Abstract
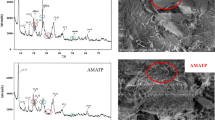
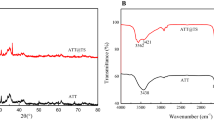
Melamine (MA)-trimesic acid (TMA) modified attapulgite (ATP–TMA–MA) with high adsorption capability for Sr(II) was successfully synthesized using attapulgite (ATP) as a matrix via a facile two-step approach. Batch adsorption experiments indicated that the adsorption kinetics followed the pseudo-second-order equation and isothermal adsorption data fitted well with the Freundlich isotherm model. The adsorption thermodynamic analysis indicated that Sr(II) adsorption onto ATP–TMA–MA was a spontaneous and endothermic process. Sr(II) adsorption mechanism of ATP–TMA–MA was mainly decided by chemisorption. TMA–MA molecules grafted on ATP surface markedly improve the removal efficiency of ATP–TMA–MA for Sr(II).









Similar content being viewed by others
References
Özeroğlu C, Bilgiç ÖD (2015) Use of the crosslinked copolymer functionalized with acrylic acid for the removal of strontium ions from aqueous solutions. J Radioanal Nucl Chem 305:551–565
Liu P, Jiang LP, Zhu LX, Guo JS, Wang AQ (2015) Synthesis of covalently crosslinked attapulgite/poly (acrylic acid-co-acrylamide) nanocomposite hydrogels and their evaluation as adsorbent for heavy metal ions. J Ind Eng Chem 23:188–193
Yin HB, Kong M, Gu XH, Chen H (2017) Removal of arsenic from water by porous charred granulated attapulgite-supported hydrated iron oxide in bath and column modes. J Clean Prod 166:88–97
Zhang ZZ, Sparks DL, Scrivner NC (1993) Sorption and desorption of quaternary amine cations on clays. Environ Sci Technol 27:1625–1631
Yi XS, Zhao ZW, Shi WX, Duan YS, Sun N, Ma C, Xie YZ (2013) Organic pollutants variation and antifouling enhancement with attapulgite clayaddition in MBR treating micro-polluted surface water. Chem Eng J 223:891–898
Zheng Y, Zhu YF, Wang AQ (2014) Highly efficient and selective adsorption of malachite green onto granular composite hydrogel. Chem Eng J 257:66–73
Han HX, Cheng C, Hu SH, Li XL, Wang WJ, Xiao CJ, Xu ZM, Shao DD (2017) Facile synthesis of gelatin modified attapulgite for the uptake of uranium from aqueous solution. J Mol Liq 234:172–178
Zhu LX, Guo JS, Liu P (2016) Effects of length and organic modification of attapulgite nanorods on attapulgite/polystyrene nanocomposite via in situ radical bulk polymerization. Appl Clay Sci 119:87–95
Yang MY, Wu XL, Xi XF, Zhang PJ, Yang XL, Lu RH, Zhou WF, Zhang SB, Gao HX, Li JY (2016) Using β-cyclodextrin/attapulgite-immobilized ionic liquid as sorbent in dispersive solid-phase microextraction to detect the benzoylurea insecticide contents of honey and tea beverages. Food chem 197:1064–1072
Li Q, Liu HN, Liu TY, Guo M, Qing BJ, Ye XS, Wu ZJ (2010) Strontium and calcium ion adsorption by molecularly imprinted hybrid gel. Chem Eng J 157:401–407
Shao DD, Hou GS, Li JX, Wen T, Ren XM, Wang XK (2014) PANI/GO as a super adsorbent for the selective adsorption of uranium(VI). Chem Eng J 255:604–612
Gao J, Shi YY, Zhou HF, Ren XQ, Ji H (2016) Application of modified attapulgite in phthalate acid ester-contaminated soil: effects on phthalate acid ester dissipation and the composition of soil microbial community. Environ Sci Pollut R 23:15175–15182
Chen LF, Liang HW, Lu Y, Cui CH, Yu SH (2011) Synthesis of an attapulgite clay@carbon nanocomposite adsorbent by a hydrothermal carbonization process and their application in the removal of toxic metal ions from water. Langmuir 27:8998–9004
