Abstract
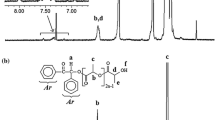
A novel well-defined end-chain functional macrophotoinitiator of poly(ε − caprolactone) (BPI-PCL) with controlled molecular weight and narrow molecular weight distribution was successfully synthesized by combination of ring opening polymerization (ROP) and “click chemistry”. The synthetic procedure for the preparation of a macrophotoinitiator involves three steps: (1) an azido-functionalized benzoin photoinitiator (BPI-N3) by transformation of a bromine-end group of BPI-Br compound into an azido group using NaN3; (2) an alkyne end-functionalized PCL (alkyne-PCL) precursor by ROP of ε − caprolactone (ε − CL) using propargyl alcohol as the initiator and stannous-2-ethylhexanoate (Sn(Oct)2) as the catalyst; and (3) BPI-PCL through “click reaction” between alkyne-PCL and BPI-N3 using Cu(I) as a catalyst. The 1H NMR, FTIR, UV–vis, fluorescence and gel permeation chromatography techniques were used to characterize the chemical structures of the intermediates and the target macrophotoinitiator. The obtained BPI-PCL macrophotoinitiator was used as a precursor in photoinitiated free radical promotedcationic polymerization to synthesize a poly(ε − caprolactone)-poly(cyclohexene oxide) (PCL-PCHO) block copolymer. The thermal behavior of the polymers was studied by differential thermal analysis and thermogravimetric analysis methods.











Similar content being viewed by others
References
Carlini C, Angiolini L, Caretti D, Corelli E (1996) Polym Adv Technol 7:379–384
Corrales T, Catalina F, Peinado C, Allen NS (2003) J Photochem Photobiol A159:103–114
Yagci Y, Mishra MK (1994) In: Mishra MK (ed) Macroinitiators. Polymer Frontiers International, Inc, New York, pp 229–264, Ch 6
Durmaz YY, Tasdelen MA, Aydogan B, Kahveci MU, Yagci Y (2009) In: Khosravi E, Yagci Y, Savelyev Y (eds) Light induced processes for the synthesis of polymers with complex structures, the NATO science for peace and security programme. Springer, Dordrecht, pp 329–341, Part 5
Degirmenci M, Hizal G, Yagci Y (2002) Macromolecules 35:8265–8270
Glaied O, Delaite C, Riess G (2012) Polym Bull 68:607–621
Degirmenci M, Acikses A, Genli N (2012) J Appl Polym Sci 123:2567–2573
Cabaret OD, Vaca BM, Bourissou D (2004) Chem Rev 104:6147–6176
Labet M, Thielemans W (2009) Chem Soc Rev 38:3484–3504
Sutar AK, Maharana T, Dutta S, Chen CT, Lin CC (2010) Chem Soc Rev 39:1724–1746
Chen HL, Chuang HJ, Huang BH, Lin CC (2013) Inorg Chem Commun 35:247–251
Kowalski A, Duda A, Penczek S (1998) Macromol Rapid Commun 19:567–572
Degirmenci M (2004) Polym J 36:542–548
Celik A, Kemikli N, Ozturk R, Muftuoglu AE, Yilmaz F (2009) React Funct Polym 69:705–713
Yurteri S, Cianga I, Degirmenci M, Yagci Y (2004) Polym Int 53:1219–1225
Colak D, Cianga I, Muftuoglu AE, Yagci Y (2006) J Polym Sci A Polym Chem 44:727–743
Hoogenboom R, Moore BC, Schubert US (2006) Chem Commun 38:4010–4012
Glaied O, Delaite C, Bistac S (2013) Crystalline properties of poly(ε-caprolactone)-block-poly(vinyl acetate) block copolymers: influence of synthesis route. Polym Int. doi:10.1002/pi.4575
Amici J, Kahveci MU, Allia P, Tiberto P, Yagci Y, Sangermano M (2012) J Mater Sci 47:412–419
Tasdelen MA (2011) Eur Polym J 47:937–941
Yang LP, Dong XH, Pan CY (2008) J Polym Sci A Polym Chem 46:7757–7772
Kolb HC, Finn MG, Sharpless KB (2001) Angew Chem Int Ed 40:2004–2021
Rostovtsev VV, Green LG, Fokin VV, Sharpless KB (2002) Angew Chem Int Ed 41:2596–2599
Eren O, Gorur M, Keskin B, Yilmaz F (2013) React Funct Polym 73:244–253
Su RJ, Yang HW, Leu YL, Hua MY, Lee RS (2012) React Funct Polym 72:36–44
He X, Liang L, Xie M, Zhang Y, Lin S, Yan D (2007) Macromol Chem Phys 208:1797–1802
Darcos V, Habnouni SE, Nottelet B, Ghzaoui AE, Coudane J (2010) Polym Chem 1:280–282
Riva R, Schmeits S, Jerome C, Jerome R, Lecomte P (2007) Macromolecules 40:796–803
Darcos V, Tabchi HA, Coudane J (2011) Eur Polym J 47:187–195
Shi GY, Pan CY (2009) J Polym Sci A Polym Chem 47:2620–2630
Tu Q, Wang JC, Liu R, Chen Y, Zhang Y, Wang DE, Yuan MS, Xu J, Wang J (2013) Colloids Surf B-Biointerfaces 108:34–43
Degirmenci M, Alter S, Genli N (2011) Macromol Chem Phys 212:1575–1581
Degirmenci M, Genli N (2009) Macromol Chem Phys 210:1617–1623
Zhao YF, Fan X, Chen X, Wan X, Zhou QF (2005) Polymer 46:5396–5405
Arnal ML, Balsamo V, Lopez-Carrasquero F, Contreras J, Carrillo M, Schmalz H, Abetz V, Laredo E, Müller AJ (2001) Macromolecules 7973–7982
Raquez JM, Degee P, Narayan R, Dubois P (2000) Macromol Rapid Commun 21:1063–1071
Nojima S, Tanaka H, Rohadi A, Sasaki S (1998) Polymer 39:1727–1734
Degirmenci M, Cianga I, Yagci Y (2002) Macromol Chem Phys 203:1279–1284
Yagci Y, Degirmenci M (2003) In: Matyjaszewski K (ed) Photoinduced free radical promoted cationic block copolymerization by using macrophotoinitiators prepared by ATRP and Ring-Opening Polymerization methods, ACS Symposium Series, American Chemical Society, Washington DC, vol. 854, Ch. 27, pp 383–393
Bottcher A, Hasebe K, Hizal G, Yagci Y, Stellberg P, Schnabel W (1991) Polymer 32:2289–2293
Dobis P, Barakat I, Jerome R, Teyssie P (1993) Macromolecules 26:4407–4412
Huang Y-p, Xu X, Luo X-l, Ma D-z (2002) Chin J Polym Sci 20:45–51
Lenoir S, Riva R, Lou X, Detrembleur C, Jerome R, Lecomte P (2004) Macromolecules 37:4055–4061
Acknowledgments
The authors would like to thank Harran University's Scientific Research Council (HÜBAK) for financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Degirmenci, M., Besli, P.A. & Genli, N. Synthesis of a well-defined end-chain macrophotoinitiator of poly(ε − caprolactone) by combination of ring-opening polymerization and click chemistry. J Polym Res 21, 540 (2014). https://doi.org/10.1007/s10965-014-0540-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-014-0540-2