Abstract
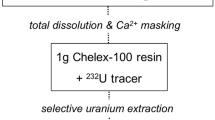
Τhe determination of uranium in calcareous samples has been investigated by means of alpha-spectroscopy after dissolution of the samples by nitrilotriacetic solution (NTA) and uranium separation by cation-exchange. Method recovery, uranium analysis and isotopic ratio composition of the samples was performed using a standard tracer solution (U-232). Comparison of the data with corresponding data obtained from experiments after EDTA-dissolution indicated lower selectivity for uranium, but higher recovery yields for phosphate rock samples. Moreover, using NTA results in the co-extraction of radium and thorium isotopes which are present in the samples, assuming broader applicability of the present method.


Similar content being viewed by others
References
Lysandrou M, Pashalidis I (2008) Uranium chemistry in stack solutions and leachates of phosphogypsum disposed at a coastal area in Cyprus. J Environ Radioact 99(2):359–366
Gorecka H, Gorecki H (1984) Determination of uranium in phosphogypsum. Talanta 31(12):1115–1117
van Calsteren P, Thomas L (2006) Uranium-series dating applications in natural environmental science. Earth-Sci Rev 75:155–175
Hennig GJ, Bangert U, Herr W, Freundlich J. Uranium series dating of calcite formations in caves: recent results and a comparative study on age determinations via 230Th/234U, 14C, TL and ESR. ArchéoSciences, revue d'Archéométrie. 1980;4(1):91-100.
Sanna L, Saez F, Simonsen S, Constantin S, Calaforra J-M, Forti P, Lauritzen S-E (2010) Uranium-series dating of gypsum speleothems: methodology and examples. Int J Speleol 39(1):35–46
Zhao M-Y, Zheng Y-F, Zhao Y-Y (2016) Seeking a geochemical identifier for authigenic carbonate. Nat Commun 7:10885. doi: https://doi.org/10.1038/ncomms10885
Kitano Y, Oomori T (1971) The Coprecipitation of Uranium with Calcium Carbonate. J Oceanogr Soc Jpn 27(1):34–42
Rajkovic MB, Karljikovic-Rajic K, Vladjisavlevic GT, Ciric IS (1999) Investigation of Radionuclides in Phosphogypsum. Measur Tech 42:299–305
Singhal RK, Sharma PK, Bassan MKT, Basu H, Reddy AVR (2011) Comparative determination of uranium in rock phosphates and columbite by ICP-OES, alpha & gamma spectrometry. J Radioanal Nucl Chem 288:149–156
Andreou G, Efstathiou E, Pashalidis I (2012) A simplified determination of uranium in phosphate rock and phosphogypsum by alpha-spectroscopy after its separation by liquid-extraction. J Radioanal Nucl Chem 291:865–867
El-Afifi E, Hilal MA, Attallah MF, EL-Reefy SA (2009) Characterization of phosphogypsum wastes associated with phosphoric acid and fertilizers production. J Env Rad 100:407–412
Mazzilli B, Palmiro V, Saueia C, Nisti MB (2018) Radiochemical characterization of Brazilian phosphogypsum. J Environ Radioact 49:113–122
Trifi M, Dermech C, Abdelkrim C, Azouzi R, Hjiri B (2018) Extraction procedures of toxic and mobile heavy metal fraction from complex mineralogical tailings affected by acid mine drainage. Arab J Geosci 11:328
Jagetiya B, Sharma A (2013) Optimization of chelators to enhance uranium uptake from tailings for phytoremediation. Chemosphere 91:692–696
Ji Y, Yun C, Le J, Qian M, Huan Y, Yang W, Yin X, Liu Y, Wang X (2017) Highly effective scale inhibition performance of amino trimethylenephosphonic acid on calcium carbonate. Desalination 422:165–173
Papanicolaou F, Antoniou S, Pashalidis I (2009) Experimental and theoretical studies on physico-chemical parameters affecting the solubility of phosphogypsum. J Environ Radioact 100:601–605
Paschalidou P, Pashalidis I (2019) Selective separation and determination of uranium in calcite and gypsum after EDTA-mediated sample dissolution and cation-exchange. J Radioanal Nucl Chem 320:807–812
Kiliari T, Pashalidis I (2010) Simplified alpha-spectroscopic analysis of uranium in natural waters after its separation by cation-exchange. Radiat Meas 45:966–968
Pashalidis I, Tsertos H (2004) Radiometric determination of uranium in natural waters after enrichment and separation by cation-exchange and extraction techniques. J Radioanal Nucl Chem 260:439–442
Kiliari T, Pashalidis I (2010) Alpha spectroscopic analysis of actinides (Th, U and Pu) after separation from aqueous solutions by cation-exchange and liquid extraction. J Radioanal Nucl Chem 284:547–551
Anderegg G (1982) Critical survey of stability constants of NTA complexes. Pure Appl Chem 54:2693–2758
Kiliari T, Pashalidis I (2012) Selective separation of actinyl(V,VI) cations from aqueous solutions by Chelex-100. Radiochim Acta 100:439–444
Paschalidou P, Pashalidis I (2019) Recovery of uranium from phosphate rock with EDTA-mediated dissolution and cation exchange. Hydrometallurgy 189:105118
Paschalidou P, Pashalidis I (2019) Alpha-spectroscopic analysis of uranium in ground- and seawater samples after EDTA-masking of interfering cations. J Radioanal Nucl Chem 320:807–812
Azouazi M, Ouahidi Y, Fakhi S, Andres Y, Abbe J, Ch, Benmansour M (2001) Natural radioactivity in phosphates, phosphogypsum and natural waters in Morocco. J Environ Radioact 54:231–242
Rutherford PM, Dudas MJ, Samek RA (1994) Environmental impacts of phosphogypsum. Sci Total Environ 149:1–38
Ptáček P (2016) ‘Phosphate Rocks’. In: Ptacek P (ed) Apatites and their Synthetic Analogues - Synthesis, Structure, Properties and Applications. IntechOpen, London
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Michael, A., Pashalidis, I. Determination of uranium in calcareous rocks/minerals after separation by nitrilotriacetic-mediated sample dissolution and cation-exchange. J Radioanal Nucl Chem 332, 1399–1403 (2023). https://doi.org/10.1007/s10967-022-08643-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-022-08643-0