Abstract
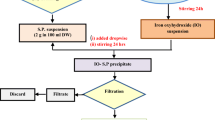
For employing strontium incorporated sodium iron titanate (Sr-SIT) in applications related to aqueous environments, the assessment of leach resistance of constituent elements assumes significance. The sodium iron titanate was prepared by the conventional co-precipitation method, and an ion exchange method was adopted for introducing strontium. 28-day-long Soxhlet leaching studies were undertaken for irradiated and unirradiated sintered solid pellets of Sr-SIT. The leaching propensities of the Sr-SIT constituents were analyzed by ICP-OES and neutron activation analysis respectively, for the unirradiated and irradiated Sr-SIT. These studies established the chemical stability of Sr-SIT towards leaching.





Similar content being viewed by others
References
Yao YFY, Kummer JT (1967) Ion exchange properties of and rates of ionic diffusion in beta-alumina. J Inorgan Nucl Chem 29:2453–2475
Goodenough JB, Hong HP, Kafalas JA (1976) Fast Na+-ion transport in skeleton structures. Mater Res Bul 11:203–220
Hayashi A, Masuzawa N, Yubuchi S, Tsuji F, Hotehama C, Sakuda A, Tatsumisago M (2019) A sodium-ion sulfide solid electrolyte with unprecedented conductivity at room temperature. Nat commun 10:5266
Sunatkari AL, Talwatkar SS, Bhaina AH (2023) A review on the development of na+ ion conducting solid electrolytes for batteries. IOP Publ J Phys Conf Ser 2426:012042
Stenina IA, Kozina LD, Kulova TL, Skundin AM, Chekannikov AA, Yaroslavtsev AB (2016) Synthesis and ionic conduction of sodium titanate Na2Ti3O7. Russian J InorgChem 61:1235–1240
Jia H, Peng L, Zhang Z, An T, Xie J (2020) Na3.8 [Sn0.67Si0.33] 0.8Sb0. 2S4: A quinary sodium fast ionic conductor for all-solid-state sodium battery. J Energ Chem 48:102–106
Zhang W, Li H, Zhang Z, Xu M, Lai Y, Chou SL (2020) Full activation of Mn4+/Mn3+ redox in Na4MnCr (PO4) 3 as a high-voltage and high-rate cathode material for sodium-ion batteries. Small 16:2001524
Sun Y, Wang Y, Liang X, Xia Y, Peng L, Jia H, Li H, Bai L, Feng J, Jiang H, Xie J (2019) Rotational cluster anion enabling superionic conductivity in sodium-rich antiperovskite Na3OBH4. J Am Chem Soc 141:5640–5644
Yang F, Zhang H, Li L, Reaney IM, Sinclair DC (2016) High ionic conductivity with low degradation in A-site strontium-doped nonstoichiometric sodium bismuth titanateperovskite. Chem Mater 28:5269–5273
Yang F, Wu P, Sinclair DC (2017) Enhanced bulk conductivity of A-site divalent acceptor-doped non-stoichiometric sodium bismuth titanate. Solid State Ionics 299:38–45
Kim JJ, Yoon K, Park I, Kang K (2017) Progress in the development of sodium-ion solid electrolytes. Small Methods 1:1700219
Shapovalov V, Kutukova K, Maletti S, Heubner C, Butova V, Shukaev I, Guda A, Soldatov A, Zschech E (2022) Laboratory X-ray microscopy study of microcrack evolution in a novel sodium iron titanate-based cathode material for Li-Ion batteries. Crystals 12:3
Sarkar A, ManoharCV MS (2020) A simple approach to minimize the first cycle irreversible loss of sodium titanate anode towards the development of sodium-ion battery. Nano Energy 70:104520
Shapovalov V, Guda A, Butova V, Shukaev I, Soldatov A (2021) Laboratory operando XAS study of sodium iron titanite cathode in the Li-ion half-cell. Nanomaterials 11:156
Guimarães ACR, Costa KÁ, de Miranda RM, Santana CSA, Castro CD (2021) Study of controlled leaching process of steel slag in Soxhlet extractor aiming employment in pavements. Transp Geotech 27:100485
Amesh P, Suneesh AS, Venkatesan KA, Maheswari RU, Vijayalakshmi S (2020) Preparation and ion exchange studies of cesium and strontium on sodium iron titanate. Sep Purif Technol 238:116393
Lindsay SA, Sriram S, Chand M, Annapoorani S, Lakshmi KU, Jena H, Vijayalakshmi S, Kumar R, Jayaraman V (2023) Evaluation of leaching behaviour of Nd from Ca10-x Nd x(PO4)F2,(x= 0–1.2) matrix and its borosilicate glass-bonded analogues by neutron activation and ICP-OES analysis. Chem Pap 77:509–516
Akieh MN, Lahtinen M, Väisänen A, Sillanpää M (2008) Preparation and characterization of sodium iron titanate ion exchanger and its application in heavy metal removal from waste waters. J Hazard Mat 152:640–647
Lehto J, Clearfield A (1987) The ion exchange of strontium on sodium titanate Na4 Ti9 O20. xH2 O. J Radioanal Nucl Chem 118:1–13
Yang D, Zheng Z, Liu H, Zhu H, Ke X, Xu Y, Wu D, Sun Y (2008) Layered titanate nanofibers as efficient adsorbents for removal of toxic radioactive and heavy metal ions from water. J Phys Chem C 112:16275–16280
Kim G, Lee DS, Eccles H, Kim SM, Cho HU, Park JM (2022) Selective strontium desorption using synthesized sodium titanate in aqueous solution. RSC Adv 12:18936–18944
Merceille A, Weinzaepfel E, Barré Y, Grandjean A (2011) Effect of the synthesis temperature of sodium nonatitanate on batch kinetics of strontium-ion adsorption from aqueous solution. Adsorption 17:967–975
Amesh P, Venkatesan KA, Suneesh AS, Maheswari U (2021) Tuning the ion exchange behavior of cesium and strontium on sodium iron titanate. Sep Purif Technol 267:118678
(1981) A State-of-the- art review of materials properties of nuclear waste forms. PNL Report, U.S. Department of Energy, Report No. PNL-3802, UC-70
Agrawal S, Dhawan N (2021) Investigation of mechanical and thermal activation on metal extraction from red mud. Sustain Mater Technol 27:00246
Xu B, Smith P, Wingate C, De Silva L (2010) The effect of calcium and temperature on the transformation of sodalite to cancrinite in Bayer digestion. Hydrometallurgy 105:75–81
Smith P (2017) Reactions of lime under high temperature Bayer digestion conditions. Hydrometallurgy 170:16–23
Acknowledgements
Authors thank Dr. N. Sivaraman, Director, MC&MFCG for his constant support and encouragement.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors wish to confirm that there are no known conflicts of interest associated with this publication and there has been no significant financial support for this work that could have influenced its outcome.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Selvan, B.R., Sriram, S., Chand, M. et al. Preparation and Soxhlet leaching studies of strontium incorporated sodium iron titanate by ICP-OES and NAA methods. J Radioanal Nucl Chem 333, 1419–1426 (2024). https://doi.org/10.1007/s10967-023-09016-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-09016-x