1.5126467
Wang H, Cheng F, Shen W et al. (2016) Amino acid-based anti-fouling functionalization of silica nanoparticles using divinyl sulfone. Acta Biomater 40:273–281. https://doi.org/10.1016/j.actbio.2016.03.035
Shahabi S, Treccani L, Dringen R, Rezwan K (2015) Modulation of silica nanoparticle uptake into human osteoblast cells by variation of the ratio of amino and sulfonate surface groups: effects of serum. ACS Appl Mater Interfaces 7:13821–13833. https://doi.org/10.1021/acsami.5b01900
Encinas N, Angulo M, Astorga C et al. (2019) Mixed-charge pseudo-zwitterionic mesoporous silica nanoparticles with low-fouling and reduced cell uptake properties. Acta Biomater 84:317–327. https://doi.org/10.1016/j.actbio.2018.12.012
Scheffer FR, Silveira CP, Morais J et al. (2020) Tailoring pseudo-zwitterionic bifunctionalized silica nanoparticles: from colloidal stability to biological interactions. Langmuir 36:10756–10763. https://doi.org/10.1021/acs.langmuir.0c01545
Sun JT, Yu ZQ, Hong CY, Pan CY (2012) Biocompatible zwitterionic sulfobetaine copolymer-coated mesoporous silica nanoparticles for temperature-responsive drug release. Macromol Rapid Commun 33:811–818. https://doi.org/10.1002/marc.201100876
Runser A, Dujardin D, Ernst P et al. (2020) Zwitterionic stealth dye-loaded polymer nanoparticles for intracellular imaging. ACS Appl Mater Interfaces 12:117–125. https://doi.org/10.1021/acsami.9b15396
Dong Z, Mao J, Yang M et al. (2011) Phase behavior of poly(sulfobetaine methacrylate)-grafted silica nanoparticles and their stability in protein solutions. Langmuir 27:15282–15291. https://doi.org/10.1021/la2038558
Zhu Y, Sundaram HS, Liu S et al. (2014) A robust graft-to strategy to form multifunctional and stealth zwitterionic polymer-coated mesoporous silica nanoparticles. Biomacromolecules 15:1845–1851. https://doi.org/10.1021/bm500209a
Chen K, Hu F, Gu H, Xu H (2017) Tuning of surface protein adsorption by spherical mixed charged silica brushes (MCB) with zwitterionic carboxybetaine component. J Mater Chem B 5:435–443. https://doi.org/10.1039/c6tb02817a
Jia G, Cao Z, Xue H et al. (2009) Novel zwitterionic-polymer-coated silica nanoparticles. Langmuir 25:3196–3199. https://doi.org/10.1021/la803737c
Matsuda Y, Kobayashi M, Annaka M et al. (2008) Dimensions of a free linear polymer and polymer immobilized on silica nanoparticles of a zwitterionic polymer in aqueous solutions with various ionic strengths. Langmuir 24:8772–8778. https://doi.org/10.1021/la8005647
Sanchez-Salcedo S, Vallet-Regí M, Shahin SA et al. (2018) Mesoporous core-shell silica nanoparticles with anti-fouling properties for ovarian cancer therapy. Chem Eng J 340:114–124. https://doi.org/10.1016/j.cej.2017.12.116
Safavi-Sohi R, Maghari S, Raoufi M et al. (2016) Bypassing protein corona issue on active targeting: Zwitterionic coatings dictate specific interactions of targeting moieties and cell receptors. ACS Appl Mater Interfaces 8:22808–22818. https://doi.org/10.1021/acsami.6b05099
Pálmai M, Nagy LN, Mihály J et al. (2013) Preparation, purification, and characterization of aminopropyl-functionalized silica sol. J Colloid Interface Sci 390:34–40. https://doi.org/10.1016/j.jcis.2012.09.025
Picco AS, Ferreira LF, Liberato MS et al. (2018) Freeze-drying of silica nanoparticles: Redispersibility toward nanomedicine applications. Nanomedicine 13:16–18. https://doi.org/10.2217/nnm-2017-0280
Du X, Kleitz F, Li X et al. (2018) Disulfide-bridged organosilica frameworks: designed, synthesis, redox-triggered biodegradation, and nanobiomedical applications. Adv Funct Mater 28:1707325. https://doi.org/10.1002/adfm.201707325
