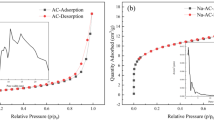
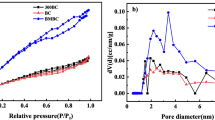
Abstract
With the exploitation of uranium–beryllium ore, the waste and wastewater produced have received extensive attention. There are a lot of toxic and harmful substances in uranium–beryllium ore, among which beryllium has not been widely concerned. This paper used hydrochloric acid (HCl) pretreatment activated carbon (H-AC) to study the removal of beryllium from uranium beryllium ore wastewater. This experiment explored the influence of reaction time, reaction temperature, and other conditions on the adsorption effect. It concluded that the adsorption efficiency was 99% when pH = 5, the amount of adsorbent was 2 g/L, and the temperature was 25 °C. The effects of Be–U, Be–Mn, Be–Zn, Be–Fe and, Be–Al binary systems on the adsorption of beryllium by H-AC were analyzed. The results show that the adsorption capacity of H-AC to beryllium is not affected by the coexisting ions. According to thermodynamics, the adsorption process is a spontaneous endothermic single-layer adsorption, and the maximum adsorption capacity fitted by thermodynamics is 30.63 mg/g. Kinetic fitting showed that chemical adsorption was the primary process, and the reaction was intense in the early stage. This study provides a new idea for removing beryllium from uranium beryllium ore wastewater, and H-AC can be considered a potential adsorbent for removing beryllium from industrial wastewater.











Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article.
References
Jia J, Li X, Wu P et al (2015) Human health risk assessment and safety threshold of harmful trace elements in the soil environment of the Wulantuga open-cast uranium–beryllium ore. Minerals 5(4):837–848
Qi H, Hu R, Zhang Q (2007) Concentration and distribution of trace elements in lignite from the Shengli Coalfield, Inner Mongolia, China: implications on origin of the associated Wulantuga germanium deposit. Int J Coal Geol 71(2–3):129–152
Dai SF, Seredin VV, Ward CR, Jiang JH, Hower JC, Song XL, Jiang YF, Wang XB, Gornostaeva T, Li X et al (2013) Composition and modes of occurrence of minerals and elements in coal combustion products derived from high-Ge coals. Int J Coal Geol 121:79–97
Wang JN, Liu ZX, Rong LI, Qiang LI, Deng YF (2014) Analysis of element content and discussion on migration of coal and flyash in coal-fired power plant. J Henan Univ Urban Constr 1(23):27–31
Chen S, Gui H (2019) The geochemical (alkaline earth) characteristics of groundwater in the taiyuan formation limestone aquifer in Huaibei coalfield. Water Pract Technol 14(1):217–228
Chiarappa-Zucca ML, Finkel RC, Martinelli RE et al (2004) Measurement of beryllium in biological samples by accelerator mass spectrometry: applications for studying chronic beryllium disease. Chem Res Toxicol 17(12):1614–1620
Jitka P, Vladimír B (2020) Health aspects of exposure to emissions from burning coal of high beryllium content: interactions with the immune system. Cent Eur J Publ Heal 128(3):198–201
Sun F, Sun WL (2012) A simultaneous removal of beryllium and ammonium–nitrogen from smelting wastewater in bench-and pilot-scale biological aerated filter. Chem Eng J 210:263–270
Wan C, Xie Q, Liu J et al (2021) Pilot-scale combined adsorption columns using activated carbon and zeolite for hazardous trace elements removal from wastewater of entrained-flow coal gasification. Process Saf Environ 147:439–449
Tokarčíková M, Motyka O, Peikertová P, Gabor R, Seidlerová J (2021) Magnetically modified biosorbent for rapid beryllium elimination from the aqueous environment. Materials 14(21):6610
Vetrivel SA, Diptanghu M, Ebhin MR, Sydavalli S, Gaurav N, Tiger KP (2017) Green algae of the genus spirogyra: a potential absorbent for heavy metal from uranium–beryllium ore water. Remediat J 27(3):81–90
Tsai WT, Huang PC (2018) Characterization of acid-leaching cocoa pod husk (CPH) and its resulting activated carbon. Biomass Convers Biorfin 8(3):521–528
Ahmad F, Daud WMAW, Ahmad MA, Radzi R (2013) The effects of acid leaching on porosity and surface functional groups of cocoa (Theobroma cacao)-shell based activated carbon. Chem Eng Res Des 91(6):1028–1038
Topcu NS, Duman G, Olgun H et al (2022) Evaluation of poultry manure: combination of phosphorus recovery and activated carbon production. ACS Omega 7(24):20710–20718
Saini VK, Suthar S, Karmveer C, Kumar K (2017) Valorization of toxic weed Lantana camara L. biomass for adsorptive removal of lead. J Chem-NY. https://doi.org/10.1155/2017/5612594
Zhao X, Su Y, Wang H, Lei Z, Hu E, Hu F, Hao X (2023) Modification of activated carbon from agricultural waste lotus leaf and its adsorption mechanism of beryllium. Korean J Chem Eng 40(1):255–266
