Abstract
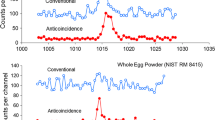
It is rather difficult to measure low levels of arsenic by instrumental neutron activation analysis (INAA) in biological materials containing high levels of bromine and antimony in particular, as well as sodium and potassium. The 559.1-keV photopeak of 76As is interfered with by the 554.3- and 564.1-keV photopeaks of 82Br and 122Sb, respectively. The use of INAA in conjunction with anticoincidence spectrometry (INAA-AC) was found to reduce the background under the 559.1-keV photopeak by factors of 4–16 for the biological reference materials analyzed and to decrease the detection limit to 0.35 µg kg−1 making the measurement of nanogram amounts arsenic in them possible.







Similar content being viewed by others
References
Shi Y, Acharya R, Chatt A (2004) J Radioanal Nucl Chem 262:277–286
Menendez Sanchez W, Zwicker B, Chatt A (2009) J Radioanal Nucl Chem 282:133–138. https://doi.org/10.1007/s10967-009-0224-2
Zwicker R, Zwicker BM, Laoharojanaphand S, Chatt A (2011) J Radioanal Nucl Chem 287:211–216. https://doi.org/10.1007/s10967-010-0670-x
Shi Y, Chatt A (2014) J Radioanal Nucl Chem 299:867–877. https://doi.org/10.1007/s10967-013-2694-5
Shi Y, Chatt A (2018) J Radioanal Nucl Chem 318:785–795. https://doi.org/10.1007/s10967-018-6164-y
Shi, Y (2000), PhD Thesis, Speciation of arsenic by chemical separations and neutron activation analysis, Department of Chemistry, Dalhousie University, Halifax, NS, Canada
Menendez Sanchez W (2010) PhD Thesis, Simultaneous speciation of arsenic, antimony and selenium in water by solid phase extraction and neutron activation, Department of Chemistry, Dalhousie University, Halifax, NS, Canada
Cubadda F, Jackson BP, Cottingham KL, Van Horne YO, Kurzius-Spencer M (2017) Sci Total Environ 579:1228–1239. https://doi.org/10.1016/j.scitotenv.2016.11.108
Han T, Ji H, Li H, Cui H, Song T, Duan X, Zhu Q, Cai F, Zhang L (2017) J Ocean Univ China (Ocean Coast Sea Res) 16:455–460. https://doi.org/10.1007/s11802-017-3070-9
Rabb SA, Le My D, Yu Lee L (2018) Microchem J 143:133–139. https://doi.org/10.1016/j.microc.2018.07.022
Zmozinski AV, Llorente-Mirandes T, López-Sánchez JF, da Silva MM (2015) Food Chem 173:1073–1082. https://doi.org/10.1016/j.foodchem.2014.10.102
Qiua Z, Lva Z, Wanga K, Lana Y, Yanga X, Rensingc C, Fub F, Yanga G (2018) J Food Compos Anal 72:132–140. https://doi.org/10.1016/j.jfca.2018.07.002
Dos Santosa QO, Silva JMM, Lemos VA, Ferreira SLC, de Andradea JB (2018) Microchem J 143:175–180. https://doi.org/10.1016/j.microc.2018.08.004
Wolle MM, Conklin SD (2018) Anal Bioanal Chem 410:5675–5687. https://doi.org/10.1007/s00216-018-0906-0
Wolle MM, Conklin SD (2018) Anal Bioanal Chem 410:5689–5702. https://doi.org/10.1007/s00216-018-0910-4
Greenberg RR, Bode P, De Nadai Fernandes EA (2011) Spectrochim Acta B 66:193–241. https://doi.org/10.1016/j.sab.2010.12.011
Greenberg RR (1987) J Radioanal Nucl Chem 113:233–247
Becker DA (1993) Unique quality assurance aspects of INAA for reference material homogeneity and certification. Fresenius J Anal Chem 345:298–301
Lindstrom RM (2018) J Radioanal Nucl Chem. https://doi.org/10.1007/s10967-018-6192-7
Neto MJL, Nascimento EdeS, Maihara VA, Silva PSC, Landgraf M (2014) J Radioanal Nucl Chem 301:573–579. https://doi.org/10.1007/s10967-014-3167-1
Acharya R, Swain KK, Shinde AD, Bhamra NS, Chakrabarty K, Karhadkar CG, Singh T, Rana YS, Pujari PK, Shukla DK, Reddy AVR (2014) J Radioanal Nucl Chem 302:1525–1530. https://doi.org/10.1007/s10967-014-3625-9
Patra AC, Mohapatra S, Kumar AV, Ravi PM, Tripathi RM (2015) J Radioanal Nucl Chem 303:315–323. https://doi.org/10.1007/s10967-014-3419-0
Kucera J, Cabalka M, Ferencei J, Kubesova M, Strunga V (2016) J Radioanal Nucl Chem 309:1341–1348. https://doi.org/10.1007/s10967-016-4739-z
Lange CN, Figueiredo AMG, Enzweiler J, Monteiro LR (2018) J Radioanal Nucl Chem 316:819–830. https://doi.org/10.1007/s10967-018-5729-0
Kato LS, Fernandes EADN, Bacchi MA, Sarries GA (2018) J Radioanal Nucl Chem 318:745–751. https://doi.org/10.1007/s10967-018-6122-8
Bedregal PS, Ubillús MS, Mendoza PA (2018) J Radioanal Nucl Chem 315:309–314. https://doi.org/10.1007/s10967-017-5684-1
