Abstract
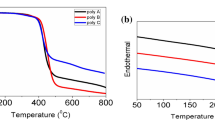
Considering the significant synthetic efforts for the preparation of poly(3,4-ethylenedioxythiophene) (PEDOT) and its soluble derivatives, very little is known about poly(3,4-ethylenedioxyselenophene) (PEDOS), especially for soluble polymers. Here, we report the synthesis of soluble dodecyl-substituted poly(3,4-ethylenedioxyselenophene) (PEDOS-C12) by two different chemical polymerization methods- transition metal-mediated and Grignard metathesis (GRIM). The polymer PEDOS-C12 prepared by GRIM was characterized using NMR, gel permeation chromatography, UV–vis-NIR spectroscopy, cyclic voltammetry and shows high conductivity. The obtained neutral polymer PEDOS-C12 (hydrazine-dedoped) prepared by GRIM was used for spray processable electrochromic property and organic solar cells. The optical contrast ratio and coloration efficiency of the polymer film were found to be 20.8% and 131.9 cm2/C at 770 nm. The photovoltaic device was fabricated using solution-processable PEDOS-C12 as hole transport layer (HTL) with the simple geometry ITO/PEDOS-C12(HTL)/P3HT:PC61BM/Al and achieved a power conversion efficiency up to ~ 0.52% under ambient conditions.






Similar content being viewed by others
References
Skotheim TA, Reynolds JR (eds) (2007) Handbook of conducting polymers, 3rd ed. CRC Press, Boca Raton, FL
Perepichka IF, Perepichka DF (eds) (2009) Handbook of thiophene-based materials. Wiley-VCH, Chichester, UK
Swager TM (2017) 50th anniversary perspective: Conducting/semiconducting conjugated polymers. A personal perspective on the past and the future. Macromolecules 50:4867–4886
Mishra A, Ma CQ, Bäuerle P (2009) Functional oligothiophenes: molecular design for multidimensional nanoarchitectures and their applications. Chem Rev 109:1141–1276
Gupta S, Patra A (2000) Facile polymerization method for poly(3,4-ethylenedioxythiophene) and related polymers using iodine vapour. New J Chem 44:6883–6888
Chen J, Shao M, Xiao K, He Z, Li D, Lokitz BS, Hensley DK, Kilbey SM, Anthony JE, Keum JK, Rondinone AJ, Lee W-Y, Hong S, Bao Z (2013) Conjugated polymer-mediated polymorphism of a high performance, small-molecule organic semiconductor with tuned intermolecular interactions, enhanced long-range order, and charge transport. Chem Mater 25:4378–4386
Patra A, Bendikov M, Chand S (2014) Poly(3,4-ethylenedioxyselenophene) and its derivatives: novel organic electronic materials. Acc Chem Res 47:1465–1474
Patra A, Kumar R, Chand S (2014) Selenium-containing p-conjugated polymers for organic solar cells. Isr J Chem 54:621–641
Patra A, Bendikov M (2010) Polyselenophenes. J Mater Chem 20:422–433
Yadav P, Patra A (2020) Recent advances in poly(3,4-ethylenedioxyselenophene) and related polymers. Polym Chem 11:7275–7292
Yadav P, Singhal S, Patra A (2020) Electropolymerized poly(3,4-ethylenedioxyselenophene) on flexiblesubstrate: A comparative study of electronic and electrochromic properties with sulfur analogue and rigid substrate. Synth Met 260:116264
Yadav P, Naqvi S, Patra A (2020) Poly (3, 4-ethylenedioxyselenophene): effect of solvent and electrolyte on electrodeposition, optoelectronic and electrochromic properties. RSC Adv 10:12395–12406
Patra A, Wijsboom YH, Zade SS, Li M, Sheynin Y, Leitus G, Bendikov M (2008) Poly(3,4-ethylenedioxyselenophene). J Am Chem Soc 130:6734–6736
Zhang W, Zhang W, Xue Z, Xue Y, Xu J, Chen S, Zhang G (2018) Effect of chalcogen substitution on aqueous dispersions of poly(3,4-ethylenedioxythiophene)s:poly(4-styrenesulfonate) and their flexible conducting films. J Mater Sci Mater Electron 29:18566–18572
Patra A, Agrawal V, Bhargav R, Shahjad D, Bhardwaj S, Chand Y, Sheynin MB (2015) Metal free conducting PEDOS, PEDOT, and their analogues via an unusual bromine-catalyzed polymerization. Macromolecules 48:8760–8764
Li M, Sheynin Y, Patra A, Bendikov M (2009) Tuning the electrochromic properties of poly(alkyl-3,4-ethylenedioxyselenophenes) having high contrast ratio and coloration efficiency. Chem Mater 21:2482–2488
Li M, Patra A, Sheynin Y, Bendikov M (2009) Hexyl-derivatized poly(3,4-ethylenedioxyselenophene): novel highly stable organic electrochromic material with high contrast ratio, high coloration efficiency, and low-switching voltage. Adv Mater 21:1707–1711
Amb CM, Beaujuge PM, Reynolds JR (2010) Spray-processable blue-to-highly transmissive switching polymer electrochromes via the donor-acceptor approach. Adv Mater 22:724–728
Kumar A, Reynolds JR (1996) Soluble alkyl-substituted poly(ethylenedioxythiophenes) as electrochromic materials. Macromolecules 29:7629–7630
Henson ZB, Müllen K, Bazan GC (2012) Design strategies for organic semiconductors beyond the molecular formula. Nat Chem 4:699–704
