Abstract
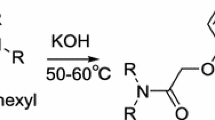
The liquid–liquid extraction, recovery of Np(VI) and Np(V) from the solution of Na2CO3 by methyltrioctylammonium carbonate in toluene was investigated. By the graphic slope analyses method, as well as by recording of absorption spectra for obtained solutions and method of calculated determination of solution properties taking into account water activity, composition was established for the carbonate complexes of Np(VI) and Np(V) being extracted, which, depending on extraction conditions, can be expressed by the following formulae’s: (R4N)2[NpVIO2(CO3)2], (R4N)3[NpVO2(CO3)2], (R4N)6[NpVIO2(CO3)4], (R4N)2[NpVIO2(CO3)2]·2(R4N)2CO3, (R4N)5[NpVO2(CO3)3], (R4N)7[NpVO2(CO3)4], (R4N)3[NpVO2(CO3)2]·(1–2)(R4N)2CO3, where R4N is the quaternary ammonium cation. The results have been obtained in the experiments, has much interest both in the field of alkaline radioactive waste management and with respect to their usage for extraction of transuranium elements during reprocessing of alkaline radioactive waste, as well as for liquid–liquid extraction purification of uranium and plutonium from admixtures of fission products. These techniques are used within the framework of a new alternative method for reprocessing of spent nuclear fuel in carbonate media the CARBEX (CARBonate EXtraction) process.







Similar content being viewed by others
References
Tomiyasu H, Asano Y (1998) Environmentally acceptable nuclear fuel cycle development of a new reprocessing system. Prog Nucl Energy 32(3–4):421–427
Asanuma N, Harada M, Ikeda Y, Tomiyasu H (2001) New approach to the nuclear fuel reprocessing in non-acidic aqueous solutions. Nucl Sci Tech 38(10):866–871
Kim KW, Kim YH, Kim SM et al (2008) A study on a process for recovery of uranium alone from spent nuclear fuel in a high alkaline carbonate media. NRC 7, Budapest, Hungary
Kim KW, Lee EH, Chung DY et al (2009) Process for recovering isolated uranium from spent nuclear fuel using a highly alkaline carbonate solution. Patent US2009269261A1
Goff GS, Brodnax LF, Cisneros MR et al (2007) Development of a novel alkaline based process for spent nuclear fuel recycling. In: AIChE annual meeting, Salt Lake City, United States
Peper SM, Brodnax LF, Field SE et al (2004) Kinetic study of the oxidative dissolution of UO2 in aqueous carbonate media. Ind Eng Chem Res 43(26):8188–8193
Soderquist C, Hanson B (2010) Dissolution of spent nuclear fuel in carbonate–peroxide solution. J Nucl Mater 396(2–3):159–162
Soderquist CZ, Johsen AM, McNamara BK et al (2011) Dissolution of irradiated commercial UO2 fuels in ammonium carbonate and hydrogen peroxide. J Ind Eng Chem Res 50:1813–1818
Nogami M, Kim SY, Asanuma N, Ikeda Y (2004) Adsorption behavior of amidoxime resin for separating actinide elements from aqueous carbonate solutions. J Alloys Compd 374(1–2):269–271
Kim KW, Lee JW, Chung DY et al (2012) Preparation of uranium oxide powder for nuclear fuel pellet fabrication with uranium peroxide recovered from uranium oxide scraps by using a carbonate–hydrogen peroxide solution. J Radioanal Nucl Chem 292:909–916
Stepanov SI, Chekmarev AM (2008) Concept of spent nuclear fuel reprocessing. Dokl Chem 423(1):276–278
Stepanov SI, Chekmarev AM (2013) CARBEX application prospects in the reprocessing of RBN spent nuclear fuel. Issues of atomic science and technology. Series of materials science and new materials 2(75):108–115
Htun S, Boyarintsev AV, Aung MM et al (2012) Solvent extraction of uranium(VI) from fluoride–carbonate solutions by methyltrioctylammonium fluoride. Prog Chem Technol 26(6):118–122
Boyarintsev AV, Stepanov SI, Chekmarev AM, Tsivadze AY (2020) Reprocessing of fluorination ash surrogate in the CARBOFLUOREX process. Nucl Eng Tech 52(1):109–114
Stepanov SI, Chekmarev AM, Boyarintsev AV, Tsivadze AY (2018) Set of SNF reprocessing concepts on the basis of the CARBEX process. In: 9th Russian conference on radiochemistry, September 17–21, 2018, St. Petersburg, Russia
Rovny SI, Shevtsev PP (2007) Current status of the radiochemical technology of U and Pu extraction and purification and potential of its development. Radiat Saf Issues 2(46):5–13
Fanghänel T, Glatz JP, Konings RJM et al (2010) Transuranium elements in the nuclear fuel cycle. In: Cacuci DG (ed) Handbook of nuclear engineering. Springer, Boston
Yoshida Z, Johnson SG, Kimura T, Krsul JR (2008) Neptunium. In: Morss LR, Edelstein NM, Fuger J (eds) The chemistry of the actinide and transactinide elements. Springer, Dordrecht
