Abstract
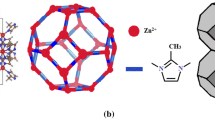
Crystalline solids of zeolitic imidazolate framework (ZIF)-69 with a suitable pore window size matching with atomic diameter of xenon is utilized for this task, showing notable performance for Xe/Kr capture and separation as well as decent hydrolytic and irradiation stabilities. The results illustrate that the saturated uptake amount of Xe by ZIF-69 is 2.46 mmol/g at 298 K, while Kr uptake is only 0.55 mmol/g. Importantly, the separation ratio of Xe/Kr is 8.35, one of the highest values among all solid sorbent material reported up to now. These findings suggest that ZIF-69 is a potential candidate for the application of xenon capture and purification form a gas mixture containing Kr.





Similar content being viewed by others
References
Z IAEA (2002) Issues and trends in radioactive waste management. Proceeding Series, STI/PUB/1175, IAEA, Vienna
Kharecha AP, Hansen EJ (2013) Prevented mortality and greenhouse gas emissions from historical and projected nuclear power. Environ Sci Technol 47:4889–4895
Ma F, Shi W, Meng H, Li Z, Zhou W, Zhang L (2016) Preparation, characterization and ion-exchange behavior of polyantimonic acid-polyacrylonitrile (PAA-PAN) composite beads for strontium(II). J Radioanal Nucl Chem 308(1):155–163
Hitoshi MIMURA, Kenichi AKIBA (1994) Separation of heatgenerating nuclides from high-level liquid wastes through zeolite columns. J Nucl Sci Technol 31:463–469
Banerjee D, Cairns AJ, Liu J, Motkuri RK, Nune SK, Fernandez CA, Krishna R, Strachan DM, Thallapally PK (2015) Potential of metal-organic frameworks for separation of xenon and krypton. Acc Chem Res 48:211–219
Soelberg NR, Garn TG, Greenhalgh MR, Law JD, Jubin R, Strachan DM (2013) Thallapally PK (2013) radioactive iodine and krypton control for nuclear fuel reprocessing facilities. Sci Technol Nucl Ins 2013:1–12
Chen XY, Plonka AM, Banerjee D, Krishna R, Schaef HT, Ghose S, Thallapally PK, Parise JB (2015) Direct Observation of Xe and Kr Adsorption in a Xe-Selective Microporous Metal-Organic Framework. J Am Chem Soc 137:7007–7010
Li LY, Guo LD, Zhang ZG, Yang QW, Yang YW, Bao Z, Ren QL, Li J (2019) A robust squarate-based metal-organic framework demonstrates record high affinity and selectivity for xenon over krypton. J Am Chem Soc 141:9358–9364
Banerjee D, Simon CM, Elsaidi SK, Haranczyk M, Thallapally PK (2018) Xenon gas separation and storage using metal-organic frameworks. Chem 4:1–29
Chen L, Reiss PS, Chong SY, Holden D, Jelfs KE, Hasell T, Little MA, Kewley A, Briggs ME, Stephenson A, Thomas KM, Armstrong JA, Bell J, Busto J, Noel R, Liu J, Strachan DM, Thallapally PK, Cooper AI (2014) Separation of rare gases and chiral molecules by selective binding in porous organic cages. Nat Mater 13:954–960
Fernandez CA, Liu J, Thallapally PK, Strachan DM (2012) Switching Kr/Xe Selectivity with Temperature in a Metal-Organic Framework. J Am Chem Soc 134:9046–9049
Li JL, Huang L, Zou XQ, Zheng AM, Li HY, Rong HZ, Zhu GS (2018) Porous organic materials with ultra-small pores and sulfonic functionality for xenon capture with exceptional selectivity. J Mater Chem A 6:11163–11168
Gong YJ, Tang YM, Mao ZH, Wu XN, Liu Q, Hu S, Xiong SS, Wang XL (2018) Metal-organic framework derived nanoporous carbons with highly selective adsorption and separation of xenon. J Mater Chem A 6:13696–13704
Banerjee D, Simon CM, Plonka AM, Motkuri RK, Liu J, Chen XY, Smit B, Parise JB, Haranczyk M, Thallapally PK (2016) Metal-organic framework with optimally selective xenon adsorption and separation. Nat Commun 7:11831
Thallapally PK, Grate JW, Motkuri RK (2012) Facile xenon capture and release at room temperature using a metal-organic framework: a comparison with activated charcoal. Chem Commun 48:347–349
Zhou HC, Long JR, Yaghi OM (2012) Introduction to metal-organic frameworks. Chem Rev 112:673–674
Burtch NC, Jasuja H, Walton KS (2014) Water stability and adsorption in metal-organic frameworks. Chem Rev 114:10575–10612