Fuentes C, Ruiz-Rico M, Fuentes A et al. (2020) Degradation of silica particles functionalised with essential oil components under simulated physiological conditions. J Hazard Mater 399:123120. https://doi.org/10.1016/j.jhazmat.2020.123120
Liu L, Kong C, Huo M et al. (2018) Schiff base interaction tuned mesoporous organosilica nanoplatforms with pH-responsive degradability for efficient anti-cancer drug delivery in vivo. Chem Commun 54:9190–9193. https://doi.org/10.1039/C8CC05043K
Croissant JG, Fatieiev Y, Julfakyan K et al. (2016) Biodegradable oxamide-phenylene-based mesoporous organosilica nanoparticles with unprecedented drug payloads for delivery in cells. Chemistry 22:14806–14811. https://doi.org/10.1002/chem.201601714
Fei W, Chen D, Tang H et al. (2020) Targeted GSH-exhausting and hydroxyl radical self-producing manganese–silica nanomissiles for MRI guided ferroptotic cancer therapy. Nanoscale 12:16738–16754. https://doi.org/10.1039/D0NR02396E
Yang G, Phua SZF, Bindra AK, Zhao Y (2019) Degradability and clearance of inorganic nanoparticles for biomedical applications. Adv Mater 31:e1805730. https://doi.org/10.1002/adma.201805730
Dietzel M (2000) Dissolution of silicates and the stability of polysilicic acid. Geochim Cosmochim Acta 64:3275–3281. https://doi.org/10.1016/S0016-7037(00)00426-9
He Q, Zhang Z, Gao Y et al. (2009) Intracellular localization and cytotoxicity of spherical mesoporous silica nano- and microparticles. Small 5:2722–2729. https://doi.org/10.1002/smll.200900923
Popplewell J, King S, Day J et al. (1998) Kinetics of uptake and elimination of silicic acid by a human subject: a novel application of 32Si and accelerator mass spectrometry. J Inorg Biochem 69:177–180. https://doi.org/10.1016/S0162-0134(97)10016-2
Ogura M, Shinomiya S, Tateno J et al. (2001) Alkali-treatment technique—new method for modification of structural and acid-catalytic properties of ZSM-5 zeolites. Appl Catal A Gen 219:33–43. https://doi.org/10.1016/S0926-860X(01)00645-7
Rimer JD, Trofymluk O, Navrotsky A et al. (2007) Kinetic and thermodynamic studies of silica nanoparticle dissolution. Chem Mater 19:4189–4197. https://doi.org/10.1021/cm070708d
Etienne M, Walcarius A (2003) Analytical investigation of the chemical reactivity and stability of aminopropyl-grafted silica in aqueous medium. Talanta 59:1173–1188. https://doi.org/10.1016/S0039-9140(03)00024-9
Nangia S, Garrison BJ (2008) Reaction rates and dissolution mechanisms of quartz as a function of pH. J Phys Chem A 112:2027–2033. https://doi.org/10.1021/jp076243w
Nangia S, Garrison BJ (2009) Ab initio study of dissolution and precipitation reactions from the edge, kink, and terrace sites of quartz as a function of pH. Mol Phys 107:831–843. https://doi.org/10.1080/00268970802665621
Kosmulski M (2011) The pH-dependent surface charging and points of zero charge. J Colloid Interface Sci 353:1–15. https://doi.org/10.1016/j.jcis.2010.08.023
Kosmulski M (2018) The pH dependent surface charging and points of zero charge. VII. Update. Adv Colloid Interface Sci 251:115–138. https://doi.org/10.1016/j.cis.2017.10.005
Ratirotjanakul W, Suteewong T, Polpanich D, Tangboriboonrat P (2019) Amino acid as a biodegradation accelerator of mesoporous silica nanoparticles. Microporous Mesoporous Mater 282:243–251. https://doi.org/10.1016/j.micromeso.2019.02.033
Choi E, Kim S (2019) Surface pH buffering to promote degradation of mesoporous silica nanoparticles under a physiological condition. J Colloid Interface Sci 533:463–470. https://doi.org/10.1016/j.jcis.2018.08.088
