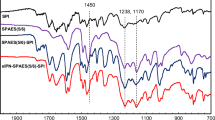
Abstract
A poly(amide-imide) (PAI) bearing imidazole groups on the polymer chain was synthesized via direct polycondensation of a synthesized diacid-diimide and 4,4′-(1,4-Phenylenediisopropylidene) bisaniline (PDBA). Diacide-diimide was synthesized by the condensation of an amino acid compound, (S)-(+)-Histidine hydrochloride monohydrate and 3,3′,4,4′-Benzophenone tetracarboxylic dianhydride (BTDA). On the other hand, a sulfonated polyimide (SPI) was also synthesized by the solution imidization of sulfonated, (4,4-diaminostilbene-2,2-disulfonic acid) (DSDSA) and non-sulfonated, 4,4′-(1,4-Phenylenediisopropylidene) bisaniline (PDBA) diamines in reaction with a six-membered naphthalene base dianhydride, 1,4,5,8-Naphthalenetetracarboxylic dianhydride (NTDA). A strong and flexible SPI membrane with good uniformity and proper thermal and mechanical properties was achieved. The SPI was then blended with different amounts of PAI and doped with phosphoric acid (PA), in order to investigate the blending influence of PAI in PA-doped blend membranes compared to the pure SPI membrane. It was found that a proper amount of PAI could effectively improve the water uptake, IEC and proton conductivity of the PA doped SPI/PAI membranes. Nevertheless the excess PAI negatively affected the membrane properties. The pure SPI with an IEC of 1.76 meq.g−1 showed a proton conductivity of 29.4 mS cm−1 at 120 °C, while PA doped SPI/PAI-10 % (w/w) as the most optimal PAI containing sample, with an IEC of 2.23, showed a proton conductivity of 69.7 mS cm−1 at 140 °C. The proton conductivity measurements were performed at 40 % relative humidity.















Similar content being viewed by others
References
Zhang H, Shen PK (2012) Chem Rev 112:2780–2832
Hickner MA, Ghassemi H, Kim YS, Einsla BR (2004) Chem Rev 104:4587–4612
Arslantas A, Sinirlioglu D, Eren F, Muftuoglu AE, Bozkurt A (2014) J Polym Res 21:437
Wang Y, Chen KS, Mishler J, Cho SC, Adroher XC (2011) Appl Energy 88:981–1007
Abu-Saied MA, Fontananova E, Drioli E, Eldin MSM (2013) J Polym Res 20:187
Zhang J, Xie Z, Zhang J, Tang Y, Song C (2006) J Power Sources 160:872–891
Einsla ML, Kim YS, Hawley M, Lee HS, McGrath JE, Liu B, Guiver MD, Pivovar BS (2008) Chem Mater 20:5636–5642
Bose S, Kuila T, Nguyen TXH, Kim NH, Lau KT, Lee JH (2011) Prog Polym Sci 36:813–843
Ma L, Xu J, Han S, Yang M, Wang Z, Ni H, Gui Y (2014) J Polym Res 21:1–10
Chandan A, Hattenberger M, El-Kharouf A, Du S, Dhir A, Self V, Pollet BG, Ingram A, Bujalski W (2013) J Power Sources 231:264–278
Yuan S, Guo X, Aili D, Pan C, Li Q, Fang J (2014) J Membr Sci 454:351–358
Xu JM, Cheng HL, Ma L, Han HL, Huang YS, Wang Z (2014) J Polym Res 21:423
He R, Li Q, Xiao G, Bjerrum NJ (2003) J Membr Sci 226:169–184
Seel DC, Benicewicz BC (2012) J Membr Sci 405:57–67
Mader JA, Benicewicz BC (2010) Macromolecules 43:6706–6715
