Abstract
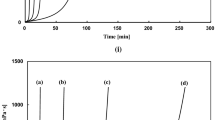
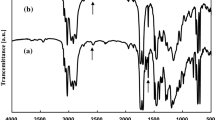
Network polymers have been successfully synthesized by ring-opening addition reaction of multi-functional epoxy with α,ω-dithiol compounds in the presence of triethyl amine as a base catalyst. The reactions with alkane dithiol in dimethyl sulfoxide yielded transparent gels. Young’s modulus of the gels, which was evaluated by compression test, increased with increasing in number of epoxy group of the multi-functional epoxy compound and decreasing in methylene length of the alkane dithiol compound used due. The reaction of pentaerythritol polyglycidyl ether (PPE) with a dithiol compound having polyethylene glycol linkage, 1,2-bis(2-mercaptoethoxy)ethane (BME), in ethanol induced phase separation during the polymerization and produced the porous polymer under the appropriate conditions (temperature, monomer concentration, and catalyst amount). The porous polymers showed the surface morphology formed by connected particles in the diameters ranged from about 2 to 30 μm. The particles size tended to decrease with increasing of the monomer concentration and decreasing of the catalyst concentration. The PPE-BME porous polymers were not breakable under the compression of 50 N, and their Young’s moduli increased with increasing in the monomer concentration and the amount of the catalyst in the preparation reaction systems.








Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Code availability
Not applicable.
References
Jin FL, Li X, Park SJ (2015) Synthesis and application of epoxy resins. Rev J Ind Eng Chem 29:1–11. https://doi.org/10.1016/j.jiec.2015.03.026
Mohan P (2013) A Critical Review: The Modification, Properties, and Applications of Epoxy Resins. Polym-Pastic Tech Eng 52:107–125. https://doi.org/10.1080/03602559.2012.727057
Naga N, Oda E, Toyota A, Horie K, Furukawa H (2006) Tailored synthesis and fundamental characterization of organic-inorganic hybrid gels by means of hydrosilylation reaction. Macromol Chem Phys 207:627–635. https://doi.org/10.1002/macp.200500501
Naga N, Kihara Y, Miyanaga T, Furukawa H (2009) Synthesis of Organic-Inorganic Hybrid Gels from Siloxane or Silsesquioxane and α,ω-Nonconjugated Dienes by Means of a Photo Hydrosilylation Reaction. Macromolecules 42:3454–3462. https://doi.org/10.1021/ma802745x
Naga N, Nagino H, Furukawa H (2016) Synthesis of organic-inorganic hybrid gels by means of thiol-ene and azide-alkene reactions. J Polym Sci Part A Polym Chem 54:2229–2238. https://doi.org/10.1002/pola.28096
Ahmed K, Inagaki A, Naga N (2020) Joint-Linker Type Ionic Gels Using Polymerizable Ionic Liquid as s Crosslinker via Thiol-Ene Click Reactions. Polymers 12:2844. https://doi.org/10.3390/polym12122844
Naga N, Sato M, Ahmed K, Nakano T (2021) Synthesis of gels by means of Michael addition reaction of multifunctional-acetoacetate and diacrylate compounds and their application to ionic conductive gels. J Polym Sci 59:2129–2139. https://doi.org/10.1002/pol.20210388
Naga N, Michida R, Kudo S, Nagami Y, Miriyama K, Nageh H, Furukawa H, Nakano T (2019) Synthesis of joint-linker type gels and porous polymers by addition reactions of multi-functional thiol and alkyl diacrylate, diisocyanate compounds. Mater Today Commun 153–162. https://doi.org/10.1016/j.mtcomm.2018.11.013
Naga N, Fujioka S, Inose D, Ahmed K, Nageh H, Makano T (2020) Synthesis and properties of porous polymers synthesized by Michael addition reactions of multi-functional acrylate and diamine, dithiol compounds. RSC Adv 10:60–69. https://doi.org/10.1039/C9RA09684A
Naga N, Miyanaga T, Wang Y, Nakano T (2020) Synthesis and properties of σ-π conjugated porous polymers obtained with Mizoroki-Heck reaction of tetra vinyl cyclic siloxane with dibromo fluorene. J Polym Sci 58:2301–2309. https://doi.org/10.1002/pol.20200268
Naga N, Sato M, Mori K, Nageh H, Nakano T (2020) Synthesis of Network Polymers by Means of Addition Reactions of Multifunctional-Amine and Poly(ethylene glycol) Diglycidyl Ether or Diacrylate Compounds. Polymers 12:2047. https://doi.org/10.3390/polym12092047
Miyanaga T, Naga N (2020) Phase and morphology control of organic-inorganic hybrid network polymer by means of hydrosilylation reaction of cubic silsesuquioxane or cyclic siloxane and divinyl or diallyl compounds. Chem Lett 49:1236–1239. https://doi.org/10.1246/cl.200421