Yang Y, Wan J, Niu Y et al. (2016) Structure-dependent and glutathione-responsive biodegradable dendritic mesoporous organosilica nanoparticles for safe protein delivery. Chem Mater 28:9008–9016. https://doi.org/10.1021/acs.chemmater.6b03896
Shen D, Yang J, Li X et al. (2014) Biphase stratification approach to three-dimensional dendritic biodegradable mesoporous silica nanospheres. Nano Lett 14:923–932. https://doi.org/10.1021/nl404316v
Yamada H, Urata C, Aoyama Y et al. (2012) Preparation of colloidal mesoporous silica nanoparticles with different diameters and their unique degradation behavior in static aqueous systems. Chem Mater 24:1462–1471. https://doi.org/10.1021/cm3001688
Hadipour Moghaddam SP, Saikia J, Yazdimamaghani M, Ghandehari H (2017) Redox-responsive polysulfide-based biodegradable organosilica nanoparticles for delivery of bioactive agents. ACS Appl Mater Interfaces 9:21133–21146. https://doi.org/10.1021/acsami.7b04351
Möller K, Bein T (2019) Degradable drug carriers: vanishing mesoporous silica nanoparticles. Chem Mater 31:4364–4378. https://doi.org/10.1021/acs.chemmater.9b00221
Choi E, Lim D-K, Kim S (2020) Hydrolytic surface erosion of mesoporous silica nanoparticles for efficient intracellular delivery of cytochrome c. J Colloid Interface Sci 560:416–425. https://doi.org/10.1016/j.jcis.2019.10.100
Yang SA, Choi S, Jeon SM, Yu J (2018) Silica nanoparticle stability in biological media revisited. Sci Rep 8:1–9. https://doi.org/10.1038/s41598-017-18502-8
Quignard S, Masse S, Laurent G, Coradin T (2013) Introduction of disulfide bridges within silica nanoparticles to control their intra-cellular degradation. Chem Commun 49:3410–3412. https://doi.org/10.1039/c3cc41062e
Yu L, Chen Y, Wu M et al. (2016) “Manganese extraction” strategy enables tumor-sensitive biodegradability and theranostics of nanoparticles. J Am Chem Soc 138:9881–9894. https://doi.org/10.1021/jacs.6b04299
Sha Z, Yang S, Fu L et al. (2021) Manganese-doped gold core mesoporous silica particles as a nanoplatform for dual-modality imaging and chemo-chemodynamic combination osteosarcoma therapy. Nanoscale 13:5077–5093. https://doi.org/10.1039/d0nr09220g
Marques MRC, Loebenberg R, Almukainzi M (2011) Simulated biological fluids with possible application in dissolution testing. Dissolution Technol 18:15–28. https://doi.org/10.14227/DT180311P15
Yang W, Tam J, Miller DA et al. (2008) High bioavailability from nebulized itraconazole nanoparticle dispersions with biocompatible stabilizers. Int J Pharm 361:177–188. https://doi.org/10.1016/j.ijpharm.2008.05.003
Dong R, Liu Y, Zhou Y et al. (2011) Photo-reversible supramolecular hyperbranched polymer based on host–guest interactions. Polym Chem 2:2771. https://doi.org/10.1039/c1py00426c
Wang D, Xu Z, Chen Z et al. (2014) Fabrication of single-hole glutathione-responsive degradable hollow silica nanoparticles for drug delivery. ACS Appl Mater Interfaces 6:12600–12608. https://doi.org/10.1021/am502585x
Tang H, Li C, Zhang Y et al. (2020) Targeted manganese doped silica nano GSH-cleaner for treatment of liver cancer by destroying the intracellular redox homeostasis. Theranostics 10:9865–9887. https://doi.org/10.7150/thno.46771
Maggini L, Travaglini L, Cabrera I et al. (2016) Biodegradable peptide-silica nanodonuts. Chemistry 22:3697–3703. https://doi.org/10.1002/chem.201504605
Croissant J, Cattoën X, Man MWC et al. (2014) Biodegradable ethylene-bis(propyl)disulfide-based periodic mesoporous organosilica nanorods and nanospheres for efficient in-vitro drug delivery. Adv Mater 26:6174–6180. https://doi.org/10.1002/adma.201401931
