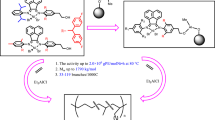
Abstract
A series of salicylaldimine ligands 2-tert-butyl-6-((2,4-bis(diphenylmethyl)-8-arylnaphthalen)imine)-phenol (L1: Aryl =4-tBu-phenyl; L2: Aryl = p-tolyl; L3: Aryl = phenyl; L4: 4-CF3-phenyl) and the corresponding neutral salicylaldimine nickel complexes (Ni1-Ni4) were prepared and characterized. In ethylene polymerization at 20 °C, these nickel complexes are highly active, with activities of up to 6.88 × 105 g·mol−1·h−1. Moreover, the molecular weight of the polyethylene could reach up to 8.1 × 105. Comparing with the classic salicylaldimine nickel complexes Ni5 at 20 °C, complexes Ni1 with the 4-tBu-phenyl substituent demonstrated 8 times higher activity and 35 times higher polyethylene molecular weight.

Graphical abstract



Similar content being viewed by others
References
Ittel SD, Johnson LK, Brookhart M (2000) Chem Rev 100:1169–1204
Nakamura A, Ito S, Nozaki K (2009) Chem Rev 109:5215–5244
Chen EYX (2009) Chem Rev 109:5157–5214
Keim W, Kowaldt FH, Goddard R, Krüger C (1978) Angew Chem Int Ed 17:466–467
Keim W (2013) Angew Chem Int Ed 52:12492–12496
Johnson LK, Killian CM, Brookhart M (1995) J Am Chem Soc 117:6414–6415
Johnson LK, Mecking S, Brookhart M (1996) J Am Chem Soc 118:267–268
Mecking S, Johnson LK, Wang L, Brookhart M (1998) J Am Chem Soc 120:888–899
Camacho DH, Guan ZB (2010) Chem Commun 46:7879–7893
Dong ZM, Ye ZB (2012) Polym Chem 3:286–301
Ye ZB, Xu LX, Dong ZM, Xiang P (2013) Chem Commun 49:6235–6255
Takeuchi D (2012) Polym J 44:919–928
Chen YS, Wang L, Yu HJ, Zhao YL, Sun R, Jing GH, Huang J, Khalid H, Abbasi NM, Akram M (2015) Prog Polym Sci 45:23–43
Guo LH, Chen CL (2015) SCIENCE CHINA Chem 58:1663–1673
Guo LH, Dai SY, Sui XL, Chen CL (2016) ACS Catal 6:428–441
Guan Z, Cotts PM, McCord EF, McLain SJ (1999) Science 283:2059–2062
Leung DH, Ziller JW, Guan Z (2008) J Am Chem Soc 130:7538–7539
Chen CL, Luo S, Jordan RF (2008) J Am Chem Soc 130:12892–12893
Liu FS, Hu HB, Xu Y, Guo LH, Zai SB, Song KM, Gao HY, Zhang L, Zhu FM, Wu Q (2009) Macromolecules 42:7789–7796
Wegner MM, Ott AK, Rieger B (2010) Macromolecules 43:3624–3633
Chen CL, Jordan RF (2010) J Am Chem Soc 132:10254–10255
Chen CL, Luo S, Jordan RF (2010) J Am Chem Soc 132:5273–5284
Liu H, Zhao WZ, Hao X, Redshaw C, Huang W, Sun WH (2011) Organometallics 30:2418–2424
Zhang D, Nadres ET, Brookhart M, Daugulis O (2013) Organometallics 32:5136–5143
Rhinehart JL, Brown LA, Long BK (2013) J Am Chem Soc 135:16316–16319
Rhinehart JL, Mitchell NE, Long BK (2014) ACS Catal 4:2501–2504
Vaidya T, Klimovica K, LaPointe AM, Keresztes I, Lobkovsky EB, Daugulis O, Coates GW (2014) J Am Chem Soc 136:7213–7216
Zhu L, Fu ZS, Pan HJ, Feng W, Chen CL, Fan ZQ (2014) Dalton Trans 43:2900–2906
Wang RK, Sui XL, Pang WM, Chen CL (2016) ChemCatChem 8:434–440
Wang RK, Zhao MH, Chen CL (2016) Polym Chem 7:3933–3938
Zou WP, Chen CL (2016) Organometallics 35:1794–1801
Guo L, Gao H, Guan Q, Hu H, Deng J, Liu J, Liu F, Wu Q (2012) Organometallics 31:6054–6062
Nakamura A, Anselment TMJ, Claverie J, Goodall B, Jordan RF, Mecking S, Rieger B, Sen A, Van Leeuwen PWNM, Nozaki K (2013) Acc Chem Res 46:1438–1449
Carrow BP, Nozaki K (2014) Macromolecules 47:2541–2555
Contrella ND, Sampson JR, Jordan RF (2014) Organometallics 33:3546–3555
Ota Y, Ito S, Kuroda J, Okumura Y, Nozaki K (2014) J Am Chem Soc 136:11898–11901
Nakano R, Nozaki K (2015) J Am Chem Soc 137:10934–10937
