Abstract
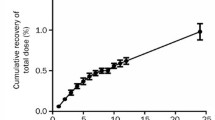
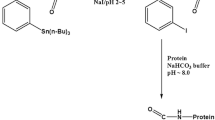
This study examined the tissues distribution and pharmacokinetics of rhTNT-IL2, a fusion protein, in rats and macaques after iv. injection. The rhTNT-IL2 was labeled with 125I. The 125I-rhTNT-IL2 was administered into rats and macaques at a dose of 250 μg·kg−1 and 125 μg·kg−1, respectively. Upon administration, rhTNT-IL2 declined in tri-exponentially with the half-lives of 1.78 h (T 1/2α), 25.06 h (T 1/2β), 114.19 h (T 1/2γ) for rats and 1.87 h (T 1/2α), 9.82 h (T 1/2β), 43.17 h (T 1/2γ) for macaques. The fraction of 125I-rhTNT-IL2 excreted in feces was low (about 1%), while the majority of 125I-rhTN-IL2 was excreted in urine. The tissue distributions showed that the liver, spleen and heart were major organs for deposition of the fusion protein in rats and macaques in 48-hour post-injection, and the fusion protein could not penetrate through blood brain barrier.
Similar content being viewed by others
References
T. A. Waldmann, S. Dubois, Y. Tagaya, Immunity, 14 (2001) 105.
T. A. Fehniger, M. A. Cooper, M. A. Caligiuri, Cytokine, 13 (2002) 169.
S. A. Rosenberg, Nature, 411 (2001) 380.
P. M. Andson, E. Katsanis, S. F. Sencer et al., J. Immunother., 12 (1992) 19.
M. E. Neville, K. W. Richau, L. T. Boni et al., Cytokine, 12 (2000) 1702.
K. Scheidhauer, I. Wolf, H.-J. Baumgartl et al., Eur. J. Nucl. Med. Mol. I, 29 (2002) 1276.
Z.-P. Yolanda, L. S. Kegeles, Ningning Guo et al., Nucl. Med. Biol., 29 (2002) 575.
Quanzhi Li, W. Hudson, Duo Wang et al., Cancer. Immunol. Immun., 47 (1998) 121.
E. S. Caldero N-Aranda, G. Riviere, V. Choumet et al., Toxicon, 37 (1999) 771.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zhang, Y., Xu, Y., Zhu, R. et al. Pharmacokinetics, tissue distribution and excretion of a recombinant fusion protein 125I-rhTNT-IL2. J Radioanal Nucl Chem 273, 3–8 (2007). https://doi.org/10.1007/s10967-007-0701-4
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-007-0701-4