Wang TT, Xuan RR, Ma JF, Tan Y, Jin ZF, Chen YH, Zhang LH, Zhang YK (2016) Using activated attapulgite as sorbent for solid-phase extraction of melamine in milk formula samples. Anal Bioanal Chem 408:6671–6677
Melataguia Tchieno FM, Guenang Sonfack L, Yme E, Ngameni E, Kenfack Tonle I (2017) Electroanalytical application of amine-grafted attapulgite to the sensitive quantification of the bioactive compound mangiferin. Electroanalysis 29:529–537
Pan DQ, Fan QH, Fan FY, Tang YF, Zhang YY, Wu WS (2017) Removal of uranium contaminant from aqueous solution by chitosan@ attapulgite composite. Sep Purif Technol 177:86–93
Majdan M, Pikus S, Gajowiak A, Sternik D, Zieba E (2010) Uranium sorption on bentonite modified by octadecyltrimethylammonium bromide. J Hazard Mater 184:662–670
Liu DC, Zheng HY (2017) Enhanced adsorption of radioactive strontium ions from aqueous solution by H2O2-modified attapulgite. J Radioanal Nucl Chem 311:1883–1890
Li B, Bai CY, Zhang S, Zhao XS, Li Y, Wang L, Ding K, Shu X, Li SJ, Ma LJ (2015) An adaptive supramolecular organic framework for highly efficient separation of uranium via an in situ induced fit mechanism. J Mater Chem A 3:23788–23798
Romero EG, Barrios MS, Revuelta MA (2004) Characteristics of a Mg-palygorskite in miocene rocks, madrid basin (Spain). Clay Clay Miner 52:484–494
Boudriche L, Calvet R, Hamdi B, Balard H (2012) Surface properties evolution of attapulgite by IGC analysis as a function of thermal treatment. Colloid Surface A 399:1–10
Zou XH, Pan JM, Ou HX, Wang X, Guan W, Li CX, Yan YS, Duan YQ (2011) Adsorptive removal of Cr(III) and Fe(III) from aqueous solution by chitosan/attapulgite composites: equilibrium, thermodynamics and kinetics. Chem Eng J 167:112–121
Huo Q, Dziri L, Desbat B, Russell KC, Leblanc RM (1999) Polarization-modulated infrared reflection absorption spectroscopic studies of a hydrogen-bonding network at the air-water interface. J Phys Chem B. 103:2929–2934
Jun YS, Lee EZ, Wang XC, Hong WH, Stucky GD (2013) A Thomas, From melamine-cyanuric acid supramolecular aggregates to carbon nitride hollow spheres. Adv Funct Mater 23:3661–3667
James WH III, Müller CW, Buchanan EG, Nix MGD, Guo L, Roskop L, Gordon MS, Slipchenko LV, Gellman SH, Zwier TS (2009) Intramolecular amide stacking and its competition with hydrogen bonding in a small foldamer. J Am Chem Soc 131:14243–14245
Lee HS, Wang YK, Hsu SL (1987) Spectroscopic analysis of phase separation behavior of model polyurethanes. Macromolecules 20:2089–2095
Huang JH, Liu YF, Jin QZ, Wang XG, Yang J (2007) Adsorption studies of a water soluble dye, Reactive Red MF-3B, using sonication-surfactant-modified attapulgite clay. J Hazard Mater 143:541–548
Wu WS, Fan QH, Xu JZ, Niu ZW, Lu SS (2007) Sorption-desorption of Th(IV) on attapulgite: effects of pH, ionic strength and temperature. Appl Radiat Isotopes 65:1108–1114
You J, Chen F, Zhao XB, Chen ZG (2010) Preparation, characterization and catalytic oxidation property of CeO2/Cu2+-attapulgite (ATP) nanocomposites. J Rare Earth 28:347–352
Jeon HJ, Yi SC, Oh SG (2003) Preparation and antibacterial effects of Ag-SiO2 thin films by sol-gel method. Biomaterials 24:4921–4928
Beijer FH, Kooijman H, Spek AL, Sijbesma RP, Meijer EW (1998) Self-complementarity achieved through quadruple hydrogen bonding. Angew Chem Int Edit 37:75–78
