Abstract
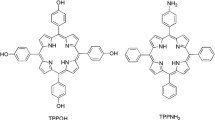
Porphyrin derivatives are usually used for photodynamic therapy (PDT) and labeled with raidonuclide for radiopharmaceuticals. In this work, 5,10,15,20-tetrakis (4-hydroxyphenyl) porphyrin (TPPOH) and 5,10,15,20-tetrakis (4-aminophenyl) porphyrin (TPPNH2) were labeled by 131I. 131I-TPPOH and 131I-TPPNH2 were cultured with SMMC-7721 and HL-7702 cells for affinity test. The results show high uptake ratios of SMMC-7721 than HL-7702. 131I-TPPOH and 131I-TPPNH2 were performed radionuclide therapy (RNT) and PDT via tumor in situ injection. At 14 days post injection, the volumes of tumors injected 131I-TPPOH and 131I-TPPNH2 diminished 64 ± 11 and 54 ± 10%, respectively. The therapeutic effect is better than either single PDT or single RNT. The combination therapy of PDT and RNT could be a more effectual method for cancer therapy.





Similar content being viewed by others
References
Firestone RA (1994) low-density-lipoprotein as a vehicle for targeting antitumor compounds to cancer-cells. Bioconjug Chem 5:105–113
Roger A, Clive K, Nicola B, Malcolm R (2001) The history of photodetection and photodynamic therapy. Photochem Photobiol 74:656–669
Chitroda PK, Katti G, Attar NM, Singh AK (2015) Photodynamic therapy-a review. Indian J Mednodent Allied Sci 3:180–187
Osterloh J, Vicente MGH (2002) Mechanisms of porphyrinoid localization in tumors. J Porphyrins Phthalocyanines 6:305–324
Hargus JA, Fronczek FR, Graca M, Vicente H, Smith KM (2007) Mono-(l)-aspartylchlorin-e(6). Photochem Photobiol 83:1006–1015
Juzeniene A (2009) Chlorin e6-based photosensitizers for photodynamic therapy and photodiagnosis. Photodiagn Photodyn 6:94–96
Jia ZY, Deng HF, Pu MF (2007) Synthesis and preliminary biological studies of the novel conjugate 188Re-labeled meso-tetrakis (4-sulfophenyl) porphyrin in mice. Nucl Med Biol 34:643–649
Jia ZY, Deng HF, Pu MF, Luo SZ (2008) 188Rh labeled meso-tetrakis[3,4-bis (carboxymethyleneoxy) phenyl] porphyrin for targeted radiotherapy: preliminary biological evaluation in mice. Eur J Nucl Med Mol Imaging 35:734–742
Liu YP, Shen B, Liu F, Zhang B, Chu TW, Bai J, Bao SL (2012) Synthesis, radiolabeling, biodistribution and fluorescent imaging of histidine-coupled hematoporphyrin. Nucl Med Biol 39:579–585
Santos PM, Laranjo M, Serra AC, Abrantes AM, Pineiro M, Casalta-Lopes J, Trindade D, Maia J, Rocha-Gonsalves A, Botelho MF (2014) Evaluation of a 99mTc-labelled mesobisphenylporphyrinas a tumor image agent. J Label Compd Radiopharm 57:141–147
Mukai H, Wada Y, Watanabe Y (2013) The synthesis of 64Cu-chelated porphyrin photosensitizers and their tumor-targeting peptide conjugates for the evaluation of target cell uptake and PET image-based pharmacokinetics of targeted photodynamic therapy agents. Ann Nucl Med 27:625–639
Fazaeli Y, Jalilian AR, Amini MM, Aboudzadeh M, Feizi S, Rahiminezhad A, Yousefi K (2013) Preparation, nano purification, quality control and labeling optimization of [64Cu]-5,10,15,20-tetrakis (penta fluoro phenyl) porphyrin complex as a possible imaging agent. J Radioanal Nucl Chem 295:255–263
Chen YH, Zheng X, Dobhal MP, Gryshuk A, Morgan J, Dougherty TJ, Oseroff A, Pandey RK (2005) Methyl pyropheophorbide-a analogues: potential fluorescent probes for the peripheral-type benzodiazepine receptor effect of central metal in photosensitizing efficacy. J Med Chem 48:3692–3695
Vahidfar N, Jalilian AR, Fazaeli Y, Bahrami-Samani A, Beiki D, Khalaj A (2014) Radiosynthesis and biological evaluation of 166Ho labeled methoxylated porphyrins as possible therapeutic agents. J Radioanal Nucl Chem 301:269–276
Dasa T, Chakrabortya S, Sarma HD, Banerjeea S, Venakatesh M (2010) A novel 177Lu-labeled porphyrin for possible use in targeted tumor therapy. Nucl Med Biol 37:655–663
Yang YQ, Song H, Song HT, Zhao WW, Pu MF (2011) Preparation of 99mTc(CO)3-TPPS4 and its biological behavior evaluation. J Porphyrins Phthalocyanines 15:271–276
Gomer CJ, Dougherty TJ (1979) Determination of [3H]-and [14C] hematoporphyrin derivative distribution in malignant and normal tissue. Cancer Res 39:146–151
Pandey SK, Gryshuk AL, Sajjad M, Zheng X, Chen YH, Abouzeid MM, Morgan J, Charamisinau I, Nabi HA, Oseroff A, Pandey RK (2005) Multimodality agents for tumor imaging (PET, Fluorescence) and photodynamic therapy. a possible, “see and treat” approach. J Med Chem 48:6286–6295
Srivatsan A, Wang YF, Joshi P, Sajjad M, Chen YH, Liu C, Thankppan K, Missert JR, Tracy E, Morgan J, Rigual N, Baumann H, Pandey RK (2011) In vitro cellular uptake and dimerization of signal transducer and activator of transcription-3 (STAT3) identify the photosensitizing and imaging-potential of isomeric photosensitizers derived from chlorophyll-a and bacteriochlorophyll-a. J Med Chem 54:6859–6873
Sun ZZ, Wei HY, Wang GQ, Zhou ZJ, He JH, Wang J, Luo SZ (2014) Preparation and the biodistribution study of [131I]-5,10,15,20-tetrakis(4-hydroxyphenyl)porphyrin and 5-(4-Aminophenyl)-10,15,20-triphenylporphyrin. J Radioanal Nucl Chem 302:123–130
Nakajimaa S, Moriyamab T, Hayashi H, Sakata I, Nakae Y, Takemura T (2000) Hemopexin as a carrier protein of tumor-localizing Ga-metalloporphyrin-ATN-2. Cancer letter 149:221–226
Acknowledgements
This work was financially supported by the National Nature Science Foundation of China (NSFC-21471138, NSFC-21401176). We are also grateful to Nuclear Medicine Department of West China Hospital for the performance of SPECT.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Song, H., Wang, G., Wang, J. et al. 131I-labeled 5,10,15,20-tetrakis(4-hydroxyphenyl)porphyrin and 5,10,15,20-tetrakis(4-aminophenyl)porphyrin for combined photodynamic and radionuclide therapy. J Radioanal Nucl Chem 316, 363–368 (2018). https://doi.org/10.1007/s10967-018-5735-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-018-5735-2