Abstract
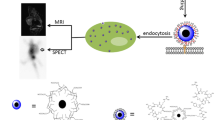
This study aimed to create a novel bimodal probe. For this purpose, cubic iron oxide nanoparticles (C-Fe3O4 NPs) conjugated with an EDTMP derivative called DPAPA thought to be a targeting tool for prostate cancer and bone metastases were studied. NPs were synthesized and conjugated with DPAPA, and then characterization studies were carried out. The particles were demonstrated to be cubic shaped, with an average size of 68.55 ± 1 0.03 (n = 3) nm. Then the nanoconjugates were radiolabeled with 99mTc. In vitro cellular affinities were conducted using PC-3, RWPE-1, LNCaP, K562, and Saos-2 cell lines. In conclusion, radio-nanoconjugates have the potential for bimodal MRI/SPECT imaging.









Similar content being viewed by others
Abbreviations
- C-Fe3O4 :
-
Cubic iron oxide
- C-Fe3O4-SiO2 :
-
Cubic iron oxide coated with TEOS
- C-Fe3O4-SiO2-HPG:
-
C-Fe3O4-SiO2 modified with hyperbranched polyglycerol (HPG)
- C-Fe3O4-SiO2-HPG-NH2 :
-
Coupled ethylene diamine with C-Fe3O4-SiO2-HPG
- C-Fe3O4-SiO2-HPG-NH2-DPAPA:
-
DPAPA conjugated with C-Fe3O4-SiO2-HPG-NH2 (NC)
- 99mTc-DPAPA-Fe3O4 :
-
Radiolabelled-nanocaonjugate (99mTc-C-Fe3O4-SiO2-HPG-NH2-DPAPA)
- DPAPA:
-
(S)-2,5-Bis(bis(phosphonomethyl)amino) pentanoic acid
- NPs:
-
Nanoparticles
- NC:
-
Nanoconjugate
References
Rawla P (2019) Epidemiology of prostate cancer. World J Oncol 10:63–89. https://doi.org/10.14740/wjon1191
Hensel J, Thalmann GN (2016) Biology of bone metastases in prostate cancer. Urology 92:6–13. https://doi.org/10.1016/j.urology.2015.12.039
Lübbe AS, Alexiou C, Bergemann C (2001) Clinical applications of magnetic drug targeting. J Surg Res 95:200–206. https://doi.org/10.1006/jsre.2000.6030
Sadri N, Moghadam M, Abbasi A (2018) MoO2(acac)2@Fe3O4/SiO2/HPG/COSH nanostructures: novel synthesis, characterization and catalyst activity for oxidation of olefins and sulfides. J Mater Sci Mater Electron 29:11991–12000. https://doi.org/10.1007/s10854-018-9301-z
Yurt Kilcar A, Biber Muftuler FZ, Enginar H et al (2014) Synthesis, characterization and biodistribution of 99mTc-Bioquin-HMPAO (99mTc-BH) as a novel brain imaging agent. J Radioanal Nucl Chem 302:563–573. https://doi.org/10.1007/s10967-014-3175-1
Shubayev VI, Pisanic TR, Jin S (2009) Magnetic nanoparticles for theragnostics. Adv Drug Deliv Rev 61:467–477. https://doi.org/10.1016/j.addr.2009.03.007
Abdeen S (2013) Diagnostics and treatment of metastatic cancers with magnetic nanoparticles. J Nanomed Biother Discov. https://doi.org/10.4172/2155-983X.1000115
Hu X, Zhou L, Gao C (2011) Hyperbranched polymers meet colloid nanocrystals: a promising avenue to multifunctional, robust nanohybrids. Colloid Polym Sci 289:1299–1320. https://doi.org/10.1007/s00396-011-2457-1
Jahandar M, Zarrabi A, Shokrgozar MA, Mousavi H (2015) Synthesis, characterization and application of polyglycerol coated Fe3O4 nanoparticles as a nano-theranostics agent. Mater Res Express 2:125002. https://doi.org/10.1088/2053-1591/2/12/125002
Fidler M (1981) Incidence of fracture through metastases in long bones. Acta Orthop Scand 52:623–627. https://doi.org/10.3109/17453678108992157
Robins HI, D’Oleire F, Grosen E, Spriggs D. Rationale and clinical status of 418 degrees C systemic hyperthermia tumor necrosis factor, and melphalan for neoplastic disease. Anticancer Res 17:2891–2894
Martinez-Boubeta C, Simeonidis K, Makridis A et al (2013) Learning from nature to improve the heat generation of iron-oxide nanoparticles for magnetic hyperthermia applications. Sci Rep 3:1652. https://doi.org/10.1038/srep01652
Ünak P, Yasakçı V, Tutun E et al (2023) Multimodal radiobioconjugates of magnetic nanoparticles labeled with 44sc and 47sc for theranostic application. Pharmaceutics 15:850. https://doi.org/10.3390/pharmaceutics15030850
