Abstract
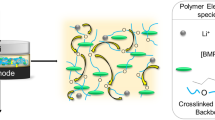
We report here a multiple-nitrile based lithium-salt liquid electrolyte. The ionic conductivity of poly (propyl ether imine) (abbreviated as PETIM) lithium salt dendrimer liquid electrolyte was observed to be a function of dendrimer generation number, n = 0 (monomer)−3. While the highest room temperature ionic conductivity value (~ 10−1 Sm−1) was recorded for the bis-2cyanoethyl ether monomer (i.e. zeroth generation; G 0 -CN), conductivity decreased progressively to lower values (~ 10−3 Sm−1) with increase in generation number (G 1 -CN → G 3 -CN). The G 0 -CN and higher dendrimer generations showed high thermal stability (≈150 to 200 °C), low moisture sensitivity and tunable viscosity (~10−2 (G 0 -CN) to 3 (G 3 -CN) Pa s). The linker ether group was found to be crucial for ion transport and also eliminated a large number of detrimental features, chiefly moisture sensitivity, chemical instability associated typically with prevalent molecular liquid solvents. Based on the combination of several beneficial physicochemical properties, we presently envisage that the PETIM dendrimers especially the G 0 -CN electrolytes hold promise as electrolytes in electrochemical devices such as lithium-ion batteries.





Similar content being viewed by others
References
Xu K (2004) Chem Rev 104(10):4303
Armand M, Endres F, MacFarlane DR, Ohno H, Scrosati B (2009) Nat Mater 8:621
Armand M, Tarascon J-M (2008) Nature 451:652
Barthel J, Gorres HJ (1999) Liquid nonaqueous electrolytes in Handbook of battery materials Ed. J. O. Besenhard. Besenhard Wiley-VCH, Weinheim
Galinski M, Lewandowski A, Stepniak I (2006) Electrochim Acta 51:5567
MacFarlane DR, Pringle JM, Howlett PC, Forsyth M (2010) Phys Chem Chem Phys 12:1659
Hwang B-J, Liu Y-C, Lin H-C (1997) J Polym Res 4:147
Liao C-S, Ye W-B (2003) J Polym Res 10:241
Li M, Ren W, Zhang Y, Zhang Y (2012) J Polym Res 19:9853
Zhjiang C (2006) J Polym Res 13:207
Kaskhedikar N, Paulsdorf J, Burjanadze M, Karatas Y, Roling B, Wiemhöfer HD (2006) Solid State Ionics 177:2699
Karatas Y, Kaskhedikar N, Burjanadze M, Wiemhöfer H-D (2006) Macromol Chem Phy 207:419
Ratner MA, Shriver DF (1988) Chem Rev 88:109
Zhang Z, Lyons LJ, Jin JJ, Amine K, West R (2005) Chem Mater 17:5646
Amine K, Wang Q, Vissers DR, Zhang Z, Rossi NAA, West R (2006) Electrochem Comm 8:429
Croce F, Appetecchi GB, Persi L, Scrosati B (1998) Nature 394:456
Croce F, Settimi L, Scrosati B (2006) Electrochem Comm 8:364
Tomalia DA, Baker H, Dewald J, Hall M, Kallos G, Martin S, Roeck J, Ryder J, Smith P (1985) Polym J 17:117
Hawker CJ, Fréchet JMJ (1990) J Am Chem Soc 112:7638
de Brabander-van den Berg EMM, Meijer EW (1993) Angew Chem Int Ed 32:1308
Newkome GR, Shreiner CD (2008) Polymer 49:1
Astruc D, Boisselier E, Ornelas C (2010) Chem Rev 110:1857
Lee B, Park YH, Hwang YT, Oh W, Yoon J, Ree M (2005) Nat Mater 4:147
Delort E, Darbre T, Reymond JL (2004) J Am Chem Soc 126:15642
Aulenta F, Hayes W, Rannard S (2003) Eur Polym J 39:1741
Ramírez RE, Torres-González LC, Hernández A, García A, Sánchez EM (2010) J Phys Chem B 114:4271
Dillon REA, Shriver DF (2001) Chem Mater 13:1369
Krishna TR, Jayaraman N (2003) J Org Chem 68:9694
Jayamurugan G, Jayaraman N (2006) Tetrahedron 62:9582
Fleig J (2003) Solid State Ionics 121:279
Mourey TH, Turner SR, Rubinstein M, Fréchet JMJ, Hawker CJ, Wooley KL (1992) Macromolecules 25:2401
Lebdeh YA, Davidson I (2009) J Electrochem Soc 156:A60
Das S, Bhattacharyya AJ (2010) Solid State Ionics 181:1732
Das SK, Darmakolla S, Bhattacharyya AJ (2010) J Mater Chem 20:1600
Dominko R, Bele M, Gaberscek M, Ramskar M, Hanzel D, Goupil JM, Pejovnik S, Jamnik J (2006) J Power Sources 153:274
Acknowledgements
The authors thank Uday Maitra and S. Banerjee for rheology measurements. SD acknowledges CSIR for SRF. NJ and AJB thank DST, Nano Mission Government of India for a financial support. AJB also thanks CEN, IISc for infrastructural support. AJB also thanks DST, Nano Mission for a financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 876 kb)
Rights and permissions
About this article
Cite this article
Das, S., Suresh, B.R., Jayaraman, N. et al. Ionic conductivity of bis(2-cyanoethyl) ether-lithium salt and poly(propylether imine)-lithium salt liquid electrolytes. J Polym Res 19, 9924 (2012). https://doi.org/10.1007/s10965-012-9924-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-012-9924-3