Wang CH, Liu XL, Demir NK, Chen JP, Li K (2016) Applications of water stable metal-organic frameworks. Chem Soc Rev 45:5107
Zhang H, Liu W, Li A, Zhang D, Li X, Zhai F, Chen L, Chen L, Wang Y, Wang S (2019) Three mechanisms in one material: uranium capture by a polyoxometalate-organic framework through combined complexation, chemical reduction, and photocatalytic reduction. Angew Chem Int Ed 58:1–6
Li H, Zhai FW, Gui DX, Wang XX, Wu CF, Duo Z, Xing D, Hong D, Su XT, Juan D, Zhang L, Chai ZF, Wang S (2019) Powerful uranium extraction strategy with combined ligand complexation and photocatalytic reduction by postsynthetically modified photoactive metal-organic frameworks. Appl Catal B 254:47–54
Liu W, Wang YL, Bai ZL, Li YX, Wang YX, Chen LH, Xu L, Diwu J, Chai ZF, Wang S (2017) Hydrolytically stable luminescent cationic metal organic framework for highly sensitive and selective sensing of chromate anions in natural water systems. ACS Appl Mater Interfaces 9:16448–16457
He C, Lu K, Lin W (2014) Nanoscale metal-organic frameworks for real-time intracellular pH sensing in live cells. J Am Chem Soc 136:12253–12256
Mason JA, Oktawiec J, Taylor MK, Hudson MR, Rodriguez J, Bachman JE, Gonzalez MI, Cervellino A, Guagliardi A, Brown CM, Llewellyn PL, Masciocchi N, Long JR (2015) Methane storage in flexible metal-organic frameworks with intrinsic thermal management. Nature 527:357–361
Ding ML, Flaig RW, Jiang HL, Yaghi OM (2019) Carbon capture and conversion using metal-organic frameworks and MOF-based materials. Chem Soc Rev 48:2783–2828
Lin YC, Kong CL, Zhang QJ, Chen L (2016) Metal-organic frameworks for carbon dioxide capture and methane storage. Adv Energy Mater 7:1601296
Kim JY, Oh H, Moon HR (2018) Hydrogen isotope separation in confined nanospaces: carbons, zeolites, metal-organic frameworks, and covalent organic frameworks. Adv Mater 31:1805293
Wang H, Yao KX, Zhang ZJ, Jagiello J, Gong QH, Han Y, Li J (2014) The first example of commensurate adsorption of atomic gas in a MOF and effective separation of xenon from other noble gases. Chem Sci 5:620
Yu GL, Liu YQ, Zou XQ, Zhao N, Rong HZ, Zhu GS (2018) A nanosized metal-organic framework with small pores for kinetic xenon separation. J Mater Chem A 6:11797
Magdysyuk OV, Denysenko D, Weinrauch I, Volkmer D, Hirscher M, Dinnebier RE (2015) Formation of a quasi-solid structure by intercalated noble gas atoms in pores of CuI-MFU-4l metal-organic framework. Chem Commun 51:714
Liu J, Thallapally PK, Strachan D (2012) Metal-organic frameworks for removal of Xe and Kr from nuclear fuel reprocessing plants. Langmuir 28:11584–11589
Perry JJ, Teich-McGoldrick SL, Meek ST, Greathouse JA, Haranczyk M, Allendorf MD (2014) Noble gas adsorption in metal-organic frameworks containing open metal sites. J Phys Chem C 118:11685–11698
Thallapally PK, Grate JW, Motkuri RK, (2012) Facile xenon capture and release at room temperature using a metal-organic framework: a comparison with activated charcoal. Chem Commun 48:347–349
Liu J, Strachan DM, Thallapally PK (2014) Enhanced noble gas adsorption in Ag@MOF-74Ni. Chem Commun 50:466–468
Banerjee R, Phan A, Wang B, Knobler C, Furukawa H, O’Keeffe M, Yaghi MO (2008) High-Throughput Synthesis of Zeolitic Imidazolate Frameworks and Application to CO2 Capture. Science 319:939–943
Phan A, Doonan CJ, Uribe-Romo FJ, Knobler BC, O’Keeffe M, Yaghi MO (2010) Synthesis, structure, and carbon dioxide capture properties of zeolitic imidazolate frameworks. Acc Chem Res 43:58–67
Xiong SS, Gong YJ, Hu SL, Wu XN, Li W, He YB, Chen BL, Wang XL (2018) A microporous metal-organic framework with commensurate adsorption and highly selective separation of xenon. J Mater Chem A 6:4752–4758
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (21906112, 21790374)
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, S., Ma, F., Wang, X. et al. Uptake and separation of Xe and Kr by a zeolitic imidazolate framework with a desirable pore window. J Radioanal Nucl Chem 324, 1275–1281 (2020). https://doi.org/10.1007/s10967-020-07147-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-020-07147-z