Babu P, Sinha S, Surolia A (2007) Sugar−quantum dot conjugates for a selective and sensitive detection of lectins. Bioconjug Chem 18:146–151. https://doi.org/10.1021/bc060204q
Yasakci V, Tekin V, Guldu OK et al (2018) Hyaluronic acid-modified [19F]FDG-conjugated magnetite nanoparticles: in vitro bioaffinities and HPLC analyses in organs. J Radioanal Nucl Chem 318:1973–1989. https://doi.org/10.1007/s10967-018-6282-6
Bahrami-Samani A, Ghannadi-Maragheh M, Jalilian AR et al (2009) Production, quality control and biological evaluation of 153sm-EDTMP in wild-type rodents. Iran J Nucl Med 17:12–19
Heister E, Neves V, Tîlmaciu C et al (2009) Triple functionalisation of single-walled carbon nanotubes with doxorubicin, a monoclonal antibody, and a fluorescent marker for targeted cancer therapy. Carbon N Y 47:2152–2160. https://doi.org/10.1016/j.carbon.2009.03.057
Tutun E, Tekin V, Yasakcı V et al (2021) Synthesis and morphological studies of Tc-99m-labeled lupulone-conjugated Fe3O4 @TiO 2 nanocomposite, and in vitro cytotoxicity activity on prostate cancer cell lines. Appl Organomet Chem. https://doi.org/10.1002/aoc.6435
Rouhrazi H, Turgan N, Oktem G (2018) Zoledronic acid overcomes chemoresistance by sensitizing cancer stem cells to apoptosis. Biotech Histochem 93:77–88. https://doi.org/10.1080/10520295.2017.1387286
Islami M, Zarrabi A, Tada S et al (2018) Controlled quercetin release from high-capacity-loading hyperbranched polyglycerol-functionalized graphene oxide. Int J Nanomed 13:6059–6071. https://doi.org/10.2147/IJN.S178374
Shapiro EM, Skrtic S, Koretsky AP (2005) Sizing it up: cellular MRI using micron-sized iron oxide particles. Magn Reson Med 53:329–338. https://doi.org/10.1002/mrm.20342
Tang F, Li L, Chen D (2012) Mesoporous silica nanoparticles: synthesis, biocompatibility and drug delivery. Adv Mater 24:1504–1534. https://doi.org/10.1002/adma.201104763
Wang S-Y, Chen X-X, Li Y, Zhang Y-Y (2016) Application of multimodality imaging fusion technology in diagnosis and treatment of malignant tumors under the precision medicine plan. Chin Med J (Engl) 129:2991–2997. https://doi.org/10.4103/0366-6999.195467
Zhang P, Li Y, Tang W et al (2022) Theranostic nanoparticles with disease-specific administration strategies. Nano Today 42:101335. https://doi.org/10.1016/j.nantod.2021.101335
Wang A-Y, Kuo C-L, Lin J-L et al (2010) Study of magnetic ferrite nanoparticles labeled with 99mTc-pertechnetate. J Radioanal Nucl Chem 284:405–413. https://doi.org/10.1007/s10967-010-0488-6
Sigma-Aldrich IR Spectrum Table & Chart. https://www.sigmaaldrich.com/technical-documents/articles/biology/ir-spectrum-table.html. Accessed 10 Dec 2020
Rainer E. Glaser vıbratıonal spectroscopy tutorıal: sulfur and phosphorus. https://faculty.missouri.edu/~glaserr/8160f10/A03_Silver.pdf. Accessed 10 Dec 2020
Cao J (2004) Preparation and radiolabeling of surface-modified magnetic nanoparticles with rhenium-188 for magnetic targeted radiotherapy. J Magn Magn Mater 277:165–174. https://doi.org/10.1016/S0304-8853(03)00894-1
Liu X, Kaminski MD, Guan Y et al (2006) Preparation and characterization of hydrophobic superparamagnetic magnetite gel. J Magn Magn Mater 306:248–253. https://doi.org/10.1016/j.jmmm.2006.03.049
Medine EI, Ünak P, Sakarya S, Özkaya F (2011) Investigation of in vitro efficiency of magnetic nanoparticle-conjugated 125I-uracil glucuronides in adenocarcinoma cells. J Nanopart Res 13:4703–4715. https://doi.org/10.1007/s11051-011-0436-6
Xu H, Tong N, Cui L et al (2007) Preparation of hydrophilic magnetic nanospheres with high saturation magnetization. J Magn Magn Mater 311:125–130. https://doi.org/10.1016/j.jmmm.2006.11.173
Aziz M, Ismail AF (2017) X-ray photoelectron spectroscopy (XPS). In: Membrane characterization. Elsevier, pp 81–93
Lowry GV, Hill RJ, Harper S et al (2016) Guidance to improve the scientific value of zeta-potential measurements in nanoEHS. Environ Sci Nano 3:953–965. https://doi.org/10.1039/C6EN00136J
