Abstract
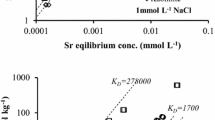
The migration and sorption of Sr in clay-sand mixture were investigated by batch experiment, column experiments and numerical simulation. The results showed that as the clay content in clay-sand mixture increased, the effective porosity, absorption capacity and retardation factor of the mixture for Sr increased, but the dispersion coefficient and migration velocity decreased. The migration of Sr was influenced strongly when clay content was in range of 0–25 %, but influenced weakly when clay content was more than 25 %. The experimental data was consistent with the calculated results by CXTFIT program.







Similar content being viewed by others
References
Jacquier P, Hainos D, Robinet JC, Herbette M, Grenut B, Bouchet A, Ferry C (2013) The influence of mineral variability of callovo–oxfordian clay rocks on radionuclide transfer properties. Appl Clay Sci 83:129–136
Kosakowski G, Berne U (2013) The evolution of clay rock/cement interfaces in a cementitious repository for low- and intermediate level radioactive waste. Phys Chem Earth 64:65–86
Landais P, Aranyossy JF (2011) Clays in natural and engineered barriers for radioactive waste confinement. Phys Chem Earth 36:14–37
Mallants D, Marivoet J, Sillen X (2001) Performance assessment of the disposal of vitrified high-level waste in a clay layer. Nucl Mater 298:125–135
Albrecht BA, Benson CH (2001) Effect of desiccation on compacted natural clay. J Geotech Geoenviron Eng 127:67–75
Rayhani MHT, Yanful EK, Fakher A (2007) Desiccation-induced cracking and its effect on the hydraulic conductivity of clayey soils from Iran. Can Geotech J 44:276–283
Huang WR, Guo ZP (2000) Experimental research on Pr eventing desiccation cracking of compacted clay liner in municipal solid waste landfill. J Hohai Univ 28:19–22
Rothfuchs T, Jockwer N, Zhang CL (2007) Self-sealing barriers of clay/mineral mixtures—the SB project at the Mont Terri Rock Laboratory. Phys Chem Earth 32:108–111
Saba S, Barnichon JD, Cui YJ (2014) Microstructure and anisotropic swelling behaviour of compacted bentonite/sand mixture. J Rock Mech Geotech Eng 6:126–132
Sun DA, Zhang JY, Zhang JR (2013) Swelling characteristics of GMZ bentonite and its mixtures with sand. Appl Clay Sci 83:224–230
Wang Q, Tang AM, Cui YJ (2013) The effects of technological voids on the hydro-mechanical behaviour of compacted bentonite–sand mixture. Soils Found 53:232–245
Chirkst DE, Litvinova TE, Cheremisina OV, Ivanov MV, Mironnenkova NA (2003) Isotherm of strontium sorption on clay. Russ J Appl Chem 76:755–758
Galambos M, Kufcakova J, Rajec P (2009) Sorption of strontium on Slovak bentonites. J Radioanal Nucl Chem 281:347–357
Khan SA (2003) Sorption of the long-lived radionuclides cesium-134, strontium-85 and cobalt-60 on bentonite. J Radioanal Nucl Chem 258:3–6
Missana T, Gutierrez MG, Alonso U (2008) Adsorption of bivalent ions (Ca(II), Sr(II) and Co(II)) onto FEBEX bentonite. Phys Chem Earth. 33:156–162
Syed HS, Syed A (1995) Competitive adsorption of 90Sr on soil sediments, pure clay phases and feldspar minerals. Appl Radiat Isot 46:287–292
Vanessa ED, Jarbas RR, Solange C, Gilberto A, Marco TG (2014) Montmorillonite and vermiculite as solid phases for the preconcentration of trace elements in natural waters: adsorption and desorption studies of As, Ba, Cu, Cd Co, Cr, Mn, Ni, Pb, Sr, V, and Zn. Appl Clay Sci 84:1–8
Zuo R, Teng YG, Wang JS (2009) Sorption and retardation of strontium in fine-particle media from a VLLW disposal site. J Radioanal Nucl Chem 279:893–899
Zhang ML, Ren A, Shao X, Wang X (2005) Effect of fluvic acid and ionic strength on the sorption of radiostrontium on Chinese calcareous soil and its solid components. J Radioanal Nucl Chem 268:33–36
Dewiere L, Bugai D, Grenier C, Kashparov V, Ahamdach N (2004) 90Sr migration to the geo-sphere from a waste burial in the chernobyl exclusion zone. J Environ Radioact 74:139–150
Takriti S, Kheitou M, Ali AF (1999) Migration study of radioisotopes incorporated in cement. Appl Radiat Isot 50:401–406
Rumyin VG, Mironova AV, Pankina EB, Chernomorova NV, Mysik SG (2004) A study of diffusion and sorption properties of Cambrian clays using radioactive tracers (36Cl and 90Sr). Radiochemistry 46:362–367
Shih CT, Shoung O, Chun NH (2001) Sorption and diffusion behavior of Cs and Sr on Jih-Hsing bentonite. Appl Radiat Isotop. 54:209–215
Van Loon LR, Baeyens B, Bradbury MH (2005) Diffusion and retention of sodium and strontium in opalinus clay: comparison of sorption data from diffusion and batch sorption measurements and geochemical calculations. Appl Geochem 20:2351–2363
Chia CC, Kim FH (1999) X-ray absorption spectroscopy investigation of aqueous Co(II) and Sr(II) sorption at clay-water interfaces. Geochim Cosmochim Acta. 63:3205–3215
Li YF, Tian SH, Qian TW (2011) Transport and retention of strontium in surface-modified quartz sand with different wettability. J Radioanal Nucl Chem 289:337–343
Zhang Y, Baeumer B, Benson DA (2006) Relationship between flux and resident concentrations for anomalous dispersion. Geophys Res Lett 33:L18407
Parker JC, Genuchten V, Baeumer B (1984) Flux averaged and volume averaged concentrations in continuum approaches to solute transport. Water Resour Res 20:866–872
Relyea JF, Serne RJ, Rai D (1980) Methods for Determining Radionuclide Retardation Factors. Appendix C, standard method used at pacific northwest national laboratory for measuring batch K d values. PNNL Status Report, Richland, p. 13
Missana T, Geckeis H (2006) Supporting laboratory experiments with radionuclides and bentonite colloids. Grimsel Test Site Investigation Phase V, The CRR Final Project Report Series II
Acknowledgments
The research was supported by the Program for Changjiang Scholars and Innovative Research Team in University (IRT-13026). The authors would also like to thank the financial support provided by the Open Research Fund Program of Key Laboratory of Advanced Reactor Engineering and Safety, Ministry of Education of China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, J., Wang, J. & Jiang, Y. Migration and sorption of strontium in clay-sand mixtures. J Radioanal Nucl Chem 308, 439–445 (2016). https://doi.org/10.1007/s10967-015-4477-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-015-4477-7