Giammar DE, Hering JG (2001) Time scales for sorption-desorption and surface precipitation of uranyl on goethite. Environ Sci Technol 35:3332–3337
Li CX, Zhang XJ, Pan JM, Xu PP, Liu Y, Yan YS, Zhang ZL (2009) Strontium(II) ion surface-imprinted polymers supported by potassium tetratitanate whiskers: synthesis, characterization and adsorption behaviours. Adsorpt Sci Technol 27:845–859
Sharma P, Tomar R (2008) Synthesis and application of an analogue of mesolite for the removal of uranium(VI), thorium(IV), and europium(III) from aqueous waste. Micropor Mesopor Mat 116:641–652
Tian GY, Wang WB, Zong L, Wang AQ (2017) MgO/palygorskite adsorbent derived from natural Mg-rich brine and palygorskite for high-efficient removal of Cd (II) and Zn (II) ions. J Environ Chem Eng 5:1027–1036
Tan KL, Hameed BH (2017) Insight into the adsorption kinetics models for the removal of contaminants from aqueous solutions. J Taiwan Inst Chem E 74:25–48
Wang H, Wang XJ, Ma JX, Xia P, Zhao JF (2017) Removal of cadmium (II) from aqueous solution: a comparative study of raw attapulgite clay and a reusable waste-struvite/attapulgite obtained from nutrient-rich wastewater. J Hazard Mater 329:66–76
Foo KY, Hameed BH (2010) Insights into the modeling of adsorption isotherm systems. Chem Eng J 156:2–10
Dolatyari L, Yaftian MR, Rostamnia S (2016) Removal of uranium (VI) ions from aqueous solutions using Schiff base functionalized SBA-15 mesoporous silica materials. J Environ Manage 169:8–17
Lalhmunsiama Tiwari D, Lee SM (2016) Surface-functionalized activated sericite for the simultaneous removal of cadmium and phenol from aqueous solutions: mechanistic insights. Chem Eng J 283:1414–1423
Singh KK, Singh AK, Hasan SH (2006) Low cost bio-sorbent ‘wheat bran’ for the removal of cadmium from wastewater: kinetic and equilibrium studies. Bioresour Technol 97:994–1001
Cui H, Qian Y, Li Q, Wei ZB, Zhai JP (2013) Fast removal of Hg(II) ions from aqueous solution by amine-modified attapulgite. Appl Clay Sci 72:84–90
Chu ZC, Liu JH, Han CL (2015) Removal of strontium ions from aqueous solution using hybrid membranes: kinetics and thermodynamics. Chin J Chem Eng 23:1620–1626
Chen Z, Wu Y, Wei Y, Mimura H (2016) Preparation of silica-based titanate adsorbents and application for strontium removal from radioactive contaminated wastewater. J Radioanal Nucl Chem 307:931–940
Lalhmunsiama Tiwari D, Lee SM (2015) Physico-chemical studies in the removal of Sr (II) from aqueous solutions using activated sericite. J Environ Radioact 147:76–84
Hu WY, Dong FQ, Yang GM, Peng X, Huang XJ, Liu MX, Zhang J (2018) Synergistic interface behavior of strontium adsorption using mixed microorganisms. Environ Sci Pollut R 25:22368–22377
Hafizi M, Abolghasemi H, Moradi M, Milani SA (2011) Strontium adsorption from sulfuric acid solution by Dowex 50 W-X resins. Chin J Chem Eng 19:267–272
Ryu J, Kim S, Hong HJ, Hong J, Kim M, Ryu T, Kim BG (2016) Strontium ion (Sr2+) separation from seawater by hydrothermally structured titanate nanotubes: removal vs Recovery. Chem Eng J 304:503–510
Asadollahi N, Yavari R, Ghanadzadeh H (2015) Preparation, characterization and analytical application of stannic molybdophosphate immobilized on multiwalled carbon nanotubes as a new adsorbent for the removal of strontium from wastewater. J Radioanal Nucl Chem 303:2445–2455
