Abstract
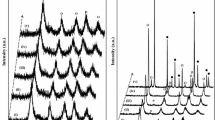
Synthesis by sol-gel method of mixed semiconductor ZnO-Fe2O3 was carried out using inorganic salts as precursors. The powders were synthesized at different pH (neutral and basic) in order to study its influence on the properties of the catalysts. These materials were characterized by thermal analysis TGA-DTA, X-ray Diffraction, FTIR spectroscopy, scanning electron microscopy and X-ray energy dispersive spectroscopy. The specific surface area of the solids was calculated from nitrogen adsorption isotherms by BET method. The band gap energy (E g ) of the catalysts was obtained from it UV-Vis spectra (diffuse reflectance) when the crystalline phase was formed under thermal treatment at 350°C by 3 h. The incorporation of Fe2O3 to ZnO semiconductor gives as a result a lower E g value for zinc oxide. Catalytic activity of the materials was evaluated using as test reaction the photocatalytic degradation of potassium cyanide in aqueous dissolution. The catalyst ZnO-Fe2O3 synthesized at pH 7 turned out to be superior than sol-gel and commercial ZnO under similar conditions.






Similar content being viewed by others
References
Herrmann JM, Guillard Ch, Disdier J, Lehaut C, Malato S, Blanco J (2002) Appl Catal B: Environ 35:281
Herrmann JM (1994) Trends Photochem Photobiol 3:633
Fox MA, Dulay MT (1993) Chem Rev 93:341
Herrmann JM (2001) Helvetica Chimica Acta 84:2731
Malato S (2004) Thessalonica
Valenzuela MA, Bosch P, Jimenez-Becerril J, Quiroz O, Páez AI (2002) J Photochem Photobiol A: Chem 148:177
Sene Jeosadeque J, Mzeltner WA, Anderson MA (2003) J Phys Chem B 107:1597
Chiang K, Amal R, Tran T (2002) Adv Environ Res 6:471
Sadeghi M, Liu W, Zhang T-G, Stavropoulos P, Levy B (1996) J Phys Chem 100:19466
Ho W, Yu JC, Lin J, Yu J, Li P (2004) Langmuir 20:5865
Serpone N (1995) Photochem Photobiol A: Chem 85:247
Villaseñor J, Reyes P, Pecchi G (1998) J Chem Technol Biotechnol 72:105
Pal B, Sharon M (2002) Mater Chem Phys 76:82
Marci G, Augugliaro V, López-Muñoz MJ, Martín C, Palmisano L, Rives V, Schiavello M, Tilley RJD, Venezia AM (2001) J Phys Chem B 105:1026
Marci G, Augugliaro V, López-Muñoz MJ, Martín C, Palmisano L, Rives V, Schiavello M, Tilley RJD, Venezia AM (2001) J Phys Chem B 105:1033
Sánchez-Mora E, Gómez-Barojas E, Gracia-Jiménez JM, Silva-González R, Pérez-Rodríguez F (2004) Phys Stat Sol (c) 1: s-116
Shaogui Y, Xie Q, Xinyong L, Yazi L, Shuo C, GuoHua C (2004) Phys Chem Chem Phys 6:659
Yuan Z-H, Zhang L-D (2001) J Mater Chem 11:1265
Brinker CJ, Scherer GW (1990) Sol gel science. Academic Press, New York
Livage J (1986) J Solid State Chem 64:322
Livage J, Henry M, Sánchez C (1988) Prog Solid St Chem 18:259
Lopez T, Sanchez E, Bosch P, Meas Y, Gomez R (1992) Mater Chem Phys 32:141–152
Hernandez A, Torres-Martinez LM, Lopez T (2002) Mater Lett 54:62
Nakamoto K (1978) Infrared and raman spectra of inorganic and coordination compounds. John Wiley & Sons
Ferraro JR (1971) Low frequency vibrations of inorganic and coordination compounds. Plenum Press, New York
Lopez T, Mendez J, Zamudio T, Villa M (1992) Mater Chem Phys 30:161–167
JCPDS 36-1451, McMurdie H (1986) Powder Diffraction 1:76
West AR (1992) Solid state chemistry and its applications. John Wiley & Sons, Great Britain
Sakthivel S, Geissen S-U, Bahnemann DW, Murugesan V, Vogelpohi A (2002) J Photochem Photobiol A: Chem 148:283
Grumes A, Leapman RD, Wilker CN, Hoffmann R, Kuns AB (1982) Phys Rev B 25:7157
López T, Alvarez M, Gomez R, Aguilar DH, Quintana P (2005) J Sol-Gel Sci Technol 33:93
Serpone N, Pellizeti E (1989) Photocatalysis fundamentals and applications. John Wiley & Sons
Ding Z, Lu GQ, Greenfield PF (2000) J Phys Chem B 104:4815
Acknowledgments
The authors gratefully acknowledge the financial support by grants: PROMEP/103.5/05/227 and PAICYT CA1048-05. We are grateful to Esthela Gomez for technical assistance in EDS analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hernández, A., Maya, L., Sánchez-Mora, E. et al. Sol-gel synthesis, characterization and photocatalytic activity of mixed oxide ZnO-Fe2O3 . J Sol-Gel Sci Technol 42, 71–78 (2007). https://doi.org/10.1007/s10971-006-1521-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-006-1521-7