Abstract
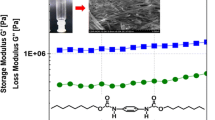
A novel class of toluene based bis urea compounds carrying linear fatty acid units and semicarbazide linkages has been synthesised. The compounds were exhibiting thermoreversible gelation at concentrations below 10 mg/mL in common organic solvents, both aliphatic and aromatic. The effect of the chain length variation of fatty acid units on gelation properties like gelation concentration, gelation time and gel melting temperatures were studied. Choosing a particular gelator of fixed chain length and a specific solvent, the effect of the concentration on the gelation properties were studied. The thermal studies using DSC revealed the presence of phase transitions corresponding to the premelting and melting of the gels during the heating cycle. The morphology of the xerogels studied using SEM revealed a three dimensional network structure while the WAXS studies showed no crystallinity in the xerogels. IR spectra of the gels (solvent subtracted) and solutions in the corresponding solvent showed that a high degree of inter-molecular H bonding exists and absorptions corresponding to NH stretching shifted to lower wave numbers. Thus simple bisurea type of compounds exhibiting gelation ability in a wide range of solvents can be used for making functional gels for various applications.








Similar content being viewed by others
References
van Esch J, Kellogg RM, Feringa BL (1997) Di-urea compounds as gelators for organic solvents. Tetrahedron Lett 38:281–284
de Loos M, van Esch J, Stokroos I, Kellogg RM, Feringa RL (1997) Remarkable stabilization of self-assembled organogels by polymerization. J Am Chem Soc 119:12675–12676
van Esch J, Schoonbeek F, de Loos M, Kooijman H, Spek AL, Kellogg RM, Feringa BL (1999) Cyclic bis-urea compounds as gelators for organic solvents. Chem Eur J 5:937–950
Schoonbeek FS, van Esch JH, Hulst R, Kellogg RM, Feringa BL (2000) Geminal bis-ureas as gelators for organic solvents: gelation properties and structural studies in solution and in the gel state. Chem Eur J 6:2633–2643
Brinksma J, Feringa BL, Kellogg RM, Vreeker R, van Esch J (2000) Rheology and thermotropic properties of bis-urea-based organogels in various primary alcohols. Langmuir 16:9249–9255
de Loos M, van Esch J, Kellogg RM, Feringa BL (2001) Chiral recognition in bis-urea-based aggregates and organogels through cooperative interactions. Angew Chem Int Ed 40:613–616
van der Laan S, Feringa BL, Kellogg RM, van Esch J (2002) Remarkable polymorphism in gels of new azobenzene bis-urea gelators. Langmuir 18:7136–7140
De Loos M, Friggeri M, Van Esch J, Kellogg RM, Feringa BL (2005) Cyclohexane bis-urea compounds for the gelation of water and aqueous solutions. Org Biomol Chem 3:1631–1639
Carr J, Melendez R, Geib SJ, Hamilton AD (1998) The design of organic gelators: solution and solid state properties of a family of bis-ureas. Tetrahedron Lett 39:7447–7450
Shi C, Huang Z, Kilic S, Xu J, Enick RM, Beckman EJ, Carr AJ, Melendez RE, Hamilton AD (1999) The gelation of CO2: a sustainable route to the creation of microcellular materials. Science 286:1540–1543
Estroff LA, Hamilton AD (2000) Effective gelation of water using a series of bis-urea dicarboxylic acids. Angew Chem Int Ed 39:3447–3450
Wang G, Hamilton AD (2002) Synthesis and self-assembling properties of polymerizable organogelators. Chem Eur J 8:1954–1961
Hanabusa K, Shimura K, Hirose K, Kimura M, Shirai H (1996) Formation of organogels by intermolecular hydrogen bonding between ureylene segment. Chem Lett 10:885–886
Kato T, Kutsuna T, Hanabusa K, Ukon M (1998) Gelation of room-temperature liquid crystals by the association of a trans-1,2-bis(amino)cyclohexane derivative. Adv Mater 10:606–608