Zhang MD, Gu P, Zhang ZG, Liu L, Dong LH, Zhang GH (2018) Effective, rapid and selective adsorption of radioactive Sr2+ from aqueous solution by a novel metal sulfide adsorbent. Chem Eng J 351:668–677
Kaveeshwar AR, Kumar PS, Revellame ED, Gang DD, Zappi ME, Subramaniam R (2018) Adsorption properties and mechanism of barium (II) and strontium (II) removal from fracking wastewater using pecan shell based activated carbon. J Clean Prod 193:1–13
Sivaiah MV, Venkatesan KA, Krishna RM, Sasidhar P, Murthy GS (2005) Ion exchange properties of strontium on in situ precipitated polyantimonic acid in amberlite XAD-7. Sep Purif Technol 44:1–9
Lalhmunsiama Lalhriatpuia C, Tiwari D, Lee SM (2014) Immobilized nickel hexacyanoferrate on activated carbons for efficient attenuation of radio toxic Cs(I) from aqueous solutions. Appl Surf Sci 321:275–282
Batzias FA, Sidiras DK (2007) Simulation of dye adsorption by beech sawdust as affected by pH. J Hazard Mater 141:668–679
Ali SA, Haladu SA (2013) A novel cross-linked polyzwitterion/anion having pH-responsive carboxylate and sulfonate groups for the removal of Sr2+ from aqueous solution at low concentrations. React Funct Polym 73:796–804
Dong ZM, Qiu YF, Dai Y, Cao XH, Wang L, Wang PF, Lai ZJ, Zhang WL, Zhang ZB, Liu YH, Le ZG (2016) Removal of U (VI) from aqueous media by hydrothermal cross-linking chitosan with phosphate group. J Radioanal Nucl Chem 309:1217–1226
Zhang S, Zhao XS, Li B, Bai CY, Li Y, Wang L, Wen R, Zhang MC, Ma LJ, Li SJ (2016) “Stereoscopic” 2D super-microporous phosphazene-based covalent organic framework: design, synthesis and selective sorption towards uranium at high acidic condition. J Hazard Mater 314:95–104
Wan H, Que YG, Chen C, Wu ZW, Gu Z, Meng J, Wang L, Guan GF (2017) Preparation of metal-organic framework/attapulgite hybrid material for CO2 capture. Mater Lett 194:107–109
Ravikovitch PI, Neimark AV (2001) Characterization of nanoporous materials from adsorption and desorption isotherms. Colloid Surface A 187:11–21
Awual MR, Suzuki S, Aguchi T, Shiwaku H, Okamoto Y, Yaita T (2014) Radioactive cesium removal from nuclear wastewater by novel inorganic and conjugate adsorbents. Chem Eng J 242:127–135
Corma A, Mifsud A, Sanz E (1987) Influence of the chemical composition and textural characteristics of palygorskite on the acid leaching of octahedral cations. Clay Miner 22:225–232
Jiang JL, Duanmu C, Yang Y, Gu X, Chen J (2014) Synthesis and characterization of high siliceous ZSM-5 zeolite from acid-treated palygorskite. Powder Technol 251:9–14
Li HL, Li Y, Zhou YZ, Li BL, Liu DB, Liao HY (2019) Efficient removal of uranium using a melamine/trimesic acid-modified hydrothermal carbon-based supramolecular organic framework. J Colloid Interf Sci 544:14–24
Acknowledgements
This work is Supported by the Project of State Key laboratory of Environment-friendly Energy Materials, Southwest University of Science and Technology (No. 18zxhk15), and the PhD Fund of Southwest University of Science and Technology (No. 13zx7132).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, D., Zheng, H., Yang, W. et al. Efficient removal of Sr(II) from aqueous solution by melamine-trimesic acid modified attapulgite composite. J Radioanal Nucl Chem 321, 97–108 (2019). https://doi.org/10.1007/s10967-019-06570-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-019-06570-1