Lee KH, Lee SY, Shin DW, Wang C, Ahn SH, Lee KJ, Guiver MD, Lee YM (2014) Polymer 55:1317–1326
Asano N, Aoki M, Suzuki S, Miyatake K, Uchida H, Watanabe M (2006) J Am Chem Soc 128:1762–1769
Hu Z, Yin Y, Kita H, Okamoto KI, Suto Y, Wang H, Kawasato H (2007) Polymer 48:1962–1971
Yin Y, Yamada O, Tanaka K, Okamoto K, Liu S, Ye H, Zhou Y, Zhao J, Yanai H, Sato T (2006) Polym J 38:197–219
Lee CH, Park CH, Lee YM (2008) J Membr Sci 313:199–206
Einsla BR, Kim YS, Hickner MA, Hong YT, Hill ML, Pivovar BS, McGrath JE (2005) J Membr Sci 255:141–148
Liaw DJ, Wang KL, Huang YC, Lee KR, Lai JY, Ha CS (2012) Prog Polym Sci 37:907–974
Akbarian-Feizi L, Mehdipour-Ataei S, Yeganeh H (2010) Int J Hydrog Energy 35:9385–9397
Jiang G, Qiao J, Hong F (2012) Int J Hydrog Energy 37:9182–9192
Seo DW, Lim YD, Lee SH, Jeong IS, Kim DI, Lee JH, Kim WG (2012) Int J Hydrog Energy 37:6140–6147
Sinirlioglu D, Muftuoglu AE, Bozkurt A (2013) J Polym Res 20:242
Zhao C, Lin H, Han M, Na H (2010) J Membr Sci 353:10–16
Li X, Liu C, Zhang S, Yu G, Jian X (2012) J Membr Sci 423:128–135
Lin HL, Hu CR, Lai SW, Yu TL (2012) J Membr Sci 389:399–406
Li Q, Rudbeck HC, Chromik A, Jensen JO, Pan C, Steenberg T, Calverley M, Bjerrum N, Kerres J (2010) J Membr Sci 347:260–270
Yu S, Benicewicz BC (2009) Macromolecules 42:8640–8648
Xu H, Chen K, Guo X, Fang J, Yin (2007) Polymer 48:5556–5564
Li Q, Jensen JO, Savinell RF, Bjerrum NJ (2009) Prog Polym Sci 34:449–477
Pu HT, Qiao L, Liu QZ, Yang ZL (2005) Eur Polym J 41:2505–2510
Pu H, Meyer WH, Wegner G (2002) J Polym Sci B Polym Phys 40:663–669
Pu H, Wang D (2006) Electrochim Acta 51:5612–5617
Hazarika M, Jana T (2012) ACS Appl Mater Interfaces 4:5256–5265
Wu H, Shen X, Cao Y, Li Z, Jiang Z (2014) J Membr Sci 451:74–84
Boaventura M, Ponce M, Brandao L, Mendes A, Nunes S (2010) Int J Hydrog Energy 35:12054–12064
Wasserscheid P, Welton T (2008) Ionic liquids in synthesis, vol. 1. Wiley Online Library, Weinheim, Germany
Mistri EA, Mohanty AK, Banerjee S (2012) J Membr Sci 411:117–129
Zhang F, Li N, Cui Z, Zhang S, Li S (2008) J Membr Sci 314:24–32
Li N, Cui Z, Zhang S, Xing W (2007) J Membr Sci 295:148–158
Wu S, Qiu Z, Zhang S, Yang X, Yang F, Li Z (2006) Polymer 47:6993–7000
Yang J, Li Q, Jensen JO, Pan C, Cleemann LN, Bjerrum NJ, He R (2012) J Power Sources 205:114–121
Schmidt C, Schmidt‐Naake G (2007) Macromol Mater Eng 292:1164–1175
He R, Li Q, Bach A, Jensen JO, Bjerrum NJ (2006) J Membr Sci 277:38–45
Alberti G, Casciola M, Massinelli L, Bauer B (2001) J Membr Sci 185:73–81
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kowsari, E., Ansari, V., Moradi, A. et al. Poly(amide-imide) bearing imidazole groups/sulfonated polyimide blends for low humidity and medium temperature proton exchange membranes. J Polym Res 22, 77 (2015). https://doi.org/10.1007/s10965-015-0729-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-015-0729-z