Fatieiev Y, Croissant JG, Julfakyan K et al. (2015) Enzymatically degradable hybrid organic–inorganic bridged silsesquioxane nanoparticles for in vitro imaging. Nanoscale 7:15046–15050. https://doi.org/10.1039/C5NR03065J
Croissant JG, Mauriello-Jimenez C, Maynadier M et al. (2015) Synthesis of disulfide-based biodegradable bridged silsesquioxane nanoparticles for two-photon imaging and therapy of cancer cells. Chem Commun 51:12324–12327. https://doi.org/10.1039/C5CC03736K
Xu Z, Zhang K, Liu X, Zhang H (2013) A new strategy to prepare glutathione responsive silica nanoparticles. RSC Adv 3:17700. https://doi.org/10.1039/c3ra43098g
Tang H, Chen D, Li C et al. (2019) Dual GSH-exhausting sorafenib loaded manganese-silica nanodrugs for inducing the ferroptosis of hepatocellular carcinoma cells. Int J Pharm 572:118782. https://doi.org/10.1016/j.ijpharm.2019.118782
Li XW, Zhao WR, Liu YJ et al. (2016) Facile synthesis of manganese silicate nanoparticles for pH/GSH-responsive T1-weighted magnetic resonance imaging. J Mater Chem B 4:4313–4321. https://doi.org/10.1039/C6TB00718J
Fontecave T, Sanchez C, Azaïs T, Boissière C (2012) Chemical modification as a versatile tool for tuning stability of silica based mesoporous carriers in biologically relevant conditions. Chem Mater 24:4326–4336. https://doi.org/10.1021/cm302142k
Kempen PJ, Greasley S, Parker KA et al. (2015) Theranostic mesoporous silica nanoparticles biodegrade after pro-survival drug delivery and ultrasound/magnetic resonance imaging of stem cells. Theranostics 5:631–642. https://doi.org/10.7150/thno.11389
Ryabchikova EI, Chelobanov BP, Parkhomenko RG et al. (2017) Degradation of core-shell Au@SiO 2 nanoparticles in biological media. Microporous Mesoporous Mater 248:46–53. https://doi.org/10.1016/j.micromeso.2017.04.006
Prasetyanto EA, Bertucci A, Septiadi D et al. (2016) Breakable hybrid organosilica nanocapsules for protein delivery. Angew Chem Int Ed 55:3323–3327. https://doi.org/10.1002/anie.201508288
Lu J, Liong M, Li Z et al. (2010) Biocompatibility, biodistribution, and drug-delivery efficiency of mesoporous silica nanoparticles for cancer therapy in animals. Small 6:1794–1805. https://doi.org/10.1002/smll.201000538
Bindini E, Ramirez M, de los A, Rios X et al. (2021) In vivo tracking of the degradation of mesoporous silica through 89Zr radio-labeled core–shell nanoparticles. Small 2101519:1–10. https://doi.org/10.1002/smll.202101519
Bhavsar D, Patel V, Sawant K (2019) Systemic investigation of in vitro and in vivo safety, toxicity and degradation of mesoporous silica nanoparticles synthesized using commercial sodium silicate. Microporous Mesoporous Mater 284:343–352. https://doi.org/10.1016/j.micromeso.2019.04.050
Allen LH, Matijević E (1970) Stability of colloidal silica II. Ion exchange. J Colloid Interface Sci 33:420–429. https://doi.org/10.1016/0021-9797(70)90234-1
Acknowledgements
The authors also acknowledge the financial support of the Fundação de Amparo à Pesquisa do Estado de São Paulo (processes 2018/00763-8, 2019/24894-7, 2016/16905-0, 2020/00767-3, and 2015/25406-5). ASP and MBC acknowledge UNLP and CONICET for their support. ASP and MC staff members of CONICET.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
da Cruz Schneid, A., Albuquerque, L.J.C., Mondo, G.B. et al. Colloidal stability and degradability of silica nanoparticles in biological fluids: a review. J Sol-Gel Sci Technol 102, 41–62 (2022). https://doi.org/10.1007/s10971-021-05695-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-021-05695-8