Abstract
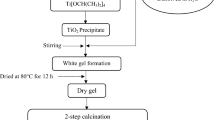
Magnesium-based Nasicon-type compounds with formula Mg0.9+0.5y Zn0.4Al y Zr1.6−y (PO4)3, (0.0 ≤ y ≤ 0.4) were prepared by the nitrate sol–gel method and characterized by X-ray diffraction, particle size analyser, scanning electron microscopy, energy-dispersive X-ray spectroscopy and impedance spectroscopy. The substitution of Zr4+ by a smaller Zn2+ and Al3+ cations reduced the unit cell dimensions of the parent compound’s structure to form a more stable framework and enhanced its conductivity by about three orders of magnitude. X-ray diffraction spectra clearly indicated the formation of single-phase compounds without any impurity. The compounds belong to the monoclinic structure with P21/n space group. The substitution of Al3+ and Zn2+ in zirconium sites did not affect the formation of the single phase, and this probably due to the concentration of both Al3+ and Zn2+ was too low to induce structural changes. The AC conductivity analysis demonstrated that the increase in conductivity was mainly due to an increase in the number of mobile ions. The Mg1.05Zn0.4Al0.3Zr1.3(PO4)3 exhibited the highest conductivity in the order of 10−4 S cm−1.
Graphical Abstract










Similar content being viewed by others
References
Adnan SBRS, Mohamed NS (2014) Characterization of novel Li4Zr0.06Si0.94O4 and Li3.94Cr0.02Zr0.06Si0.94O4 ceramic electrolytes for lithium cells. Ceram Int 40:6373–6379
Anuar NK, Adnan SBRS, Mohamed NS (2014) Characterization of Mg0.5Zr2(PO4)3 for potential use as electrolyte in solid state magnesium batteries. Ceram Int 40:13719–13727
Berry FJ, Vithal M (1995) New compounds with NASICON-related structures of the type NaM′M″P3O12 (M′ = Nb, Sb; M″ = Al, Ga, In, Fe). Polyhedron 14:1113–1115
Besenhard JO, Winter M (2002) Advances in battery technology: rechargeable magnesium batteries and novel negative-electrode materials for lithium ion batteries. Chem Phys Chem 3:155–159
Dorner G, Durakpasa H, Fafilek G, Breiter MW (1992) Production and characterization of polycrystalline (Na, Ca)β″-alumina. Solid State Ionics 53:553–558
Duluard S, Paillassa A, Puech L, Vinatier P, Turq V, Rozier P, Lenormand P, Taberna P-L, Simon P, Ansart F (2013) Lithium conducting solid electrolyte Li1.3Al0.3Ti1.7(PO4)3 obtained via solution chemistry. J Eur Ceram Soc 33:1145–1153
Dunn B, Farrington GC (1980) Fast divalent ion conduction in Ba++, Cd++ and Sr++ beta″ aluminas. Mater Res Bull 15:1773–1777
Farrington GC, Dunn B (1982) Divalent beta″-aluminas: high conductivity solid electrolytes for divalent cations. Solid State Ionics 7:267–281
Ikeda S, Kanbayashi Y, Nomura K, Kasai A, Ito K (1990) Solid electrolytes with multivalent cation conduction (2) zinc ion conduction in Zn–Zr–PO4 system. Solid State Ionics 40–41(Part 1):79–82
Ikeda S, Nomura K, Ito K, Einaga H (1994) Electrical conduction mechanism in solid electrolytes with zirconium phosphate framework. Solid State Ionics 70:153–156
Ikeda S, Takahashi M, Ishikawa J, Ito K (1987) Solid electrolytes with multivalent cation conduction. 1. Conducting species in Mg–Zr–PO4 system. Solid State Ionics 23:125–129
Imanaka N, Itaya M, Ueda T, Adachi G (2002) Tetravalent Zr4+ or Hf4+ ion conduction in NASICON type solids. Solid State Ionics 154–155:319–323
