Abstract
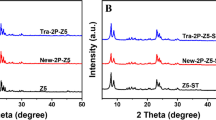
A mesoporous titania material with a single X-ray diffraction signal at small-angle and anatase TiO2 walls was prepared by a sol–gel method using hexadecylamine as a templating agent and titanium isopropoxide as a titanium precursor. The mesoporous solid was used to prepare a novel layered titanium phosphate by Soxhlet extraction reflux with phosphoric acid. The obtained α-TiP material showed a typical wide-angle X-ray diffraction pattern with a monoclinic structure, N2 adsorption–desorption isotherms Type II, and large crystals of rectangular lamellae, confirming its layered nature by transmission electron microscopy. The elimination by adsorption of Hg2+ ions from aqueous samples was studied using Langmuir, Freundlich and Temkin models, calculating its different parameters. The adsorption of Hg2+ ions could be performed via active P-OH groups (observed by Fourier Transform Infrared results) in the lamellar structure showing excellent properties that make the layered material possible excellent adsorbent to remove heavy metals ions contaminants from potable water.

Highlights
-
The synthesis of a novel layered titanium phosphate has been studied.
-
A mesoporous titania prepared by sol–gel method was used as titanium source.
-
The successful formation of the layered titanium phosphate in the alpha phase was confirmed by several characterization techniques.
-
The adsorption experimental data for Hg2+ uptake from aqueous solutions were analyzed using the Langmuir, Freundlich, and Temkin models.
-
The results propose the material might be an efficient adsorbent for wastewater treatment and water purification.






Similar content being viewed by others
References
Roy Chowdhury A, Datta R, Sarkar D (2018) Heavy metal pollution and remediation. In: Török, B, Dransfield, T (eds) Green chemistry: an inclusive approach, Elsevier, Ámsterdam, pp 359–373. https://doi.org/10.1016/B978-0-12-809270-5.00015-7
Gautam RK, Sharma SK, Mahiya S, Chattopadhyaya MC (2014) Contamination of heavy metals in aquatic media: transport, toxicity and technologies for remediation. In: Sharma SK (ed) Heavy metals in water: presence, removal and safety, Royal Chemistry Society, United Kingdom, pp 1–24. https://doi.org/10.1039/9781782620174-00001
Xia M, Chen Z, Li Y, Li C, Ahmad NM, Cheema WA, Zhu S (2019) Removal of Hg(II) in aqueous solutions through physical and chemical adsorption principles. RSC Adv 9:20941–20953. https://doi.org/10.1039/c9ra01924c
Malm O (1998) Gold mining as a source of mercury exposure in the Brazilian Amazon. Environ Res 77:73–78. https://doi.org/10.1006/enrs.1998.3828
Daza L, Mendioroz S, Pajares JA (1991) Mercury adsorption by sulfurized fibrous silicates. Clays Clay Miner 39:14–21. https://doi.org/10.1346/CCMN.1991.0390102
Al-Ghouti MA, Da’ana D, Abu-Dieyeh M, Khraisheh M (2019) Adsorptive removal of mercury from water by adsorbents derived from date pits. Sci Rep 9:15327. https://doi.org/10.1038/s41598-019-51594-y
Musmarra D, Karatza D, Lancia A, Prisciandaro M, Mazziotti, Di-Celso G (2015) A comparison among different sorbents for mercury adsorption from flue gas. Chem Eng Trans 43:2461–2466. https://doi.org/10.3303/CET1543411
Rungnim C, Promarak V, Hannongbua S, Kungwan N, Namuangruk S (2016) Complete reaction mechanisms of mercury oxidation on halogenated activated carbon. J Hazard Mater 310:253–260. https://doi.org/10.1016/j.jhazmat.2016.02.033
Khunphonoi R, Khamdahsag P, Chiarakorn S, Grisdanurak N, Paerungruang A, Predapitakkun S (2015) Enhancement of elemental mercury adsorption by silver supported material. J Environ Sci 32:207–216. https://doi.org/10.1016/j.jes.2015.01.008
Xie J, Xu H, Qu Z, Huang W, Chen W, Ma Y, Zhao S, Liu P, Yan N (2014) Sn-Mn binary metal oxides as non-carbon sorbent for mercury removalin a wide-temperature window. J Colloid Interface Sci 428:121–127. https://doi.org/10.1016/j.jcis.2014.04.032
Wdowin M, Macherzynski M, Panek R, Górecki J, Franus W (2015) Investigation of the sorption of mercury vapour from exhaust gas by an Ag-X-zeolite. Clay Miner 50:31–40. https://doi.org/10.1180/claymin.2015.050.1.04
Guerra DL, Santos MRMC, Airoldi C (2009) Mercury adsorption on natural and organofunctionalized smectites: thermodynamics of cation removal. J Braz Chem Soc 20:594–603. https://doi.org/10.1590/S0103-50532009000400003
