Abstract
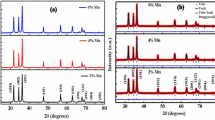
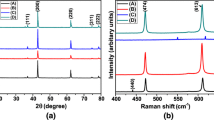
Undoped and Mn doped ZnO nanoparticles were synthesized by two wet chemical techniques: sol–gel and gel-combustion. We were able to prepare Mn-doped ZnO nanoparticles free from the second phases at calcining temperatures 400 and 500 °C using sol–gel and gel combustion, respectively. Complete crystallization occurs in both methods, but it is found that the crystallization is better performed in the ZnO based solid solution prepared by the sol–gel method. TEM images show that the average size of the nanoparticles synthesized by gel-combustion is smaller than that of prepared by sol–gel method. Optical characterizations such as vibrational properties, lattice dynamical parameters, absorption edges and optical band gap energies were also carried out by FTIR and UV–Vis spectroscopies. The quantitative estimations led to the conclusion that the effects of the method on the particle size and the optical band gap energy of the prepared samples are very significant.









Similar content being viewed by others
References
Brus L (1990) J Phys Chem 90:2555–2560
Nepijko SA, Pippel E, Woltersdorf J (2006) Phys Stat Sol A 61:469–475
Dinesha ML, Jayanna HS, Mohanty S, Ravi S (2010) J Alloy Compd 490:618–623
Bhargava R, Sharma PK, Dutta RK, Kumar S, Panday AC, Kumar N (2010) Mat Chem Phys 120:393–398
Jung D (2010) Solid State Sci 12:466–470
Fang Y, Wen X, Yang S, Pang Q, Ding L, Wang J, Ge W (2005) J Sol-Gel Sci Techn 36:227–234
Wang J, Shi N, Qi Y, Liu M (2010) J Sol-Gel Sci Techn 53:101–106
Yang Z, Ye Z, Zhao B, Zong X, Wang P (2010) J Sol-Gel Sci Techn 54:282–285
Selim MM, Deraz NM, El-Asmi AA, Elshfay O (2010) J Alloy Compd 506:541–547
Jayakumar OD, Salunke HG, Kadam RM, Mohapatra M, Yaswant G, Kulshreshta SK (2006) Nanotechnology 17:1278–1285
Lojkowski W, Gedanken A, Grzanka E, Opalinska A, Strachowski T, Pielaszek R, Tomaszewska-Grzeda A, Yatsunenko S, Godlewski M, Matysiak H, Kurzydlowski KJ (2009) J Nanopart Res 11:1991–2002
Hwang CC, Wu TY (2004) J Mater Sci 39:6111–6115
Chattopadhyay S, Dutta S, Banerjee A, Jana D, Bandyopadhyay S, Chattopadhyay S, Sarkar A (2009) Phys B 404:1509–1510
Ebrahimizadeh Abrishami M, Hosseini SM, Kompany A (2011) J Appl Sci 11:1411–1415
Kwon YJ, Kim KH, Lim CS, Shim KB (2002) J Ceram Process Res 3:146–149
Wang H, Xie C (2008) J Phys Chem Solids 69:2440–2444
Shinde VR, Gujar TP, Lokhande CD, Mane RS, Han S-W (2006) Mat Chem Phys 96:326–330
Morales AE, Mora ES, Pal U (2007) Rev Mex Fis 53:18–22
Senthilkumaar S, Rajendran K, Banerjee S, Chini TK, Sengodan V (2008) Mat Sci Semicon Proc 11:6–12
Olguin D, Baquero R, de Coss R (2001) Rev Mex Fis 47:43–49
Kim KJ, Park YR (2002) Appl Phys Lett 81:1420–1422
Ebrahimizadeh Abrishami M, Attaran Kakhki E, Hosseini SM, Kompany A (2010) Mod Phys Lett B 24:749–760
Cedillo E, Ocampo J, Rivera V, Valenzuela R (1980) J Phys E Sci Instrum 13:383–386
Rekha K, Nirmala M, Nair MG, Anukaliani A (2010) Phys B 405:3180–3185
Deepa M, Bahadur N, Srivastava AK, Chaganti P, Sood KN (2009) J Phys Chem Solids 70:291–297
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ebrahimizadeh Abrishami, M., Kompany, A., Hosseini, S.M. et al. Preparing undoped and Mn-doped ZnO nanoparticles: a comparison between sol–gel and gel-combustion methods. J Sol-Gel Sci Technol 62, 153–159 (2012). https://doi.org/10.1007/s10971-012-2701-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-012-2701-2