Imanaka N, Okazaki Y, G-Y Adachi (1999) Divalent magnesium ionic conduction in the magnesium phosphate based composites. Chem Lett 9:939–940
Kazakos-Kijowski A, Komarneni S, Agrawal D, Roy R (1988) Synthesis, crystal data and thermal stability of magnesium zirconium phosphate [MgZr4(PO4)6]. Mater Res Bull 23:1177–1184
Makino K, Katayama Y, Miura T, Kishi T (2001) Magnesium insertion into Mg0.5+y (Fe y Ti1−y )2(PO4)3. J Power Sources 97–98:512–514
Makino K, Katayama Y, Miura T, Kishi T (2002) Preparation and electrochemical magnesium insertion behaviors of Mg0.5+y (Me y Ti1−y )2(PO4)3 (Me = Cr, Fe). J Power Sources 112:85–89
Mančić D, Paunović V, Vijatović M, Stojanović B, Živković L (2008) Electrical characterization and impedance response of lanthanum doped barium titanate ceramics. Sci Sinter 40:283–294
Mustaffa NA, Adnan SBRS, Sulaiman M, Mohamed NS (2015) Low-temperature sintering effects on NASICON-structured LiSn2P3O12 solid electrolytes prepared via citric acid-assisted sol–gel method. Ionics 21:955–965
Mustaffa NA, Mohamed NS (2015) Zirconium-substituted LiSn2P3O12 solid electrolytes prepared via sol–gel method. J Sol–Gel Sci Technol 77:585–593
Nomura K, Ikeda S, Ito K, Einaga H (1992) Framework structure, phase transition and ionic conductivity of MgZr4(PO4)6 and ZnZr4(PO4)6. J Electroanal Chem 326:351–356
Nomura K, Ikeda S, Ito K, Einaga H (1992) Framework structure, phase transition, and transport properties in MIIZr4(PO4)6 compounds (MII = Mg, Ca, Sr, Ba, Mn Co, Ni, Zn, Cd, and Pb). Bull Chem Soc Jpn 65:3221–3227
Nomura K, Ikeda S, Ito K, Einaga H (1993) Ionic conduction behavior in zirconium phosphate framework. Solid State Ionics 61:293–301
Norhaniza R, Subban RHY, Mohamed NS (2013) Cr and V substituted LiSn2P3O12 solid electrolyte materials. J Power Sources 244:300–305
Seevers R, deNuzzio J, Farrington GC, Dunn B (1983) Ion transport in Ca2+, Sr2+, Ba2+, and Pb2+ beta″ aluminas. J Solid State Chem 50:146–152
Semkow KW, Sammells AF (1988) Ionic and electronic conductivity measurements on polycrystalline calcium conducting β″-alumina. J Electrochem Soc 135:244–247
Takahashia H, Takamura H (2012) Ionic conductivity and crystal structure of TM-doped Mg0.5Ti2(PO4)3 (TM = Fe, Mn, Co and Nb). Key Eng Mater 508:291
Tomita Y, Matsushita H, Yonekura H, Yamauchi Y, Yamada K, Kobayashi K (2004) Li ion conductivity of solid electrolyte, Li3−2x M x InBr6 (M = Mg, Ca, Sr, Ba). Solid State Ionics 174:35–39
West AR (1984) Solid state chemistry and its applications. Wiley, Chichester
Xu X, Yang T, Shui M, Lu Z, Gao S, Shu J, Zheng W, Cheng L, Feng L, Ren Y (2014) The preparation and lithium mobility of zinc based NASICON-type solid electrolyte Li1+2x+2y Al x Zn y Ti2−x−y Si x P3−x O12. Ceram Int 40:3819–3822
Zhang TS, Ma J, Cheng H, Chan SH (2006) Ionic conductivity of high-purity Gd-doped ceria solid solutions. Mater Res Bull 41:563–568
Acknowledgments
Financial support (Research Grants RP013A-13AFR and PG072-2015A) provided by the University of Malaya is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anuar, N.K., Mohamed, N.S. Structural and electrical properties of novel Mg0.9+0.5y Zn0.4Al y Zr1.6−y (PO4)3 ceramic electrolytes synthesized via nitrate sol–gel method. J Sol-Gel Sci Technol 80, 249–258 (2016). https://doi.org/10.1007/s10971-016-4091-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-016-4091-3