Abstract
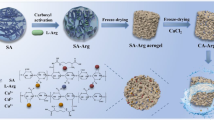
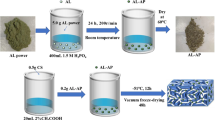
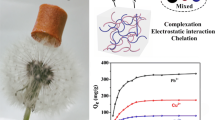
Novel chitosan crosslinked amidoxime polyacrylonitrile aerogel was synthesized and used for adsorption of uranium from seawater. The effects of initial uranium concentration, contact time, and pH value on uranium adsorption were investigated. The simulated uranium solution adsorption experiment shows that under the condition of an initial uranium concentration of 100 mg/L, the maximum adsorption capacity of CTS-g-PAO is 203.4 mg/g. The adsorption process can be well-fitted by Freundlich and Pseudo-second order models. In addition, CTS-g-PAO aerogel has good antibacterial activity against marine bacteria, with the antibacterial activity reaching 94.8%. The results demonstrated that CTS-g-PAO aerogel was an effective adsorbent for extracting uranium from seawater.












Similar content being viewed by others
References
Carvalho FP (2017) Mining industry and sustainable development: time for change. Food Energy Secur 6(2):61–77
Abney CW, Mayes RT, Saito T et al (2017) Materials for the recovery of uranium from seawater. Biochem Rev 117(23):13935–14013
Li ZM, Niu YQ, Su YT et al (2022) Latest research progress in uranium extraction technology from seawater. Nucl Radiochem 44(3):233–245
Egawa H, Nakayama M, Nonaka T et al (1987) Recovery of uranium from seawater. IV. Influence of crosslinking reagent of the uranium adsorption of macroreticular chelating resin containing amidoxime groups. J Appl Polym Sci 33(6):1993–2005
Singh K, Shah C et al (2013) Study of uranium adsorption using amidoximated polyacrylonitrile-encapsulated macroporous beads. J Appl Polym Sci 127(1):410–419
Hwang TS et al (2003) Synthesis of ion exchange fiber containing amidoxime and phosphoric acid groups and its uranium adsorption properties. Polym 27(3):242–248
Chandrasekaran K, Karunasagar et al (2013) Dispersive liquid-liquid micro extraction of boron as tetrafluoroborate ion (BF4) from natural waters, wastewater and seawater samples and determination using a micro-flow nebulizer in inductively coupled plasma-quadrupole mass spectrometry. J Anal At Spectrom 28(1):142–149
Luo W, Kelly SD, Kemner KM et al (2009) Sequestering uranium and technetium through co-precipitation with aluminum in a contaminated acidic environment. Environ Sci Technol 43(19):7516–7522
Bai Z, Wang Y, Li Y et al (2016) First cationic uranyl-organic framework with anion-exchange capabilities. Inorg Chem 55(13):6358–6360
Jin KY, Yu SC, Gao CJ (2001) The development status of extracting uranium from seawater. Mar Sci Bull 20(2):78–82
Du YF, Yin ZX, Wu HY et al (2015) Sorption of U(VI) on magnetic illite: effects of pH, ions, humic substances and temperature. J Radioanal Nucl Chem 304(2):793–804
Hamid A, Yadollah et al (2018) Highly efficient capture and recovery of uranium by reusable layered double hydroxide intercalated with 2-mercaptoethanesulfonate. Chem Eng J 337:609–615
Simsek S, Ulusoy U (2013) Adsorptive properties of sulfolignin–polyacrylamide graft copolymer for lead and uranium: Effect of hydroxilamine–hydrochloride treatment. React Funct Polym 73(1):73–82
Zeng YY, Liu S, Xu JC et al (2021) ZIF-8 in-situ growth on amidoximerized polyacrylonitrile beads for uranium sequestration in wastewater and seawater. Environ Chem Eng 9(6):96–106
Alqadami AA, Naushad M, Alothman ZA et al (2017) Novel metal-organic framework (MOF) based composite material for the sequestration of U(VI) and Th(IV) metal ions from aqueous environment. ACS Appl Mater Interfaces 9(41):36026–36037
Cui WR, Zhang CR, Jiang W et al (2020) Regenerable and stable sp(2) carbon-conjugated covalent organic frameworks for selective detection and extraction of uranium. Nat Commun 11(1):436–446
Seko N, Katakai A, Hasegawa S et al (2003) Aquaculture of uranium in seawater by a fabric-adsorbent submerged system. Nucl Technol 144(2):274–278
Li LY, Wen J, Hu S et al (2022) Research progress on biofouling resistant uranium extraction materials from seawater. Nucl Radiochem 44(3):299–312
Kumar M (2000) A review of chitin and chitosan applications. React Funct Polym 46(1):1–27
Márquez G, Imelda AJ, Cruz I et al (2013) Disruption of protein synthesis as an antifungal mode of action by chitosan. Int J Food Microbiol 164(1):108–112
Raafat D, Bargen KV, Haas A et al (2008) Insights into the mode of action of chitosan as an antibacterial compound. Appl Environ Microbiol 74(12):3764–3773
