Abstract
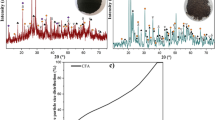
Radioactive liquid wastes containing large amounts of nitric acid (approx. 4–7 M) generated in the nuclear fuel cycle are very undesirable by-products. Denitration of highly acidic radioactive liquid wastes with organic reducing agents has several advantages, such as a significant pH increase, possibility of separation of radioisotopes which can be utilized in other technologies, reduction of volatility of radioisotopes and fixation of certain mono- and divalent radioisotopes with zeolite. In the present work some aspects of denitration of simulated radioactive liquid wastes at laboratory level were investigated. Denitration of HNO3 with formic acid was investigated for different [HCOOH]/[HNO3] ratios. The changes in the induction time, pH increase and volume reduction were determined. It was shown that by the denitration of simulated radioactive liquid waste high pH values could be achieved which in real systems would create more possibilities in radioactive waste management. Analysis of solid phases formed after the oxidation of excess HCOOH with H2O2 showed the presence of amorphous fraction as well as goethite and hematite phases, which could remove different radioactive cations from the liquid phase. The immobilization of simulated solid radioactive waste with borosilicate glass matrix was also shown.









Similar content being viewed by others
References
Ito K, Kanno T (1986) Effect of denitration with formic acid on precipitation and volatilization of ruthenium and technetium. J Nucl Sci Technol 23:37–43
Nakamura H, Yamaguchi I, Kubota M (1978) Effect of platinum group elements on denitration of high-level liquid waste with formic acid. J Nucl Sci Technol 15:760–764
Kondo Y, Kubota M (1992) Precipitation behavior of platinum group metals from simulated high-level liquid waste in sequential denitration process. J Nucl Sci Technol 29:140–148
Shirahashi K, Kubota M (1992) Precipitation behaviour of transuranium elements during denitration of high-level radioactive liquid wastes by formic acid. J Nucl Sci Technol 29:559–565
Pitt WW, Hancher CW, Patton RD (1981) Biological reduction of nitrates in wastewaters from nuclear processing using a fluidized-bed bioreactor. Nucl Chem Waste Manag 2:57–70
Shumate II SE, Hancher CW, Stranberg GW, Scott CD (1978) Biological process for environmental control of effluent streams in the nuclear fuel cycle. In: Waste management and fuel cycle’78 symposium, Tucson, Arizona, March 5–8, preprint
Cecille L, Kelm M (1986) Chemical reactions involved in the denitration process with HCOOH and HCHO. In: Cecile L, Halaszovich S (eds) Denitration of radioactive liquid waste. Graham & Trotman Ltd., London
Healy TV (1958) The reaction of nitric acid with formaldehyde and with formic acid and its application to the removal of nitric acid from mixtures. J Appl Chem 8:553–562
Kelm M, Oser B, Drobnik S, Deckwer WD (1980) Denitration of aqueous waste solutions from the nuclear fuel reprocessing. Nucl Technol 51:27–32
Holze K, Li Z, Oser B, Kelm M, Deckwer WD (1984) Occurrence of oscillations during denitration of nitric acid waste solutions containing metal salts. Ger Chem Eng 7:102–108
Drobnik S, Verfahren zum Entfernen von Salpetersaure und/oder Nitrat- und Nitrit-Ionen aus wässrigen Abfall-Lösungen. Patentschrift 1935273, Deutsches Patentamt, Bundesrepublik Deutschland
Musić S, Ristić M, Popović S (1989) Characterization of the precipitates formed during the denitration of simulated HRLW. J Radioanal Nucl Chem 134:153–365
Fletcher JM, Jenkins IL, Lever FM, Martin FS, Powell AR, Todd R (1955) Nitrato and nitro complexes of nitrosylruthenium. J Inorg Nucl Chem 1:378–401
Blasius E, Glatz J-P, Neumann W (1981) Ruthenium nitrosyl complexes in radioactive waste solutions of reprocessing plants. Radiochim Acta 29:159–166
Musić S, Wolf RHH, Šipalo-Žuljević J (1978) Sorption of trace amounts of ruthenium, manganese and europium on iron(III) hydroxide. Microchim Acta 70:303–308
Musić S, Gessner M, Wolf RHH (1979) Sorption of small amounts of cobalt(II) on iron(III) oxide. Microchim Acta 71:105–112
Musić S, Gessner M, Wolf RHH (1979) Sorption of ruthenium-97 on Fe2O3, Fe3O4 and Fe(OH)2. Microchim Acta 71:95–104
Musić S, Wolf RHH (1975) Sorption of microamounts of gallium on Fe(OH)3 and Fe2O3 precipitates. Microchim Acta 71:87–94
Musić S, Ristić M (1987) Adsorption of microamounts of ruthenium on hydrous iron oxides. J Radioanal Nucl Chem 109:495–506
Musić S, Ristić M (1988) Adsorption of trace elements or radionuclides on hydrous iron oxides. J Radioanal Nucl Chem 120:289–304
Musić S, Ristić M, Gotić M, Forić J (1988) Corrosion of simulated nuclear waste glass. J Radioanal Nucl Chem 122:91–102
Taragin MF, Eisenstein JC (1970) Mössbauer effect in some complex borosilicate glasses. J Non Cryst Solids 3:311–316
Eremyashev VE, Mironov AB (2015) Effect of Fe on the structure of potassium borosilicate glasses. Inorg Mater 51:177–181
Eremyashev VE, Shabunina LA, Mironov AB (2014) Structural particulars of iron-containing borosilicate glasses. Glass Ceram 70:391–394
Glazkova YS, Kalmykov SN, Presnyakov IA, Stefanovskaya OI, Stefanovsky SV (2015) The structural state of iron in multicomponent aluminium iron borosilicate glass depending on their composition and synthesis conditions. Glass Phys Chem 41:367–377
Tenney AS, Wong J (1972) Vibrational spectra of vapor-deposited binary borosilicate glasses. J Chem Phys 56:5516–5523
Sun Y, Zhang Z (2015) Structural roles of boron and silicon in the CaO–SiO2–B2O3 glasses using FT-IR, Raman and NMR spectroscopy. Metall Mater Trans B 46:1549–1554
Meikhail MS, Abdelghany AM (2017) Structure and electrical properties of iron borosilicate glasses. Silicon 9:895–900
Kamal H, Hezma AM (2018) Structure and physical properties of borosilicate as potential bioactive glass. Silicon 10:851–858
Madheshiya A, Dey KK, Ghosh M, Singh J, Gautam C (2019) Synthesis, structural, optical and solid state NMR study of lead, bismuth, titanate borosilicate glasses. J Non Cryst Solids 503–504:286–2896
Acknowledgements
The authors wish to thank Fedor Jakšić and Jasmin Forić for their assistance in the experiments.
Funding
This work was supported by Hrvatska Zaklada za Znanost (Grant No. IP-2016-06-8254).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ristić, M., Musić, S., Marciuš, M. et al. Denitration of simulated radioactive liquid waste. J Radioanal Nucl Chem 322, 1477–1485 (2019). https://doi.org/10.1007/s10967-019-06736-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-019-06736-x