Naga N, Yamada K, Moriyama K, Kudoh S, Nagami Y, Nageh H, Nakano T (2021) Synthesis of network polymers by photo-initiated thiol-ene reaction between multi-functional thiol and poly(ethylene glycol) diacrylate. Polym Bull 79:2411–2427. https://doi.org/10.1007/s00289-021-03643-8
Naga N, Ito M, Mezaki A, Tang HC, Chang TFM, Sone M, Nageh H, Nakano T (2021) Morphology Control and Metallization of Porous Polymers Synthesized by Michael Addition Reactions of a Multi-Functional Acrylamide with a Diamine. Materials 14:800. https://doi.org/10.3390/ma14040800
Naga N, Inose D, Ishida T, Kubota K, Nageh H, Nakano T (2021) Synthesis of polymer networks by means of addition reactions of tri-amine and poly(ethylene glycol) diacrylate or diglycidyl ether compounds. Polym Bull 78:2745–2763. https://doi.org/10.1007/s00289-020-03241-0
Naga N, Hasegawa K, Nageh H, Nakano T (2021) Synthesis and properties of degradable gels and porous polymers including acetal group in the network structure by addition reaction of multi-functional phenols and divinyl ether compounds. Polym Bull 77:5631–5645. https://doi.org/10.1007/s00289-019-03033-1
Naga N, Sato M, Magara T, Ahmed K, Nakano T (2022) Synthesis of porous polymers by means of Michael addition reaction of multifunctional-acetoacetate and poly(ethylene glycol) diacrylate. Eur Polym J 162:110901. https://doi.org/10.1016/j.eurpolymj.2021.110901
Brandle A, Khan A (2012) Thiol–epoxy ‘click’ polymerization: efficient construction of reactive and functional polymers. Polym Chem 3:3224–3227. https://doi.org/10.1039/C2PY20591B
Stuparu MC, Khan A (2016) Thiol-epoxy “click” chemistry: Application in preparation and postpolymerization modification of polymers. J Polym Sci Part A Poym Chem 54:3057–3070. https://doi.org/10.1002/pola.28195
Chen L, Zhwng Y, Meng X, Wei G, Dietliker K, Li Z (2020) Delayed Thiol-Epoxy Photopolymerization: A General and Effective Strategy to Prepare Thick Composites. ACS Omega 5:15192–15201. https://doi.org/10.1021/acsomega.0c01170
Jin K, Wilmot N, Heath WH, Torkelson JM (2016) Phase-Separated Thiol–Epoxy–Acrylate Hybrid Polymer Networks with Controlled Cross-Link Density Synthesized by Simultaneous Thiol-Acrylate and Thiol-Epoxy Click Reactions. Macromolecules 49:4115–4123. https://doi.org/10.1021/acs.macromol.6b00141
Stuparu MC, Khan A (2018) Thiol-epoxy and Amine-epoxy ‘Click’ Polymerizations. Click Polym Chapter 6. https://doi.org/10.1039/9781788010108-00191
Dhulst EA, Heath WH, Torkelson JM (2016) Hybrid thiol-acrylate-epoxy polymer networks: Comparison of one-pot synthesis with sequential reactions and shape memory properties. Polymer 96:198–204. https://doi.org/10.1016/j.polymer.2016.04.032
Grauzeliene S, Navaruckiene A, Skliutas E, Malinauskas M, Serra A, Ostrauskaite J (2021) Vegetable Oil-Based Thiol-Ene/Thiol-Epoxy Resins for Laser Direct Writing 3D Micro-/Nano-Lithography. Polymers 13:872. https://doi.org/10.3390/polym13060872
De S, Khan A (2012) Efficient synthesis of multifunctional polymersviathiol–epoxy “click” chemistry. Chem Commun 48:3130–3132. https://doi.org/10.1039/C2CC30434A
Xin F, Han J, Pan H, Sun F (2019) Surface microstructures and properties of thiol-epoxy/thiol-Si-methacrylate hybrid polymer networks prepared by UV-induced polymerization. J Macromol Sci Part A 57:355–362. https://doi.org/10.1080/10601325.2019.1698965
Carioscia JA, Stansbury JW, Bowman CN (2007) Evaluation and control of thiol–ene/thiol–epoxy hybrid networks. 48:1526–1532. https://doi.org/10.1016/j.polymer.2007.01.044
Wang Y, Tang L, Li Y, Li Q (2022) Effects of networks composed of epoxy/dual thiol-curing agents on properties of shape memory polymers. 139:51548. https://doi.org/10.1002/app.51548
Funding
This work was partially supported by JSPS KAKENHI Grant Number 24550261.
Author information
Authors and Affiliations
Contributions
Conceptualization, N.N. and T.N.; analysis, N.N. and Y.H.; investigation, N.N. and Y.H.; writing—original draft preparation, N.N.; writing—review and editing, T.N.; supervision, N.N. and T.N.; project administration, N.N.; funding acquisition, N.N. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Naga, N., Hosoi, Y. & Nakano, T. Synthesis of network polymers by ring-opening addition reaction of multi-functional epoxy and dithiol compounds. J Polym Res 30, 42 (2023). https://doi.org/10.1007/s10965-022-03429-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-022-03429-0