Cheng Y-J, Yang S-H, Hsu C-S (2009) Synthesis of conjugated polymers for organic solar cell applications. Chem Rev 109:5868–5923
Dong H, Zhu H, Meng Q, Gong X, Hu W (2012) Organic photoresponse materials and devices. Chem Soc Rev 41:1754–1808
Ye L, Zhang S, Huo L, Zhang M, Hou J (2014) Molecular design toward highly efficient photovoltaic polymers based on two-dimensional conjugated benzodithiophene. Acc Chem Res 47:1595–1603
Shahjad R, Bhargav D, Bhardwaj A, Mishra AP (2017) Synthesis and characterization of benzodithiophene-chalcogenophene based copolymers: a comparative study of optoelectronic properties and photovoltaic applications. Macromol Chem Phys 218:1700038
Hains AW, Ramanan C, Irwin MD, Liu J, Wasielewski MR, Marks TJ (2010) Designed bithiophene-based interfacial layer for high-efficiency bulk-heterojunction organic photovoltaic cells. Importance of interfacial energy level matching. ACS Appl Mater Interfaces 2:175–185
Li SS, Tu KH, Lin CC, Chen CW, Chhowalla M (2010) Solution-processable graphene oxide as an efficient hole transport layer in polymer solar cells. ACS Nano 4:3169–3174
Liu J, Xue Y, Gao Y, Yu D, Durstock M, Dai L (2012) Hole and electron extraction layers based on graphene oxide derivatives for high-performance bulk heterojunction solar cells. Adv Mater 24:2228–2233
Jouad ZE, Morsli M, Louarn G, Cattin L, Addou M, Bernède J (2015) Improving the efficiency of subphthalocyanine based planar organic solar cells through the use of MoO3/CuI double anode buffer layer. Sol Energ Mat Sol Cells 141:429–435
Chaudhary N, Kesari JP, Chaudhary R, Patra A (2016) Low band gap polymeric solar cells using solution-processable copper iodide as hole transporting layer. Opt Mater 58:116–120
Chaudhary N, Chaudhary R, Kesari JP, Patra A, Chand S (2015) Copper thiocyanate (CuSCN): an efficient solution-processable hole transporting layer in organic solar cells. J Mater Chem C 3:11886–11892
Chaudhary N, Chaudhary R, Kesari JP, Patra A (2017) An eco-friendly and inexpensive solvent for solution processable CuSCN as a hole transporting layer in organic solar cells. Opt Mater 69:367–371
Irwin MD, Buchholz DB, Hains AW, Chang RPH, Marks TJ (2008) p-Type semiconducting nickel oxide as an efficiency-enhancing anode interfacial layer in polymer bulk-heterojunction solar cells. Proc Natl Acad Sci 105:2783–2787
Steim R, Kogler FR, Brabec CJ (2010) Interface materials for organic solar cells. J Mater Chem 20:2499–2512
Pan H, Zuo L, Fu W, Fan C, Andreasen B, Jiang X, Norrman K, Krebs FC, Chen H (2013) MoO3–Au composite interfacial layer for high efficiency and air-stable organic solar cells Org. Electron 14:797–803
Bhargav R, Chaudhary N, Rathi S, Shahjad D, Bhardwaj S, Gupta AP (2019) Copper bromide as an efficient solution-processable hole transport layer for organic solar cells: effect of solvents. ACS Omega 4:6028–6034
Bhargav R, Bhardwaj D, Shahjad A, Patra SC (2017) Poly (styrene sulfonate) free poly (3, 4-ethylenedioxythiophene) as a robust and solution-processable hole transport layer for organic solar cells. ChemistrySelect 1:1347–1352
He Z, Zhang Z, Bi S, Chen J, Li D (2020) Conjugated polymer controlled morphology and charge transport of small-molecule organic semiconductors. Sci Rep 10:4344
Bi S, Li Y, He Z, Ouyang Z, Guo Q, Jiang C (2019) Self-assembly diketopyrrolopyrrole-based materials and polymer blend with enhanced crystal alignment and property for organic field-effect transistors. Org Electron 65:96–99
Chaudhary N, Chaudhary R, Kesari JP, Patra A (2018) Effect of composition ratio of P3HT:PC61BM in organic solar cells: optical and morphological properties. Mater Res Innov 22(5):282–286
Heeney M, Zhang W, Crouch DJ, Chabinyc ML, Gordeyev S, Hamilton R, Higgins SJ, McCulloch I, Skabara PJ, Sparrowe D, Tierney S (2007) Regioregular poly(3-hexyl)selenophene: a low band gap organic hole transporting polymer. Chem Commun 5061–5063
Bhardwaj D, Shahjad S, Gupta P, Yadav R, Bhargav AP (2017) All conjugated poly(3-hexylthiophene)-block-poly(hexyl-3,4-ethylenedioxythiophene) copolymers. ChemistrySelect 2:9557–9562
Organic solar cells using solid-state deposition of PEDOT as a HTL with geometry of ITO/PEDOT(HTL)/P3HT:PC BM/Al and a PCE 0.71% has been reported (for details see reference 36)
Acknowledgements
S. N. acknowledges SERI-DST, New Delhi and P. Y. acknowledges CSIR, New Delhi, for their fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Naqvi, S., Yadav, P., Pahari, P. et al. Dodecyl-substituted poly(3,4-ethylenedioxyselenophene): polymerization and its solution-processable applications for electrochromic and organic solar cells. J Polym Res 28, 250 (2021). https://doi.org/10.1007/s10965-021-02609-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-021-02609-8