Summary
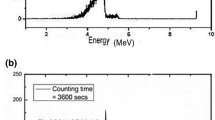
Water samples, spiked with 133Ba and 232U radiotracers, are scavenged for radium and uranium isotopes using hydrous manganese dioxide which is produced in-situ, by reacting manganese (+2) and permanganate ions at pH 8-9. The precipitate is solubilized with ascorbic and acetic acids and the resulting solution filtered through a glass fibre filter GF/F to remove particulate matter. The radium is co-precipitated with barium ions by the addition of a saturated Na2SO4 solution where a small amount of BaSO4 suspension is introduced to initiate crystallization. The micro precipitate containing the radium is collected on a 0.1 µm membrane filter and the filtrate saved for follow-up uranium analysis. The 226Ra on the filter is determined by alpha-spectrometry and its recovery is assessed by measuring the 133Ba on the same filter using gamma-spectrometry. The filtrate containing uranium is passed through a Dowex AG 1x4 ion-exchange resin in the SO42- form which retains uranium while other ions are eluted by dilute (0.25M) sulphuric acid. Uranium is eluted from the column by distilled water, electrodeposited on a silver disc and the uranium isotopes and their recovery are determined by alpha-spectrometry. The method was tested on a variety of natural and spiked water samples with known concentrations of 226Ra and 238U and was found to yield accurate results within ±10% RSD of the target values.
Similar content being viewed by others

Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Burns, K. Determination of radium and uranium isotopes in natural waters by sorption on hydrous manganese dioxide followed by alpha-spectrometry. J Radioanal Nucl Chem 264, 437–443 (2005). https://doi.org/10.1007/s10967-005-0734-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10967-005-0734-5