Abstract
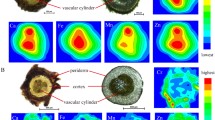
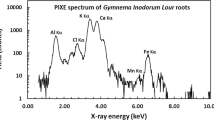
Quantitative estimation of inorganic elements of cotyledon-derived callus tissues of a medicinal legume, Abrus precatorius L. was determined using Proton induced X-ray emission technique. Nine trace elements namely Cu, Cr, Co, Zn, Mn, Fe, Se, Br and Ti in addition to two macro-elements K and Ca were identified, quantified and compared with each culture passage to monitor the variation in trace element accumulation. Experimental findings revealed that, 10-week-old calli was more efficient than others in accumulating inorganic elements. These results through light on the implication of in vitro callus cultures as a viable, alternative and proliferating renewable resource of medicinally useful elements for designing of drugs and also helping in natural germplasm conservation.




Similar content being viewed by others
References
Trejo-Tapia G, Balcazar-Aguilar JB, Martı´nez-Bonfil B, Salcedo-Morales G, Jaramillo-Flores M, Arenas-Ocampo L, Jime´ nez-Aparicio A (2008) Innov Food Sci Emerg Technol 9:32–36
Baldi A, Dixit VK (2008) Bioresour Technol 99:4609–4614
Dhakulkar S, Ganapathi TR, Bhargava S, Bapat VA (2005) Plant Sci 169:812–818
Wang B, Zhang G, Zhu L, Chen L, Zhang Y (2006) Biointerfaces 53:101–104
Namdeo AG (2007) Pharmacogn Rev 1:69–79
Ivey M, Elmen G (1989) Nutritional supplements, mineral and vitamin production v In: Berardi RR, Kroon LA, McDermott JH, Newton GD, Oszko MA, Popovich NG, Remington TL, Rollins CJ, Shimp LA, Tietze KJ (Eds.) Handbook of Nonprescription Drugs: An Interactive Approach to Self-Care 8th ed. American Pharmaceutical Association, The National Professional Society of Pharmacists, 2215 Constitution Avenue, N.W. Washington, DC 20037, pp 215
Tolonen M (1990) Vitamins and minerals in health and nutrition. Woodhead Publishing Ltd., Abington Hall, Abington, Cambridge
Chen KS, Tseng CL, Lin TH (1993) J Radioanal Nucl Chem 170:265–280
Birch NJ, Padgham C (1994) Handbook on metals in clinical and analytical chemistry. Marcel Dekker, New York
Obiajunwa EI, Adebajo AC, Omobuwajo OR (2002) J Radioanal Nucl Chem 252:473–476
Ekinci N, Ekinci R, Polat R, Budak G (2004) J Radioanal Nucl Chem 260:127–131
Kar A, Choudhury BK, Bandyopadhyay NG (1994) J Ethnopharmacol Pharmacol 64:179–184
Singh V, Garg AN (1997) Appl Radiat Isot 48:97–101
Garcia JS, de Magalh˜aes CS, Arruda MAZ (2006) Talanta 69:1–15
Fraga CG (2005) Mol Asp Med 26:235–244
Colak H, Soylak M, Turkoglu O (2005) Trace Elem Electrolyte 22:192–195
Divrikli U, Horzum N, Soylak M, Elci L (2006) Int J Food Sci Technol 41:712–716
Szoboszlai N, Polgari Z, Mihucz VG, Zaray G (2009) Anal Chim Acta 633:1–18
Majumdar S, Peralta-Videaa JR, Castillo-Michelc H, Hong J, Ricoa CM, Gardea-Torresdeya JL (2012) Anal Chim Acta 755:1–16
Yamashita CI, Saiki M, Sertie´ JAA (2006) J Radioanal Nucl Chem 270:181–186
Queralt I, Ovejero M, Carvalho ML, Marques AF, Llabres JM (2005) X-ray Spect 34(3):213–217
Sahu L, Ray DK, Chand PK (2014) J Radioanal Nucl Chem 300:345–354
