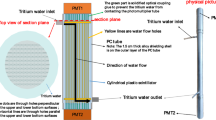
Abstract
Tritiated water is probably the most mobile manmade nuclide found in the environment. The low-energy beta decay produces few health concerns except at extremely high concentrations of tritium. However, because of the ubiquitous nature of tritiated water, when identified in the bionetwork, it can be viewed as a precursor of nuclides with greater health risks. For this reason alone it becomes desirable to determine tritium concentrations many orders of magnitude below that considered a health concern. A devise is described that eliminates the need for large water-filled chest freezers used as coolers. The enrichment cells can be employed individually or in series.


Similar content being viewed by others
References
Hill RL, Johnson JR (1993) Metabolism and dosimetry of tritium. Health Phys 65:628–647
National Council on Radiation Protection and Measurements (NCRP) (1979) Tritium in the environment, NCRP Report No. 62. National Council on Radiation Protection and Measurements, Bethesda, MD
Okada S, Momoshima N (1993) Overview of tritium: characteristics, sources, and problems. Health Phys 65:595–609
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stan Morton, J., Westmoreland, J.B. & Rhineheart, A. Self contained enrichment cell for tritium. J Radioanal Nucl Chem 282, 483–486 (2009). https://doi.org/10.1007/s10967-009-0264-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-009-0264-7