Summary
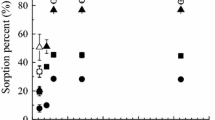
The sorption of Cs(I) and Sr(II) on bentonite and magnetite was experimentally studied and numerically simulated using surface complexation (SCM) and ion-exchange (IExM) models. The empirical system consisted of: (1) synthetic granitic water with a given ionic strength (0.1 or 0.01 NaNO3), (2) radionuclides studied (10-6M CsCl or SrCl2 . 6H2O spiked with 137Cs or 85Sr), and (3) bentonite pre-treated with the aim to remove carbonates, and magnetite as a representative of steel canister corrosion products. The parametric study covered, e.g., the influence of pH, bentonite to magnetite ratio and volume-liquid ratio on the values of selectivity coefficients and Kd.
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Filipská, H., Štamberg, K. Sorption of Cs(I) and Sr(II) on a mixture of bentonite and magnetite using SCM + IExM: A parametric study. J Radioanal Nucl Chem 270, 531–542 (2006). https://doi.org/10.1007/s10967-006-0459-0
Issue Date:
DOI: https://doi.org/10.1007/s10967-006-0459-0