Abstract
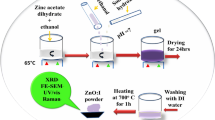
The optical properties have been investigated using EDX, UV–Visible spectroscopy and Raman spectroscopy. The nanocomposite formation was confirmed using EDX as well as UV-Visible absorption spectroscopy. Raman study confirms the β phase of PVDF. The UV–Vis spectroscopy accounts for a significant continuous decline in optical band gap and optical activation energy, while increase in refractive index with ZnO nanoparticles incorporation. The results imply that the effectiveness in shielding of UV radiation is due to absorption capacity of ZnO nanoparticles incorporated in PVDF. The increase of absorption in the UV-region of the spectrum is due to the excitations of donor level electrons to the conduction band at these energies. This decrease of band gap may be attributed by presence of unstructured bulk defects. The optical properties of nanocomposite thin films were shown to depend on ZnO content and possessed the most optimal optical properties.







Similar content being viewed by others
References
Groh W, Zimmermann A (1991) What is the lowest refractive index of an organic polymer. Macromolecules 24:6660–6663
Nanda KK, Sarangi SN, Mohanty S, Sahu SN (1998) Optical properties of CdS nanocrystalline films prepared by a precipitation technique. Thin Solid Films 322:21–27
Rozenberg BA, Tenne R (2008) Polymer-assisted fabrication of nanoparticles and nanocomposites. Prog Polym Sci 33:40–112
Rong MZ, Zhang MQ, Zheng YX, Zeng HM, Walter R, Friedrich K (2000) Irradiation graft polymerization on nano-inorganic particles: an effective means to design polymer based nanocomposites. J Mater Sci Lett 19:1159
Dimitry OIH, Abdeen ZI, Ismail EA, Saad ALG (2010) Preparation and properties of elastomeric polyurethane/organically modified montmorillonite nanocomposites. J Polym Res 17:801–813
Haridas M, Srivastava S, Basu JK (2008) Optical properties of polymer nanocomposites. Bull Mater Sci 31:213–217
Sui XM, Shao CL, Liu YC (2005) White-light emission of polyvinyl alcohol/ZnO hybrid nanofibers prepared by electrospinning. Appl Phys Lett 87:113–115
Xiong M, Gu G, You B, Wu L (2003) Preparation and characterization of poly (styrene butylacrylate) latex/nano-ZnO nanocomposites. J Appl Polym Sci 90:1923–1931
Liang S, Xiao K, Mo Y, Huang X (2012) A novel ZnO nanoparticle blended polyvinylidene fluoride membrane for anti-irreversible fouling. J Membr Sci 394:184–192
Mitra P, Chatterjee AP, Maiti HS (1998) ZnO thin film sensor. Mater Lett 35:33–38
Dimova-Malinovska D (1999) Application of stain-etched porous silicon in light emitting diodes and solar cells. J Lumin 80:207–211
Kumar AP, Depan D, Tomer NS, Singh RP (2009) Nanoscale particles for polymer degradation and stabilization-trends and future perspectives. Prog Polym Sci 34:479–515
Lee J, Bhattacharyya D, Easteal AJ, Metson JB (2008) Properties of nano-ZnO/poly(vinyl alcohol)/poly(ethylene oxide) composite thin films. Curr Appl Phys 8:42–47
Abdullah TMM, Okuyama K (2003) Generating blue and red luminescence from ZnO/Poly (ethylene glycol) nanocomposites prepared using an in-situ method. Adv Funct Mater 11:800–804
Gaur MS, Indolia AP (2010) Thermally stimulated dielectric properties of polyvinylidenefluoride–zinc oxide nanocomposites. J Therm Anal Calorim 103:977–985
Srivastava AK, Virk HS (2000) 50 MeV lithium ion beam irradiation effects in poly vinylidene fluoride (PVDF) polymer. Bull Mater Sci 23:533–538
Elilarassi R, Chandrasekaran G (2011) Synthesis, structural and optical characterization of Ni-doped ZnO nanoparticles. J Mater Sci Mater Electron 22:751–756
Islam MR, Podder J (2009) Optical properties of ZnO nano fiber thin films grown by spray pyrolysis of zinc acetate precursor. Cryst Res Technol 44:286–292
Mott NF, Davis EA (1979) Electronic Processes in Non-Crystalline Materials, 2nd edn. Clareddon, Oxford
Mathai CJ, Saravanan S, Anantharaman MR, Venkatachalam S, Jayalekshmi S (2002) Effect of iodine doping on the bandgap of plasma polymerized aniline thin films. J Phys D Appl Phys 35:2206–2210
Rathore BS, Gaur MS, Singh F, Singh KS (2012) Optical and dielectric properties of 55 MeV carbon beam-irradiated polycarbonate films. Radiat Eff Defect Solid 167:131–140
Majeed Khan MA, Zulfequar M, Husain M (2003) Optical investigation of a-Se100-xBix alloys. Opt Mater 22:21–29
Susilawati DA (2009) Dose response and optical properties of Dyed Poly Vinyl Alcohol-Trichloroacetic Acid Polymeric Blends Irradiated with Gamma-Rays. American J Appl Sci 6:2071–2077
Radwan RM (2007) Electron induced modification in optical properties of polypropylene. J Phys D Appl Phys 40:374–379
Cody GD, Tiedje T, Abeles B, Brooks B, Goldstein Y (1981) Disorder and the optical-absorption edge of hydrogenated amorphous silicon. Phys Rev Lett 47:1480–1483
Abay B, Guder HS, Yogurtchu YK (1999) Urbach–Martienssen’s tails in layered semiconductor GaSe. Solid State Commun 112:489–494
Long DA (1977) Raman spectroscopy. McGraw Hill, New York
Boerio FJ, Koenig JL (1969) Raman scattering in nonplanar poly (vinylidene fluoride). J Polym Sci Part A2 Polym Phys 7:1489–1494
Kuptsov AH, Zhizhin GN (1998) Handbook of Fourier Transform Raman and Infrared Spectra of polymers. Elsevier, Amsterdam
Acknowledgements
The authors are thankful to Defence Research & Development Organization (Vide letter no. ERIP/ER/0804419/M/01/1113) New Delhi (India) for providing financial support. One of the authors (Ajay Pal Indolia) acknowledges DRDO for a senior research fellowship. The authors are also thankful to Director, AIRF-JNU New Delhi (India) for providing Raman and EDX characterization facilities.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Indolia, A.P., Gaur, M.S. Optical properties of solution grown PVDF-ZnO nanocomposite thin films. J Polym Res 20, 43 (2013). https://doi.org/10.1007/s10965-012-0043-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10965-012-0043-y