NagaRaju GJ, Sarita P, Ramana Murty GA, Ravi Kumar M, Reddy BS, Charles MJ, Lakshminarayana S, Reddy TS, Reddy SB, Vijayan V (2006) Appl Radiat Isot 64:893–900
Nayak P, Behera PR, Thirunavoukkarasu M, Chand PK (2011) Appl Radiat Isot 69:567–573
Bhatia M, Siddiqui NA, Gupta S (2013) Indo Am J Pharm Res 3(4):3295–3315
Ray DK, Nayak PK, Panda SR, Rautray TR, Vijayan V, Christopher CC, Jena S (2006) Int J PIXE 16:47–54
Rautray TR, Vijayan V, Panigrahi S (2007) Nucl Instrum Methods Phys Res B 255:409–415
Kumar SSP, Venkateswarlu P, Rao VR, Rao GN (2013) Int Nano Lett. doi:10.1186/2228-5326-3-30
Campbell JL, Hopman TL, Maxwell JA, Nejedly Z (2000) Nucl Instrum Methods Phys Res B 170:193–204
Gomez KA, Gomez AA (1984) Statistical procedures for agricultural research, 2nd edn. Wiley, New York
Tıraşoğlu E, Cevik U, Ertuğral B, Apaydin G, Baltas H, Ertuğrul M (2005) J Quant Spectrosc Radiat Transf 94:181–187
Desai NS, Joseph D, Suprasanna P, Bapat VA (2006) Nucl Instrum Methods Phys Res B 252:299–302
de Oliveira LM, Paiva R, de Santana JRF, Pereira FD, Nogueira RC, Silva LC (2010) Cienc Agrotec Lavras 34:1439–1445
Behera PR, Nayak P, Barik DP, Rautray TR, Thirunavoukkarasu M, Chand PK (2010) Appl Radiat Isot 68:2229–2236
Valko M, Morris H, Cronin MTD (2005) Curr Med Chem 12:1161–1208
Andrews NC (1995) N Engl J Med 341:1986–1995
Nuviala RJ, Lapieza MG, Bernal E (1999) Int J Sport Nutr 9:295–309
Rajukar NS, Pardeshi BM (1997) Appl Radiat Isot 48(8):1059–1062
Baker D, Campbell RK (1992) Diabetes Educ 18(5):420–427
Swain SS, Ray DK, Chand PK (2012) J Radioanal Nucl Chem 293(2):443–453. doi:10.1007/s10967-012-1796-9
Rayman MP, Phil D (2000) The Lancet 356:177–264
Mahboobi H, Yucel M, Oktem HA (2002) J Plant Nutr 25:1829–1837
Suzuki K, Itali R, Suzuki K, Nakanishi H, Nishizawa NK, Yoshimura E, Mori S (1998) Plant Phsiol 116:725–732
Saric M, Vasic D, Vasiljevic LJ, Skoric D, Mezei S, Pajevic S (1997) Romanian Agric Res 7–8:37–42
Aloni R (2001) J Plant Growth Regul 20:22–34
Donato VMTS, Andrade AG, Souza ES, França JGE (2003) Pesquisa Agropecuária Brasileira 38:1373–1379
Barberaki M, Kintzios S (2002) Sci Hortic 95:133–150
Acknowledgments
The funding support by the University Grants Commission (UGC), New Delhi, India through a ‘UGC Research Award’ project is gratefully acknowledged. Authors acknowledge UGC for the receipt of the award of ‘Research Fellowship in Science for Meritorious Students’. The authors are also thankful to Dr. K.B Satapathy, Head of the Dept., P.G Dept. of Botany, Utkal University for providing all available facilities at the Department.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pani, D., Rath, S.K., Ray, D.K. et al. Proton induced X-ray emission-based analysis of trace element composition of cotyledon derived in vitro callus culture of Abrus precatorius L.: a multimedicinal wild legume. J Radioanal Nucl Chem 308, 113–122 (2016). https://doi.org/10.1007/s10967-015-4363-3
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-015-4363-3