Abstract
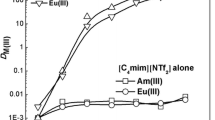
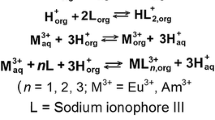
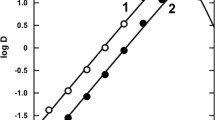
The formation constants of thiocyanate complexes of Eu(III) and Am(III) in trace concentrations were investigated in mixed solvent (CH3OH+H2O) solutions of different ionic strength. Furthermore, in paper electrophoresis, the moving velocities of the species of Eu(III) and Am(III) were investigated in 1.1M (H, Na)(SCN, ClO4) mixed solvent (CH3OH-H2O) solutions. The results showed that the difference between the velocities of Eu(III) and Am(III) is explained by the difference of the mean charges calculated by the formation constants of thiocyanate complexes of Eu(III) and Am(III) in the solution.
Similar content being viewed by others
References
M. Arisaka, T. Kimura, H. Suganuma, Z. Yoshida, Radiochim. Acta, 90 (2002) 193.
A. Ikeda, T. Suzuki, M. Aida, Y. Fujii, K. Itoh, T. Mitsugashira, M. Hara, M. Ozawa, J. Chromatogr., A1041 (2004) 195.
A. Ikeda, T. Suzuki, M. Aida, K. Ohtake, Y. Fujii, K. Itoh, M. Hara, T. Mitsugashira, J. Alloys Comp., 374 (2004) 245.
G. Suresh, M. S. Murali, J. N. Mathur, Radiochim. Acta, 91 (2003) 127.
G. R. Choppin, J. Alloys Comp., 223 (1995) 174.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mori, T., Suganuma, H., Yanaga, M. et al. The interaction between trivalent 4f and 5f-block elements and thiocyanate ion. J Radioanal Nucl Chem 272, 247–250 (2007). https://doi.org/10.1007/s10967-007-0509-2
Received:
Issue Date:
DOI: https://doi.org/10.1007/s10967-007-0509-2