Abstract
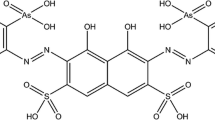
The complexation of thorium with arsenazo III in perchloric acid was quantitatively investigated with ultraviolet–visible (UV–Vis) absorption spectroscopy. The UV–Vis absorption of both 1:1 and 1:2 (thorium to arsenazo III) complexes in perchloric acid were found to be highly enhanced than the previously reported absorption of the complexes in hydrochloric acid. The stability constants of thorium-arsenazo III complexes were determined via computational analysis, and the SIT (specific ion interaction theory) was employed to evaluate the dependence on ionic strength. This work contributes to a better understanding of the speciation and spectroscopic properties of thorium-arsenazo III complexes at high ionic strength.








Similar content being viewed by others
References
Kim JI (2006) Significance of actinide chemistry for the long-term safety of waste disposal. Nucl Eng Technol 38:459–482
Altmaier M, Gaona X, Fanghänel T (2013) Recent advances in aqueous actinide chemistry and thermodynamics. Chem Rev 113:901–943
Grenthe I, Stumm W, Laaksuharju M, Nilsson AC, Wikberg P (1992) Redox potentials and redox reactions in deep groundwater systems. Chem Geol 98:131–150
Ryu JH, Koh YK, Park SW, Kim GY, Choi JW (2012) Geochemical characterization of deep groundwater in KURT using geochemical modeling. J Environ Eng 138:351–359
Petrow HG, Strehlow CD (1967) Spectrophotometric determination of thorium in bone ash using arsenazo III. Anal Chem 39:265–267
Kiriyama T, Kuroda R (1974) Ion-exchange separation and spectrophotometric determination of zirconium, thorium and uranium in silicate rocks with arsenazo III. Anal Chim Acta 71:375–381
Fukuma HT, Fernandes EAN, Nascimento MRL, Quinelato AL (2001) Separation and spectrophotometric determination of thorium contained in uranium concentrate. J Radioanal Nucl Chem 248:549–553
Shvoeva OP, Dedkova VP, Savvin SB (2007) Sorption and complexation of uranium(VI) and thorium(IV) with reagents arsenazo III and arsenazo M on fibrous filled sorbents. J Anal Chem 62:935–939
Liang Y, He Y (2016) Arsenazo III functionalized gold nanoparticles for photometric determination of uranyl ion. Microchim Acta 183:407–413
Savvin SB (1961) Analytical use of arsenazo III: determination of thorium, zirconium, uranium and rare earth elements. Talanta 8:673–685
Savvin SB (1964) Analytical applications of arsenazo III–III: the mechanism of complex formation between arsenazo III and certain elements. Talanta 11:7–19
Rohwer H, Rheeder N, Hosten E (1997) Interactions of uranium and thorium with arsenazo III in an aqueous medium. Anal Chim Acta 341:263–268
Choi S, Lee JY, Yun JI (2017) Stability constants and spectroscopic properties of thorium(IV)–arsenazo III complexes in aqueous hydrochloric medium. J Solution Chem 46:1272–1283
Leggett DJ (1985) Computational methods for the determination of formation constants. Plenum Press, New York
Wanner H, Östhols E (1999) Guidelines for the assignment of uncertainties. OECD Nuclear Energy Agency, Issy-les-Moulineaux
Grenthe I, Mompean F, Spahiu K, Wanner H (2013) Guidelines for the extrapolation to zero ionic strength. OECD Nuclear Energy Agency Data Bank, Issy-les-Moulineaux
Wanner H, Östhols E (2015) Standards and conventions for TDB publications. OECD Nuclear Energy Agency, Issy-les-Moulineaux
Renny JS, Tomasevich LL, Tallmadge EH, Collum DB (2013) Method of continuous variations: applications of job plots to the study of molecular associations in organometallic chemistry. Angew Chem Int Ed 52:11998–12013
Jackson GE, Seymour LF (1995) Formation constants at high ionic strength-II. The ionic strength correction of formation constants using a simplified pitzer equation. Talanta 42:9–16
Buděšínský B (1969) Acidity of several chromotropic acid azo derivatives. Talanta 16:1277–1288
Kim HT, Frederick WJ (1988) Evaluation of pitzer ion interaction parameters of aqueous electrolytes at 25 °C. 1. Single salt parameters. J Chem Eng Data 33:177–184
Das B (2004) Pitzer ion interaction parameters of single aqueous electrolytes at 25 °C. J Solution Chem 33:33–45
Palei PN, Udaltsova NI, Nemodruk AA (1967) Acid dissociation constants of arsenazo III. Zh Analit Khim 22:1797–1804 (in Russian)
Němcová I, Metal B (1986) Dissociation constants of arsenazo III. Talanta 33:841–842
Kufelnicki A, Lis S, Meinrath G (2005) Application of cause-and-effect analysis to potentiometric titration. Anal Bioanal Chem 382:1652–1661
Khan MH, Ali A, Khan NN (2001) Spectrophotometric determination of thorium with disodium salt of arsenazo-III in perchloric acid. J Radioanal Nucl Chem 250:353–357
Yamamoto T, Muto H, Kihara S, Motojima K (1971) Spectrophotometric determination of microamounts of plutonium in the presence of uranium. Anal Chim Acta 56:191–196
Rand M, Fuger J, Grenthe I, Neck V, Rai D (2008) Chemical thermodynamics of thorium. OECD Nuclear Energy Agency Data Bank, OECD Publications, Paris
Acknowledgements
This work was supported by the BK21 PLUS program, and a grant from the Nuclear R&D Program of the National Research Foundation of Korea funded by the Ministry of Science and ICT (Grant Nos. 2016M2B2B1945252 and 2017M2A8A5014801).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Choi, S., Yun, JI. Stability constants and spectroscopic properties of thorium(IV)-arsenazo III complexes in perchloric acid. J Radioanal Nucl Chem 319, 401–407 (2019). https://doi.org/10.1007/s10967-018-6342-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-018-6342-y