Helander IM, Nurmiaho-Lassila EL, Ahvenainen R et al (2001) Chitosan disrupts the barrier properties of the outer membrane of gram-negative bacteria. Int J Food Microbiol 71(2–3):235–244
Wang G, Liu J, Wang X et al (2009) Adsorption of uranium (VI) from aqueous solution onto cross-linked chitosan. J Hazard Mater 168(2–3):1053–1058
Wang CX, Liu YH, Huang DJ et al (2009) Synthesis of serine modified chitosan and its adsorption of uranium. Chem Res Appl 21(10):1361–1363
Liu Y, Cao X, Rong H et al (2010) Selective adsorption of uranyl ion on ion-imprinted chitosan/PVA cross-linked hydrogel. Hydrometallurgy 104(2):150–155
Gao L, Yang Z, Shi K et al (2010) U(VI) sorption on kaolinite: efects of pH, U(VI) concentration and oxyanions. J Radioanal Nucl Chem 284(3):519–526
Hashimoto Y, Taki T, Sato T (2009) Sorption of dissolved lead from shooting range soils using hydroxyapatite amendments synthesized from industrial byproducts as afected by varying pH conditions. J Environ Manag 90(5):1782–1789
He NN, Lu X, Wen J (2022) Hyperbranched antibacterial seawater uranium extraction material. J Nucl Radiochem 44(1):90–99
Xu X, Yue Y, Cai D et al (2020) aqueous solution blow spinning of seawater-stable polyamidoxime nanofibers from water-soluble precursor for uranium extraction from seawater. Small Methods 4(12):200558
Shao D, Hou G, Chi FT et al (2021) Transformation details of poly(acrylonitrile) to poly(amidoxime) during the amidoximation process. Rsc Adv 11(4):1909–1915
Sabaa MW, Mohamed NA, Ali R et al (2010) Chemically induced graft copolymerization of acrylonitrile onto carboxymethyl chitosan and its modification to amidoxime derivative. Polym Plast Technol Eng 49(11):1055–1064
Peckham J, Andrews GT (2015) Comparative study of the biodegradability of porous silicon films in simulated body fluid. Bio-Med Mater Eng 25(1):111–116
Cheng JF, Leng YC, Tuo XG et al (2017) Efect of pH on adsorption characteristics of uranium and plutonium. J Nucl Radiochem 39(3):213–217
Schmeide K, Pompe S, Bubner M et al (2000) Uranium(VI) sorption onto phyllite and selected minerals in the presence of humic acid. Radiochim Acta 88(9–11):723–728
Xu D, Tan X, Chen C et al (2008) Removal of Pb(II) from aqueous solution by oxidized multiwalled carbon nanotubes. J Hazard Mater 154(1–3):407–416
Özcan AS, Gök Ö, Özcan A (2009) Adsorption of lead( II) ions onto 8-hydroxy quinoline-immobilized bentonite. J Hazard Mater 161(1):499–509
Zhou X, Zhou J, Liu Y et al (2018) Preparation of iminodiacetic acid-modified magnetic biochar by carbonization, magnetization and functional modification for Cd(II) removal in water. J Fuel 233:469–479
Ma TX, Yang C, Jiang XY et al (2016) Preparation of nano zero valent iron modified amino biochar and its adsorption and desorption characteristics for Cd (II). J Environ Eng 10(10):5433–5439
Zhou C, Wu Q, Lei T et al (2014) Adsorption kinetic and equilibrium studies for methylene blue dye by partially hydrolyzed polyacrylamide/cellulose nanocrystal nanocomposite hydrogels. Chem Eng J 251:17–24
Zhang F, Zhang H, Chen R et al (2019) Mussel-inspired antifouling magnetic activated carbon for uranium recovery from simulated seawater. J Colloid Interface Sci 5(34):172–182
Li H, Wen J, Wang XL (2020) Polyguanidine modified antibacterial seawater uranium extraction material. J Sichuan Univ, Nat Sci Ed 57(3):569–576
Wang Y, Lin Z, Liu Q et al (2021) Simple one-step synthesis of woven amidoximated natural material bamboo strips for uranium extraction from seawater. Chem Eng J 425:131538
Tang N, Liang J, Niu C et al (2020) Amidoxime-based materials for uranium recovery and removal. J Mater Chem 8(16):7588–7652
Gao JJ, Ma JL, Zhang JQ et al (2022) Porous and biofouling-resistant amidoxime-based hybrid hydrogel with excellent interfacial compatibility for high-performance recovery of uranium from seawater. Sep Purif Technol 287:120571
Acknowledgements
This work was supported by the National Natural Science Foundation of China (U2130125); Sponsorship of the Sichuan Provincial Science and Technology Plan Project (2023 NSFSC1959).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors disclosed no relevant relationships. All relevant data are within the paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Du, Z., Ni, Y., Peng, H. et al. Adsorption of U(VI) by chitosan crosslinked PAO aerogel. J Radioanal Nucl Chem 333, 71–84 (2024). https://doi.org/10.1007/s10967-023-09261-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-09261-0