Shen B, Chen J, Yue S (2015) Removal of elemental mercury by titanium pillared clay impregnated with potassium iodine. Micropor Mesopor Mater 203:216–223. https://doi.org/10.1016/j.micromeso.2014.10.030
Clearfield A, Stynes JA (1964) The preparation of crystalline zirconium phosphate and some observations on its ion exchange behavior. J Inorg Nucl Chem 26:117–129. https://doi.org/10.1016/0022-1902(64)80238-4
Alberti G, Torracca E (1968) Crystalline insoluble salts of polybasic metals. Part II: synthesis of crystalline zirconium or titanium phosphate by direct precipitation. J Inorg Nucl Chem 30:317–318. https://doi.org/10.1016/0022-1902(68)80096-X
Clearfield A, Blessing RH, Stynes JA (1968) New crystalline phases of zirconium phosphate possessing ion-exchange properties. J Inorg Nucl Chem 30:2249–2258. https://doi.org/10.1016/0022-1902(68)80224-6
Alberti G, Costantino U, Allulli S, Tomassini N (1978) Crystalline Zr(R-PO3)2 and Zr(R-OPO3)2 compounds (R = organic radical): a new class of materials having layered structure of the zirconium phosphate type. J Inorg Nucl Chem 40:1113–1117. https://doi.org/10.1016/0022-1902(78)80520-X
Alberti G, Costantino U, Perego G (1986) Structural investigation on layered α-zirconium phosphite and zirconium phosphate-phosphites from X-ray powder data. J Solid State Chem 63:455–460. https://doi.org/10.1016/0022-4596(86)90205-7
Sahu BB, Parida K (2002) Cation exchange and sorption properties of crystalline α-titanium(IV) phosphate. J Colloid Interface Sci 248:221–230. https://doi.org/10.1006/jcis.2001.7818
Bhaumik A, Inagaki S (2001) Mesoporous titanium phosphate molecular sieves with ion-exchange capacity. J Am Chem Soc 123:691–696. https://doi.org/10.1021/ja002481s
Kishore MS, Pralong V, Caignaert V, Varadaraju UV, Raveau B (2007) Electrochemical intercalation of lithium in the titanium hydrogen phosphate Ti(HPO4)2.H2O. J Power Sources 169:355–360. https://doi.org/10.1016/j.jpowsour.2007.03.051
Curnow P, Bessette PH, Kisailus D, Murr MM, Daugherty PS, Morse DE (2005) Enzymatic synthesis of layered titanium phosphates at low temperature and neutral pH by cell-surface display of silicatein-α. J Am Chem Soc 127:15749–15755. https://doi.org/10.1021/ja054307f
Wang Q, Yu H, Zhong L, Liu J, Sun J, Shen J (2006) Incorporation of silver ions into ultrathin titanium phosphate films: in situ reduction to prepare silver nanoparticles and their antibacterial activity. Chem Mater 18:1988–1994. https://doi.org/10.1021/cm052654i
del Val S, Granados M, Fierro JLG, Santamarı́a-González J, López A, Blasco T (2001) α-TiP-supported vanadium oxide catalysts: influence of calcination pretreatments on structure and performance for o-xylene oxidation. J Catal 204:466–478. https://doi.org/10.1006/jcat.2001.3417
Clearfield A (1984) Group IV phosphates as catalysts and catalyst supports. J Mol Catal 27:251–262. https://doi.org/10.1016/0304-5102(84)85084-1
Clearfield A, Thakur DS (1980) The acidity of zirconium phosphates in relation to their activity in the dehydration of cyclohexanol. J Catal 65:185–194. https://doi.org/10.1016/0021-9517(80)90293-6
Bruque S, Aranda MAG, Losilla ER, Olivera-Pastor P, Maireles-Torres P (1995) Synthesis optimization and crystal structures of layered metal(IV) hydrogen phosphates, α-M(HPO4).H2O (M = Ti, Sn, Pb). Inorg Chem 34:893–899. https://doi.org/10.1021/ic00108a021
Llavona R, García JR, Suárez M, Rodríguez J (1985) Synthesis of γ-titanium phosphate: study of the solids obtained by using water or acetone as washing agents. Thermochim Acta 86:281–289. https://doi.org/10.1016/0040-6031(85)87056-8
Ismail AA, Bahnemann DW (2011) Mesoporous titania photocatalysts: preparation, characterization and reaction mechanisms. J Mater Chem 21:11686–11707. https://doi.org/10.1039/C1JM10407A
Chen H, Nanayakkara CE, Grassian VH (2012) Titanium dioxide photocatalysis in atmospheric chemistry. Chem Rev 112:5919–5948. https://doi.org/10.1021/cr3002092
Antonelli DM, Ying JY (1995) Synthese von hexagonal gepacktem, mesoporösem TiO2 mit einer modifizierten sol‐gel‐methode. Angew Chem 107:2202–2206. https://doi.org/10.1002/ange.19951071828
Castro AL, Nunes MR, Carvalho AP, Costa FM, Florêncio MH (2008) Synthesis of anatase TiO2 nanoparticles with high temperature stability and photocatalytic activity. Solid State Sci 10:602–606. https://doi.org/10.1016/j.solidstatesciences.2007.10.012
