Abstract
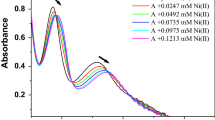
A novel method was designed for the synthesis of polyphenol capped amorphous iron oxide nanoparticles (ION) using an agro waste (peanut skin). The synthesized nanoparticles were characterized by absorption spectroscopy, TEM, SEM, IR, Raman and XRD. An increased rate of formation of the nanoparticles is observed upon low dose gamma irradiation. ION shows specific spectral features with perchlorate anion even in presence of other anions selected for the anion sensing study. The nanoparticles formed in presence of irradiation was found even more sensitive to lower concentration of perchlorate owing to its finely dispersed nature and increased surface area.








Similar content being viewed by others
References
Vella AJ, Chircop C, Micallef T, Pace C (2015) Perchlorate in dust fall and indoor dust in Malta: an effect of fireworks. Sci Total Environ 521–522:46–51
Susarla S, Collette TW, Garrison AW, Wolfe NL, McCutcheon SC (1999) Perchlorate identification in fertilizers. Environ Sci Technol 33:3469–3472
Steinmaus C, Miller MD, Cushing L, Blount BC, Smith AH (2013) Combined effects of perchlorate, thiocyanate, and iodine on thyroid function in the National Health and Nutrition Examination Survey 2007–08. Environ Res 123:17–24
Wu F, Chen H, Zhou X, Zhang R, Ding M, Liu Q, Peng KL (2013) Pulmonary fibrosis effect of ammonium perchlorate exposure in rabbit. Arch Environ Occup Health 68:161–165
Mosier-Boss PA, Putnam MD (2014) Detection of perchlorate using Ag/DMAH+ SERS-active capture matrices. Spectrochim Acta Part A 133:156–164
Nuntawong N, Eiamchai P, Limwichean S, Wong-ek B, Horprathum M, Patthanasettakul V, Leelapojanaporn A, Nakngoenthong S, Chindaudom P (2013) Trace detection of perchlorate in industrial-grade emulsion explosive with portable surface-enhanced Raman spectroscopy. Forensic Sci Int 233:174–178
Wagner HP, Pepich BV, Pohl C, Srinivasan K, Lin R, DeBorba B, Munch DJ (2007) Selective method for the analysis of perchlorate in drinking waters at nanogram per liter levels, using two-dimensional ion chromatography with suppressed conductivity detection. J Chromatogr A 1155:15–21
Braik M, Dridi C, Ali A, Abbas MN, Ben Ali M, Errachid A (2015) Development of a perchlorate sensor based on Co-phthalocyanine derivative by impedance spectroscopy measurements. Org Electr 16:77–86
Kharisov BI, Dias HVR, Kharissova OV, Jime´nez-Pe´rez VM, Pe´rez BO, Mun˜ozFloresa B (2012) Iron-containing nanomaterials: synthesis, properties, and environmental applications. RSC Adv 2:9325–9358
Hoanga VV, Ganguli D (2012) Amorphous nanoparticles—experiments and computer simulations. Phys Rep 518:81–140
Ramesh R, Ashok K, Bhalero GM, Ponnusamy S, Muthamizhchelvan C (2010) Synthesis and properties of α-Fe2O3 nanorods. Cryst Res Technol 45:965–968
Sugimoto T, Matijevic E (1980) Formation of uniform spherical magnetite particles by crystallization from ferrous hydroxide gels. J Colloid Interface Sci 74:227–243
Tartaj P, Md. Morales P, Veintemillasr SV, Gonzalez TC, Serna JC (2003) The preparation of magnetic nanoparticles for applications in biomedicine. J Phys D 36:R182–R197
Suslick SK, Choe BS, Cichowlas AA, Grinstaff WM (1991) Applications of sonochemistry to material synthesis. Nature 353:414–416
Srivastava DN, Perkas N, Gedanken A, Felner I (2002) Sonochemical synthesis of mesoporous iron oxide and accounts of its magnetic and catalytic properties. J Phys Chem B 106:1878–1883
Mittal AK, Chisti Y, Banerjee UC (2013) Synthesis of metallic nanoparticles using plant extracts. Biotechnol Adv 31:346–356
Biswal J, Misra N, Bordeand LC, Sabharwal S (2013) Synthesis of silver nanoparticles in methacrylic acid solution by gamma radiolysis and their application for estimation of dopamine at low concentrations. Radiat Phys Chem 83:67–73
Yakabuskie PA, Joseph JM, Keech P, Botton GA, Guzonas D, Wren JC (2011) Iron oxyhydroxide colloid formation by gamma-radiolysis. Phys Chem Chem Phys 13:7198–7206
dL. Francisco ML, Resurreccion AVA (2012) Development of a reversed-phase high performance liquid chromatography (RP-HPLC) procedure for the simultaneous determination of phenolic compounds in peanut skin extracts. LWT Food Sci Technol 47:189–198
Lester GE, Lewers KS, Medina MB, Saftner RA (2012) Comparative analysis of strawberry total phenolics via fast blue Bb vs. Folin–Ciocalteu: assay interference by ascorbic acid. J Food Compos Anal 27:102–107
Mahajan S, Mahajan RK (2012) Interactions of phenothiazine drugs with bile salts: micellization and binding studies. J Colloid Interface Sci 387:194–204
de Camargo AC, Bismara MA, d’Arce R, Gallo CR, Shahidi F (2015) Gamma-irradiation induced changes in microbiological status, phenolic profile and antioxidant activity of peanut skin. J Funct Foods 12:129–143
Phu ND, Ngo DT, Hoang LH, Luong NH, Chau N, Hai NH (2011) Crystallization process and magnetic properties of amorphous iron oxide nanoparticles. J Phys D 44:345002–345008
Oh SJ, Cook DC, Townsend HE (1998) Characterization of iron oxides commonly formed as corrosion products on steel. Hyperfine Interact 112:59–65
Clarkson J, Tonge PJ, Taylor KL, Dunaway-Mariano D, Carey PR (1997) Raman study of the polarizing forces promoting catalysis in 4-chlorobenzoate-CoA dehalogenase. Biochemistry 36:10192–10199
Iqbal MZ, Ma X, Chen T, Zhang L, Ren W, Xiang L, Wu A (2015) Silica-coated super-paramagnetic iron oxide nanoparticles (SPIONPs): a new type contrast agent of T1 magnetic resonance imaging (MRI) J. Mater Chem B (in press)
Hoang VV, Ganguli D (2012) Amorphous nanoparticles—experiments and computer simulations. Phys Rep 518:81–140
Racuciu M, Creanga DE, Airinei A, Chicea D, Badescu V (2010) Synthesis and properties of magnetic nanoparticles coated with biocompatible compounds. Mater Sci Pol 28:609–616
Acknowledgments
We express sincere thanks to UGC-DAE, CSR/PROJECT/ACCT/2014/0057/0085 for necessary funding. We are thankful to Mr. Nayan Saha, Department of Chemical Technology, University of Calcutta, India for measuring the XRD. We also thank Ms. Urmila Goswami, and Mr. Pratyush Sengupta Centre for Research in Nanoscience and Nanotechnology, University of Calcutta, India, for obtaining TEM and SEM images.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ansari, Z., Sarkar, K., Saha, A. et al. Enhanced anion sensing by γ-irradiated polyphenol capped iron oxide nanoparticles. J Radioanal Nucl Chem 308, 517–525 (2016). https://doi.org/10.1007/s10967-015-4473-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-015-4473-y