Altmaier M, Gaona X, Fanghänel T (2013) Recent advances in aqueous actinide chemistry and thermodynamics. Chem Rev 113(2):901–943
Goff GS, Brodnax LF, Cisneros MR, Runde WH (2007) Redox chemistry of actinides in peroxide–carbonate media: applications to developing a novel process for spent nuclear fuel reprocessing. In: AIChE annual meeting, Salt Lake City, United States
Karalova ZK, Bukina TV, Myasoedov BF, Rodionova LM (1985) The use of decylamine for the extraction of actinides and lanthanides from carbonate solutions. Sov Radiochem 27(1):41–44
Stepanov SI, Chekmarev AM (2004) Extraction of rare metals by the salts of quaternary ammonium bases. IzdAT, Moscow
Ueno K, Saito A (1971) Extraction of several elements with trioctylmonomethylammonium chloride. Anal Chim Acta 56(3):427–434
Gromov BV (1978) Introduction to the chemical technology of uranium. Atomizdat, Moscow
Alekseeva NA, Bulyanitsa LS, Kovalskaya MP (1974) On extraction of certain actinides by methyltrioctylammonium carbonate. Sov Radiochem 16(5):575–580
Goff GS, Long KM, Reilly SD et al (2012) Americium/lanthanide separation in alkaline solution for advanced nuclear fuel cycles. In: 36th actinide separations conference, Chattanooga, United States
Boyarintsev AV, Kostikova GV, Stepanov SI et al (2020) Liquid–liquid extraction of trivalent americium from carbonate and carbonate–peroxide aqueous solutions by methyltrioctylammonium carbonate in toluene. J Radioanal Nucl Chem 324:1031–1038
Pilgrim CD, Zavarin M, Casey WH (2017) Pressure dependence of carbonate exchange with [NpO2(CO3)3]4− in aqueous solutions. Inorg Chem 56(1):661–666
Nitsche H, Standifer EM, Silva RJ (1990) Neptunium complexation with carbonate. Lanthan Actin Res 3(3):203–211
Moskvin AI (1971) Complex formation of neptunium(IV, V, VI) in carbonate solutions. Sov Radiochem 13(5):694–699
Wester DW, Sullivan JC (1981) Electrochemical and spectroscopic studies on neptunium(VI) -(V) and -(IV) in carbonate–bicarbonate buffers. Inorg Nucl Chem 43(11):2919
Grenthe I, Riglet C, Vitorge P (1986) Studies of metal complexes 14. Composition and equilibria of trinuclear neptunium(VI)- and plutonium(VI)–carbonate complex. Inorg Chem 25(10):1679–1684
Pratopo MI, Moriyama H, Higashi K (1993) Carbonate complexation of Np(VI) in near-neutral solutions. J Nucl Sci Technol 30(10):1024–1029
Tait CD, Palmer PD, Ekberg SA, Clark DL (1995) Report on neptunium speciation by NMR and optical spectroscopies. LA–13012–M
Hagan PG, Cleveland JM (1966) The absorption spectra of neptunium ions in perchloric acid solution. Inorg Nucl Chem 28(12):2905–2909
Nitsche H, Muller A, Standifer EM et al (1992) Dependence of actinide solubility and speciation on carbonate concentration and ionic strength in groundwater. Radiochim Acta 58(1):27–32
Neck V, Kim JI, Kanellakopulos B (1992) Solubility and hydrolysis behavior of neptunium(V). Radiochim Acta 56:25–30
Runde W (1993) Zum chemischen verhalten von drei- und funfwertigem americium in salinen NaCl–losungen. Ph.D. thesis, Technischen Universitat Munchen, Germany
Varlashkin PG, Hobart DE, Begun GM, Peterson JR (1984) Electrochemical and spectroscopic studies of neptunium in concentrated aqueous carbonate and carbonate–hydroxide solutions. Radiochim Acta 35:91–96
Mikulin GI (1968) Physical chemistry of electrolyte solutions. Khimiya, Leningrad
Acknowledgements
The authors express their deep gratitude to Alexander Mikhailovich Chekmarev for his exceptionally well-imed and useful remarks provided during the writing of this article, and express their heartfelt condolences for his untimely departure from our life. The work was supported by Mendeleev University of Chemical Technology. Project No. T–2020–009.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by AVB and SAP. The first draft of the manuscript was written by AVB and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. Conceptualization: SIS and AVB; Methodology: SAP and AVB; Formal analysis and investigation: SAP and AVB; Writing-original draft preparation: AVB; Writing-review and editing: SIS and AMC; Funding acquisition: SIS and AMC; Resources and supervision: SAP.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Boyarintsev, A.V., Perevalov, S.A., Stepanov, S.I. et al. Liquid–liquid extraction of neptunium(VI) and neptunium(V) from carbonate solutions by methyltrioctylammonium carbonate in toluene. J Radioanal Nucl Chem 327, 385–393 (2021). https://doi.org/10.1007/s10967-020-07487-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-020-07487-w