Maroti B, Revay Z, Szentmiklosi L, Kleszcz K, Parkanyi D, Belgya T (2018) J Radioanal Nucl Chem 317:1151–1163. https://doi.org/10.1007/s10967-018-5990-2
Rahman M, Islam MA, Khan RA (2018) J Radioanal Nucl Chem. https://doi.org/10.1007/s10967-018-6222-5
Kamenik J, Simoes FRP, Costa PMFJ, Kucera J, Havranek V (2018) J Radioanal Nucl Chem. https://doi.org/10.1007/s10967-018-6200-y
Ryan DE, Stuart DC, Chattopadhyay A (1978) Anal Chim Acta 100:87–93
Elson CM, Milley JE, Chatt A (1983) Anal Chim Acta 142:269–275
Elson CM, Ackman RG, Chatt A (1983) J Am Oil Chem Soc 60:829–832
Holzbecher J, Chatt A, Ryan DE (1985) Can J Spectros 30:67–72
Chatt A, Dang HS, Fong BB, Jayawickreme CK, McDowell LS, Pegg DL (1988) J Radioanal Nucl Chem 124:65–77
Chatt A, Jayawickreme CK (1989) Trans Am Nucl Soc 60:21–22
Laoharojanaphand S, Busamongkol A, Permnamtip V, Judprasong K, Chatt A (2012) J Radioanal Nucl Chem 294:323–327. https://doi.org/10.1007/s10967-012-1884-x
Landsberger S, Swift G, Neuhoff J (1990) Biol Trace Elem Res 26–27:27–32
Lin X, Lierse Ch, Wahl W (1997) J Radioanal Nucl Chem 215:169–178
Zhang W, Chatt A (1997) Trans Am Nucl Soc 77:11–12
Zhang W (1997) PhD thesis, Studies on anticoincidence gamma-ray spectrometry in neutron activation analysis, Department of Chemistry, Dalhousie University, Halifax, NS, Canada
Ryan DE, Holzbecher J, Chattopadhyay A (1987) Anal Chim Acta 200:89–100
Milley JE, Chatt A (1987) J Radioanal Nucl Chem 110:345–363
Jayawickreme CK, Chatt A (1987) J Radioanal Nucl Chem 110:583–593
Byrne AR (1987) Fresenius J Anal Chem 326:733–735
Mok WM, Wai CM (1988) Talanta 35:183–186
Beazley PI, Rao RR, Chat A (1994) J Radioanal Nucl Chem 179:267–276
Nyarko BJB, Akaho EHK, Fletcher JJ, Chatt A (2008) Appl Rad Isot 66:1067–1072
Acharya R, Chatt A (2009) J Radioanal Nucl Chem 282:991–996
Zhang W, Chatt A (2009) J Radioanal Nucl Chem 282:139–143
Fukushima M, Chatt A (2012) J Radioanal Nucl Chem 294:471–478
Isaac-Olive K, Chatt A (2012) J Radioanal Nucl Chem 294:479–486
Fukushima M, Chatt A (2013) J Radioanal Nucl Chem 296:563–571
Zhang W, Chatt A (2013) J Radioanal Nucl Chem 296:495–501. https://doi.org/10.1007/s10967-012-2064-8
Zhang W, Chatt A (2014) J Radioanal Nucl Chem 299:1777–1785. https://doi.org/10.1007/s10967-013-2854-7
Zhang W, Chatt A (2014) J Radioanal Nucl Chem 302:1201–1211. https://doi.org/10.1007/s10967-014-3540-0
Isaac-Olive K, Chatt A (2014) J Radioanal Nucl Chem 302:1213–1224. https://doi.org/10.1007/s10967-014-3520-4
Isaac-Olive K, Kyaw TT, Chatt A (2018) J Radioanal Nucl Chem 318:247–257. https://doi.org/10.1007/s10967-018-6086-8
Serfor-Armah Y, Carboo D, Akuamoah RK, Chatt A (2018) J Radioanal Nucl Chem. https://doi.org/10.1007/s10967-018-6194-5
Drescher A, Yoho M, Landsberger S (2018) J Radioanal Nucl Chem 318:527–532. https://doi.org/10.1007/s10967-018-6033-8
Ehmann WD, McKown DM (1969) Anal Lett 2:49–60
Tomlin BE, Zeisler R, Lindstrom RM (2008) Nucl Instrum Meth Phys Res A 589:243–249
Di Piero A, Bacchi MA, Fernandes EAN (2008) J Radioanal Nucl Chem 278:761–765. https://doi.org/10.1007/s10967-008-1607-5
Zeisler R, Cho H, Ribeiro IS, Shetty MG, Turkoglu D (2017) J Radioanal Nucl Chem 314:513–519. https://doi.org/10.1007/s10967-017-5342-7
Zhang W, Chatt A (2018) J Radioanal Nucl Chem 318:445–455. https://doi.org/10.1007/s10967-018-6031-x
Acknowledgements
The authors would like to acknowledge the assistance of Dr. J. Holzbecher and Blaine Zwicker of the DUSR facility for irradiations. The financial assistance from the Natural Sciences and Engineering Research Council (NSERC) Canada in the form of Discovery and Infrastructure Grants (AC) and the Dalhousie University Faculty of Graduate Studies for a scholarship (WZ) are also acknowledged. This paper was presented at RadChem 2018 held during 2018 May 13–18 in Mariánské Lázně, Czech Republic.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, W., Chatt, A. INAA with anticoincidence counting significantly reduces interferences from the 554.3-keV photopeak of 82Br to allow reliable measurements of nanogram levels of arsenic in solid biological materials via the 559.1-keV photopeak of 76As. J Radioanal Nucl Chem 318, 1671–1680 (2018). https://doi.org/10.1007/s10967-018-6280-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-018-6280-8