Zhao X, Shuai D, Hongqiang W, Eming H, Fang H, Zhiwu L, Qingliang W, Chunze Z, Shiyao F, Xinwei L, Xuanzhang H, Yucheng S (2023) Preparation of porous calcium carbonate biochar and its beryllium adsorption performance. J Environ Chem Eng 11:110102
Zhao X, Su Y, Hao X et al (2023) Effect of mechanical− chemical modification on adsorption of beryllium by calcite. Environ Sci Pollut Res 2023:1–13. https://doi.org/10.1007/s11356-023-27275-9
Xin Q, Wang Q, Gan J, Lei Z, Hu E, Wang H, Wang H (2023) Enhanced performance in uranium extraction by the synergistic effect of functional groups on chitosan-based adsorbent. Carbohyd Polym 300:120270
Matthias T (2016) Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC technical report). Chem Int News Mag IUPAC 38(1):1051–1069
Zhao X, Su Y, Lei Z, Wang H, Hu E, Hu F, Hao X (2022) Adsorptive removal of beryllium by Fe-modified activated carbon prepared from lotus leaf. Environ Sci Pollut Res 30:1–14
Martins AC, Pezoti O et al (2015) Removal of tetracycline by NaOH-activated carbon produced from macadamia nut shells: kinetic and equilibrium studies. Chem Eng J 260:291–299
Jiang K, Zhou K, Yang Y (2018) Removal of beryllium from industrial wastewater by ferrite precipitation process. Nonferrous Met (Smelting Section) 6:83–86
Choy SY, Prasad KN, Wu TY, Raghunandan ME, Ramanan RN (2016) Performance of conventional starches as natural coagulants for turbidity removal. Ecol Eng 94:352–364
Kumar PS, Kirupha SD et al (2011) Adsorption behavior of nickel(II) onto cashew nut shell: equilibrium, thermodynamics, kinetics, mechanism and process design. Chem Eng J 167:122–131
Chen Y, Li M, Li Y, Liu Y, Chen Y, Li H, Li L, Xu F, Jiang H, Chen L (2021) Hydroxyapatite modified sludge-based biochar for the adsorption of Cu2+ and Cd2+: adsorption behavior and mechanisms. Bioresour Technol 321:124413
Zheng X, He X, Peng H, Wen J, Lv S (2021) Efficient adsorption of ciprofloxacin using Ga2S3/S-modified biochar via the high-temperature sulfurization. Bioresour Technol 334:125238
Li X, Wang H et al (2019) Low temperature reduction of NO by activated carbons impregnated with Fe based catalysts(Article). Int J Hydrogen Energ 44(46):25265–25275
Pezoti O, Cazetta AL et al (2016) NaOH-activated carbon of high surface area produced from guava seeds as a high-efficiency adsorbent for amoxicillin removal: kinetic, isotherm and thermodynamic studies. Chem Eng J 288:778–788
Singha AS, Guleria A (2014) Application of vinyl monomers functionalized cellulosic biopolymer for removal of dissolved toxic metal ions from polluted water samples. J Environ Chem Eng 2(3):1456–1466
Jiayang L, Changwei H, Qingguo H (2018) Adsorption of Cu2+, Pb2+, and Cd2+ onto oiltea shell from water. Bioresour Technol 271:487–491
Liu Y, Liu Y (2008) Biosorption isotherms, kinetics and thermodynamics. Sep Purif Technol 61:229–242
Liu W, Zhang J, Zhang C, Wang Y, Li Y (2010) Adsorptive removal of Cr (VI) by Fe-modified activated carbon prepared from Trapa natans husk. Chem Eng J 162:677–684
Barkat M, Nibou D, Chegrouche S, Mellah A (2009) Kinetics and thermodynamics studies of chromium(VI) ions adsorption onto activated carbon from aqueous solutions. Chem Eng Process 48:38–47
Elena MI, Paola GS, Cinzia C, Giovanni D (2018) Enhanced lanthanum adsorption by amine modified activated carbon. Chem Eng J 341:75–82
Atkins P, Overton T (2010) Shriver and atkins’ inorganic chemistry. Oxford University Press, Oxford USA
Polowczyk I, Bastrzyk A, Fiedot M (2016) Protein-mediated precipitation of calcium carbonate. Materials 9(11):944
Al Isawi WA, Zeller M, Mezei G (2022) Supramolecular incarceration and extraction of tetrafluoroberyllate from water by nanojars. Inorg Chem 61(23):8611–8622
Acknowledgements
We thank the following funding agencies for supporting this work: Foundation of State Key Laboratory of Nuclear Resources and Environment (2020NRE02). Research on characteristic properties of typical radioactive solid waste and radiation protection regulation technology and operation management mechanism (2019YFC1907701). Scientific Research Project of Education Department of Hunan (Project no. 21C0297).
Funding
Foundation of State Key Laboratory of Nuclear Resources and Environment (2020NRE02). Ministry of Science and Technology of China (2019YFC1907701). Scientific Research Project of Education Department of Hunan (Project no. 21C0297).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by XZ, HX, ZL, YS, HW, EH, PH, FH, QW, CZ, BZ. The first draft of the manuscript was written by XZ and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, X., Xia, H., Su, Y. et al. Study on removal of beryllium from uranium beryllium ore wastewater by acid leaching activated carbon and its mechanism. J Radioanal Nucl Chem 332, 4231–4242 (2023). https://doi.org/10.1007/s10967-023-09122-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-09122-w