Gajjela SR, Ananthanarayanan K, Yap C, Gratzel M, Balaya P (2010) Synthesis of mesoporous titanium dioxide by soft template-based approach: characterization and application in dye-sensitized solar cells. Energ Environ Sci 3:838–845. https://doi.org/10.1039/B921360K
Corma A (1997) From microporous to mesoporous molecular sieve materials and their use in catalysis. Chem Rev 97:2373–2420. https://doi.org/10.1021/cr960406n
Schüth F, Schmidt W (2002) Microporous and mesoporous materials. Adv Eng Mater 14:629–638. https://doi.org/10.1002/1521-4095(20020503)14:9
Lee J, Han S, Hyeon T (2004) Synthesis of new nanoporous carbon materials using nanostructured silica materials as templates. J Mater Chem 14:478–486. https://doi.org/10.1039/B311541K
Vanderborght BM, Van-Grieken RE (1977) Enrichment of trace metals in water by adsorption on activated carbon. Anal Chem 49:311–316. https://doi.org/10.1021/ac50010a032
Tanev PT, Pinnavaia TJ (1996) Mesoporous silica molecular sieves prepared by ionic and neutral surfactant templating: a comparison of physical properties. Chem Mater 8:2068–2079. https://doi.org/10.1021/cm950549a
Zhang W, Pauly TR, Pinnavaia TJ (1997) Tailoring the framework and textural mesopores of HMS molecular sieves through an electrically neutral (S0I0) assembly pathway. Chem Mater 9:2491–2498. https://doi.org/10.1021/cm970354y
Salvado MA, Pertierra P, Garcia-Granda S, Garcia JR, Rodriguez J, Fernandez-Diaz MT (1996) Neutron powder diffraction study of α-Ti(HPO4)2.H2O and α-Hf(HPO4)2.H2O: H-atom positions. Acta Crystallogr B 52:896–898. https://doi.org/10.1107/S0108768196006702
Christensen AN, Andersen EK, Andersen IGK, Alberti G, Nielsen M, Lehmann MS (1990) X-ray powder diffraction study of layer compounds: the crystal structure of α-Ti(HPO4)2∙H2O and a proposed structure for γ-Ti(H2PO4)(PO4).2H2O. Acta Chem Scand 44:865–872. https://doi.org/10.3891/acta.chem.scand.44-0865
Leofanti G, Padovan M, Tozzola G, Venturelli B (1998) Surface area and pore texture of catalysts. J Catal 41:207–219. https://doi.org/10.1016/S0920-5861(98)00050-9
Thommes M, Kaneko K, Neimark-Alexander V, Olivier-James P, Rodriguez-Reinoso F, Rouquerol J, Sing-Kenneth SW (2015) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl Chem 87:1051–1069. https://doi.org/10.1515/pac-2014-1117
Ortiz-Oliveros HB, Flores-Espinosa RM, Ordoñez-Regil E, Fernández-Valverde SM (2014) Synthesis of α-Ti(HPO4)2·H2O and sorption of Eu(III). Chem Eng J 236:398–405. https://doi.org/10.1016/j.cej.2013.09.103
Stanghellini PL, Boccaleri E, Diana E, Alberti G, Vivani R (2004) Vibrational study of some layered structures based on titanium and zirconium phosphates. Inorg Chem 43:5698–5703. https://doi.org/10.1021/ic049565c
Chen RY, Zhong J, Gu CR, Chen CL (2010) Computer simulation of n‐butylamine‐intercalated α‐zirconium phosphate. J Chin Chem Soc 57:1015–1021. https://doi.org/10.1002/jccs.201000141
Hall KR, Eagleton LC, Acrivos A, Vermeulen T (1966) Pore- and solid-diffusion kinetics in fixed-bed adsorption under constant-pattern conditions.Ind Eng Chem Fundam 5:212–223. https://doi.org/10.1021/i160018a011
Karadag D, Koc Y, Turan M, Armagan B (2006) Removal of ammonium ion from aqueous solution using natural Turkish clinoptilolite. J Hazard Mater 136:604–609. https://doi.org/10.1016/j.jhazmat.2005.12.042
Hameed BH, Tan IAW, Ahmad AL (2008) Adsorption isotherm, kinetic modeling and mechanism of 2,4,6-trichlorophenol on coconut husk-based activated carbon. Chem Eng J 144:235–244. https://doi.org/10.1016/j.cej.2008.01.028
Acknowledgements
The authors would like to acknowledge financial support by FONACIT through Project 2011001359, as well as the fine technical assistance of Pablo Quiroz and Oscar Zorrilla (XRD) from PDVSA-El Chaure, Yraida Díaz (nitrogen physisorption) from IVIC and Edgar Cañizales (TEM) from INTEVEP.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Carvajal, M.E., Brito, J.L., Bastardo-González, E. et al. Adsorption properties of novel layered titanium phosphate prepared from mesoporous titania by sol–gel processing. J Sol-Gel Sci Technol 97, 431–440 (2021). https://doi.org/10.1007/s10971-020-05435-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-020-05435-4