Häfeli UO, Sweeney SM, Beresford BA et al (1995) Effective targeting of magnetic radioactive90Y-microspheres to tumor cells by an externally applied magnetic field. Preliminary in vitro and in vivo results. Nucl Med Biol 22:147–155. https://doi.org/10.1016/0969-8051(94)00124-3
Munir T, Mahmood A, Fakhar-e-Alam M et al (2019) Treatment of breast cancer with capped magnetic-NPs induced hyperthermia therapy. J Mol Struct 1196:88–95. https://doi.org/10.1016/j.molstruc.2019.06.067
Liu XL, Fan HM, Yi JB et al (2012) Optimization of surface coating on Fe3O4 nanoparticles for high performance magnetic hyperthermia agents. J Mater Chem 22:8235–8244. https://doi.org/10.1039/c2jm30472d
Chapman MC, Lee AY, Hayward JH et al (2020) Superparamagnetic iron oxide sentinel node tracer injection: effects on breast MRI quality. J Breast Imaging 2:577–582. https://doi.org/10.1093/jbi/wbaa083
Krischer B, Forte S, Niemann T et al (2018) Feasibility of breast MRI after sentinel procedure for breast cancer with superparamagnetic tracers. Eur J Surg Oncol 44:74–79. https://doi.org/10.1016/j.ejso.2017.11.016
Aras O, Pearce G, Watkins AJ et al (2018) An in-vivo pilot study into the effects of FDG-mNP in cancer in mice. PLoS ONE 13:e00202482. https://doi.org/10.1371/journal.pone.0202482
Kim BM, Lee DR, Park JS et al (2016) Liquid crystal nanoparticle formulation as an oral drug delivery system for liver-specific distribution. Int J Nanomed. https://doi.org/10.2147/IJN.S97000
Arias-Ramos N, Ibarra LE, Serrano-Torres M et al (2021) Iron oxide incorporated conjugated polymer nanoparticles for simultaneous use in magnetic resonance and fluorescent imaging of brain tumors. Pharmaceutics 13:1258. https://doi.org/10.3390/pharmaceutics13081258
Zheltova V, Vlasova A, Bobrysheva N et al (2020) Fe3O4@HAp core–shell nanoparticles as MRI contrast agent: Synthesis, characterization and theoretical and experimental study of shell impact on magnetic properties. Appl Surf Sci 531:147352. https://doi.org/10.1016/j.apsusc.2020.147352
Ariza de Schellenberger A, Kratz H, Farr T et al (2016) Labeling of mesenchymal stem cells for MRI with single-cell sensitivity. Int J Nanomed. https://doi.org/10.2147/IJN.S101141
Payolla FB, Massabni AC, Orvig C (2019) Radiopharmaceuticals for diagnosis in nuclear medicine: A short review. Eclet Quim 44:11–19. https://doi.org/10.26850/1678-4618eqj.v44.3.2019.p11-19
Heery CR, Madan RA, Stein MN et al (2016) Samarium-153-EDTMP (Quadramet®) with or without vaccine in metastatic castration-resistant prostate cancer: a randomized Phase 2 trial. Oncotarget 7:69014–69023. https://doi.org/10.18632/oncotarget.10883
Acknowledgements
This work was supported financially by TUBITAK (The Scientific and Technological Research Council of Turkey) within the scope of the Bilateral Cooperation Project No. 218S749. The authors thank TUBITAK for the support. Volkan Yasakçı was also supported through the 100/2000 CoHE Doctorate Scholarship and Turkish Higher Education Council (YÖK) 100/2000 Scholarship. The authors thank the Turkish Higher Education Council (YÖK) and 100/2000 CoHE Ph.D. for their support. Dr. Omer Aras was partially supported through the NIH/NCI Cancer Support Grant P30 CA008748. The work was partially presented at the International Conference on Clinical PET-CT and Molecular Imaging in the Era of Theranostics, IPET-2020, IAEA-CN-285/52, and the International Nuclear Sciences and Technologies Conference (INSTEC-22).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.






Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yasakçı, V., Tutun, E., Karatay, K.B. et al. A novel SPECT/MRI bimodal imaging probe: 99mTc-DPAPA-Fe3O4 nanoconjugate. J Radioanal Nucl Chem 332, 4651–4666 (2023). https://doi.org/10.1007/s10967-023-08875-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-023-08875-8