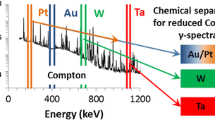
Abstract
Gamma detection of select fission products or activation products such as 153Sm and 239Np in a fission sample is difficult due to gamma interferences from radiotellurium and radioiodine. To aid in the detection of these difficult-to-detect nuclides, the radiotellurium and radioiodine were removed using Cu metal turnings. This method is highly effective at the removal of the interfering radionuclides removing > 98% of the radiotellurium and radioiodine. A mechanistic study was conducted. Determining the deposition mechanism, using a number of techniques including XANES and electrochemistry providing an understanding of the electrochemical path for the Te deposition. Initial work focused on the use of stable Te, further expanding into activated TeO2, and a fresh fission sample. This method presents a rapid method for the direct detection of select fission products that could have a significant impact on nuclear related fields including nuclear forensics.











Similar content being viewed by others
Data availability
All data and materials included in the submitted manuscript will be available upon request.
References
Wren MS, May I, Guardincerri E, Boswell MS, Herman SM, Warzecha EJ, Haney MM, Fotiades N, Dale GE, Hanson SK (2023) Chemical separation and measurement of platinum activation products. Talanta 260:124587
Pierson BD, Archambault BC, Greenwood LR, Haney MM, Cantaloub MG, Hagen AR, Herman SM, Uhnak NE, Bowen JM, Estrada JH (2022) Alpha/beta-gated gamma-gamma spectroscopy of mixed fission products for trace analysis. J Radioanal Nucl Chem 331:5453–5467
Soppera N, Bossant M, Dupont E (2014) JANIS 4: an improved version of the NEA java-based nuclear data information system. Nucl Data Sheets 120:294–296
Haney MM, Seiner BN, Finn EC, Friese JI (2016) Rapid quantification of uranium from mixed fission product samples. J Radioanal Nucl Chem 307:1737–1742
Seiner BN, Morley SM, Beacham TA, Haney GS, Metz L (2014) Effects of digestion, chemical separation, and deposition on Po-210 quantitative analysis. J Radioanal Nucl Chem 302(1):673–678
Miller DA, Sun S, Yi JH (1992) Preparation of a tellurium-118/antimony-118 radionuclide generator. J Radioanal Nucl Chem 160(2):467–476
Downs D, Miller DA (2004) Radiochemical separation of antimony and tellurium in isotope production and in radionuclide generators. J Radioanal Nucl Chem 262(1):241–247
Yu C, Cai Q, Guo Z, Yang Z, Khoo SB (2004) Simultaneous speciation of inorganic selenium and tellurium by inductively coupled plasma mass spectrometry following selective solid-phase extraction separation. J Anal At Spectrochem 19:410–413
Mostafa M, Ramadan HE, El-Amir MA (2013) Radiochemical precipitation studies for separation of iodine from tellurium and other trace impurities. J Radioanal Nucl Chem 295:115–123
Mostafa M, El-gharbawy AF, El-Absy MA, Soliman SE, Aly FA (2014) Preconcentration of radioiodine as AgI and purification from radiotellurium waste. J Radioanal Nucl Chem 300:1089–1097
Mandal DK, Bhattacharya B, Das RD (2004) Recovery of tellurium from chloride media using tri-iso-octylamine. Sep Purif Technol 40:177–182
Uhnak NE, Morrison SS, Morley S, Edwards A, Finn E, Seiner BN (2018) Cu metal based radiotellurium separation from simulated and HEU process streams. J Radioanal Nucl Chem 317:519–526
Zhao H, Chang J, Boika A, Bard A (2013) Electrochemistry of high concentration copper chloride complexes. Anal Chem 85:7696–7703
Marks J, Cone R, Leao E (1971) The spectrographic determination of traces of tellurium. Appl Spectrosc 25(4):493–496
Lilja C, King F, Puigdomenech I, Pastina B (2021) Speciation of copper in high concentrations in the context of corrosion of copper canisters. Mater Corros 72:293–299
Milne JB (1991) Chlorotellurate(IV) equilibria in aqueous hydrochloric acid. Inorg Chem 23:987–991
Barati G, Solokha V, Wandelt K, Hingerl K, Cobet C (2014) Chloride-induced morphology transformations of the Cu(110) surface in Dilute HCl. Langmuir 30:14486–14493
Goletti C, Bussetti G, Violante A, Bonanni B, Di Giovannantonio M, Serrano G, Breuer S, Gentz K, Wandelt K (2015) Cu(110) surface in hydrochloric acid solution: potential dependent chloride adsorption and surface restructuring. J Phys Chem C 119:1782–1790
Rosamilia JM, Miller B (1986) Voltammetric studies of tellurium film and hydrogen telluride formation in acidic tellurite solution. J Electroanal Chem 215:261–271
Sorgho A, Bougouma M, De Leener G, Steen J, Doneux T (2022) (2022) Impact of speciation on the tellurium electrochemistry in choline chloride-based deep eutectic solvents. Electrochem Commun 140:107327
Martell A (2004) NIST critically selected stability constants of metal complexes database
Bouroussian M (2010) In: Scholz F (ed), Electrochemistry of Metal Chalcogenides, Springer-Verlag, Berlin, Heidelberg
Suggs WD, Bard AJ (1995) Scanning tunneling microscopic study with atomic resolution of the dissolution of Cu(100) electrodes in aqueous chloride media. J Phys Chem 99:8349–8355
Mokmeli M, Dreisinger D, Wassink B (2014) Thermodynamics and kinetics study of tellurium removal with cuprous ion. Hydromettallurgy 147–148:20–29
Lide DR (2010) Handbook of chemistry and physics. CRC Press, Boca Raton, USA
Ishizaki T, Yata D, Fuwa A (2003) Electrodeposition of a copper-tellurium compound under diffusion-limiting control. Mater Trans 44(8):1583–1587
Gebregziabiher DK, Ledina MA, Preisser R, Stickney JL (2012) Cu(111) surface passivation with atomic layers of Te, Se or I, studied using auger spectroscopy. J Electrochem Soc 159(8):H675–H679
Vance E, Grant C, Karatchevtseva I, Aly Z, Stopic A, Harrison J, Thorogood G, Wong H, Gregg D (2018) Immobilization of iodine via copper iodide. J Nucl Mater 505:143–148
Andryushechkin BV, Eltsov KN, Shevlyuga VM (2004) CuI growth on copper surfaces under molecular iodine action: influence of the surface anisotropy in the iodine monolayer. Surf Sci 566–568:203–209
Riley B, Chong S, Beck C (2021) Iodine vapor reactions with pure metal wires at temperatures of 100–139 °C in air. Ind Eng Chem Res 60(47):17162–17173
Nerisson P, Barrachin M, Ohnet M, Cantrel L (2022) Behavior of ruthenium in nitric media (HLLW) in reprocessing plants: a review and some perspectives. J Radioanal Nucl Chem 331:3365–3389
Acknowledgements
This work was performed in coordination with National Nuclear Security Administration Office of Defense Nuclear Nonproliferation Research and Development. This research was conducted at the U.S. Department of Energy (DOE)’s Pacific Northwest National Laboratory, which is operated for the DOE by Battelle Memorial Institute under Contract DE-AC05-76RL1830.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing fnancial interests or personal relationships that could have appeared to infuence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uhnak, N.E., Saslow, S., Bowen, J. et al. Removal of fission product tellurium and iodine from recently irradiated HEU using copper metal. J Radioanal Nucl Chem 333, 467–479 (2024). https://doi.org/10.1007/s10967-023-09275-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-09275-8