Hanabusa K, Maesaka Y, Kimura M, Shirai H (1999) New gelators based on 2-amino-2-phenylethanol: close gelator-chiral structure relationship. Tetrahedron Lett 40:2385–2388
Mizoshita N, Hanabusa K, Kato T (1999) Self-aggregation of an amino acid derivative in a liquid-crystalline physical gel—faster response to electric fields. Adv Mater 11:392–394
Yabuuchi K, Marfo-Owusu E, Kato T (2003) A new urea gelator: incorporation of intra- and intermolecular hydrogen bonding for stable 1D self-assembly. Org Biomol Chem 1:3464–3469
Suzuki M, Nakajima Y, Yumoto M, Kimura M, Shirai H, Hanabusa K (2004) In situ organogelation at room temperature: direct synthesis of gelators in organic solvents. Org Biomol Chem 2:1155–1159
Jeong Y, Hanabusa K, Masunaga H, Akiba I, Miyoshi K, Sakurai S, Sakurai K (2005) Solvent/gelator interactions and supramolecular structure of gel fibers in cyclic bis-urea/primary alcohol organogels. Langmuir 21:586–594
Beginn U, Tartsch B (2001) Bis[(alkoxy)benzoylsemicarbazides]—a new class of powerful organogelators. Chem Commun 19:1924
Rodrguez-Llansola F, Hermida-Merino D, Nieto-Ortega B, Ramrez FJ, Lopez Navarrete JT, Casado J, Hamley IW, Escuder B, Hayes W, Miravet JF (2012) Self-assembly studies of a chiral bisurea-based superhydrogelator. Chem Eur J 18:14725–14731
Lortie F, Boileau S, Bouteiller L, Chassenieux C, Deme´ B, Ducouret G, Jalabert M, Laupreˆtre F, Terech P (2002) Structural and rheological study of a bis-urea based reversible polymer in an apolar solvent. Langmuir 18:7218–7222
Zweep N, Hopkinson A, Meetsma A, Browne WR, Feringa BL, van Esch JH (2009) Balancing hydrogen bonding and van der Waals interactions in cyclohexane-based bisamide and bisurea organogelators†. Langmuir 25(15):8802–8809
Moniruzzaman M, Goodbrand B, Sundararajan PR (2003) Morphology and thermal behavior of self-assembling carbamates. J Phys Chem B 107:8416–8423
Moniruzzaman M, Sundararajan PR (2005) Role of hydrogen bonds in controlling the morphology of self-assembling carbamate systems. J Phys Chem B 109:1192–1197
Khanna S, Moniruzzaman M, Sundararajan PR (2006) Influence of single versus double hydrogen-bonding motif on the crystallization and morphology of self-assembling carbamates with alkyl side chains: model system for polyurethanes. J Phys Chem B 110:15251–15260
Moniruzzaman M, Sundararajan PR (2005) Low molecular weight organogels based on long-chain carbamates. Langmuir 21:3802–3807
Ohsedo Y, Miyamoto M, Watanabe H, Oono M, Tanaka A (2013) Alkylhydrazide derivatives as new organogelators and their potential ability to gel electrolytes. Bull Chem Soc Jpn 86(5):671–673
Campbell TW, Foldi VS, Farago J (1959) Condensation polymers from diisocyanates with dihydrazides and hydrazine. J Appl Polym Sci 2:155–162
Vogel AI, Tatchell AR, Furnis BS, Hannaford AJ, Smith PWG (1996) Vogel’s text book of practical organic chemistry, 5th ed. ELBS, London, p 703
van Esch J, de Feyter S, Kellogg RM, de Schryver FB, Feringa L (1997) Self-assembly of bisurea compounds in organic solvents and on solid substrates. Chem Eur J 3:1238–1243
Hirst AR, Smith DK, Harrington JP (2005) Unique nanoscale morphologies underpinning organic gel-phase materials. Chem Eur J 11:6552–6559
Acknowledgments
Corresponding author wishes to acknowledge CSIR XII five year plan project INTELCOAT (CSC 0114) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sravan, B., Kamalakar, K., Karuna, M.S.L. et al. Studies on organogelation of self assembling bis urea type low molecular weight molecules. J Sol-Gel Sci Technol 71, 372–379 (2014). https://doi.org/10.1007/s10971-014-3386-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-014-3386-5