Jian ZB, Moritz BC, Mecking S (2015) J Am Chem Soc 137:2836–2839
Jian ZB, Mecking S (2015) Angew Chem Int Ed 54:15845–15849
Zhang YL, Cao YC, Leng XB, Chen C, Huang Z (2014) Organometallics 33:3738–3745
Sui XL, Dai SY, Chen CL (2015) ACS Catal 5:5932–5937
Chen M, Yang BP, Chen CL (2015) Angew Chem Int Ed 54:15520–15524
Chen M, Yang BP, Chen CL (2016) Synlett 27:1297–1302
Wu ZX, Chen M, Chen CL (2016) Organometallics 35:1472–1479
Zhou XY, Bontemps S, Jordan RF (2008) Organometallics 27:4821–4824
Guironnet D, Runzi T, Gottker-Sechnetmann I, Mecking S (2008) Chem Commun 132:4965–4967
Noda S, Kochi T, Nozaki K (2009) Organometallics 28:656–658
Perrotin P, McCahill JSJ, Wu G, Scott SL (2011) Chem Commun 47:6948–6950
Ito S, Ota Y, Nozaki K (2012) Dalton Trans 41:13807–13809
Chen M, Zou WP, Cai ZG, Chen CL (2015) Polym Chem 6:2669–2676
Younkin TR, Connor EF, Henderson JI, Friedrich SK, Grubbs RH, Bansleben DA (2000) Science 287:460–462
Wang C, Friedrich S, Younkin TR, Li RT, Grubbs RH, Bansleben DA, Day MW (1998) Organometallics 17:3149–3151
Mu HL, Pan L, Song DP, Li YS (2015) Chem Rev 115:12091–12137
Makio H, Terao H, Iwashita A, Fujita T (2011) Chem Rev 111:2363–2449
Delferro M, McInnis JP, Marks TJ (2010) Organometallics 29:5040–5049
Weberski MP, Chen CL, Delferro M, Zuccaccia C, Macchioni A, Marks TJ (2012) Organometallics 31:3773–3789
Weberski MP, Chen CL, Delferro M, Marks TJ (2012) Chem Eur J 18:10715–10732
Osichow A, Göttker-Schnetmann I, Mecking S (2013) Organometallics 32:5239–5242
Stephenson CJ, McInnis JP, Chen CL, Weberski MP, Motta A, Delferro M, Marks TJ (2014) ACS Catal 4:999–1003
Wiedemann T, Voit G, Tchernook A, Roesle P, Göttker-Schnetmann I, Mecking S (2014) J Am Chem Soc 136:2078–2085
Ölscher F, Göttker-Schnetmann I, Monteil V, Mecking S (2015) J Am Chem Soc 137:14819–14828
Wang JC, Yao ED, Chen ZT, Ma YG (2015) Macromolecules 48:5504–5510
Hu XH, Dai SY, Chen CL (2016) Dalton Trans 45:1496–1503
Chen Z, Mesgar M, White PS, Daugulis O, Brookhart M (2015) ACS Catal 5:631–636
Liu H, Sun WH (2011) Organometallics 30:2418–2424
Kong SL, Song KF, Liang TL, Guo CY, Sun WH, Redshaw C (2013) Dalton Trans 42:9176–9187
Guo LH, Sui XL, Dai SY, Chen CL (2016) Polymer 8:37
Dai SY, Sui XL, Chen CL (2015) Angew Chem Int Ed 54:9948–9953
Dai SY, Chen CL (2016) Angew Chem Int Ed 55:13281–13285
Dai SY, Sui XL, Chen CL (2016) Chem Commun 52:9113–9116
Klein HF, Karsch HH (1972) Chem Ber 105:2628–2636
Nadres ET, Santos GIF, Shabashov D, Daugulis O (2013) J Organomet Chem 78:9689–9714
Diamanti SJ, Ghosh P, Shimizu F, Bazan GC (2003) Macromolecules 36:9731–9735
Acknowledgements
This work was supported by Advanced Catalysis and Green Manufacturing Collaborative Innovation Center (ACGM2016-06-01), National Natural Science Foundation of China (NSFC, 21304054), Yixing Taodu Ying Cai Program, Jiangsu Province Science and Technology Support Project (BE2014103), Foundation of Qufu Normal University (xkJ201603), National College Students Innovation Project (201610446029).
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
ESM 1
(DOCX 4076 kb)
Rights and permissions
About this article
Cite this article
Ji, P., Guo, L., Hu, X. et al. Ethylene polymerization by salicylaldimine Nickel(II) complexes derived from arylnaphthylamine. J Polym Res 24, 30 (2017). https://doi.org/10.1007/s10965-017-1190-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-017-1190-y