Abstract
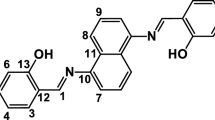
A pure benzoxazine synthesized from phenolphthalein through Mannich reaction, 3,3′-bis(3,4-dihydro-3-phenyl-2H-1,3-benzoxazinyl)-1(3H)-isobenzofuranone (Boz-BP), was characterized by FTIR and 1H NMR. Its curing behavior was different with that of 2,2′-bis(4-phenyl-3,4-dihydro-2H-1,3-benzoxazinyl)isopropane (Boz-BA) because Boz-BP possesses highly steric and inactivated phenol rings. During curing period, the reactivity of ortho position of phenolic hydroxyl group was retarded and the addition of -CH2- group generated in ring-opening reaction preferred to taking place on para position of aniline ring, which is beneficial for better thermal properties. Furthermore, additional hydrogen bonding would be formed between carbonyl groups on phthalide structure and hydroxyl groups in Boz-BP based polymer (P(Boz-BP)). Compared with the analogous polymer from Boz-BA (P(Boz-BA)), P(Boz-BP) displayed higher glass transition temperature, better thermal stability, better dimensional stability and flame-retardant properties.















Similar content being viewed by others
References
Macko JA, Ishida H (2001) Macromol Chem Phys 202:2351–2359
Macko JA, Ishida H (2001) Polymer 42:227–240
Macko JA, Ishida H (2001) Polymer 42:6371–6383
Su YC, Chang FC (2003) Polymer 44:7989–7996
Wang CF, Wang YT, Tung PH, Chang FC et al (2006) Langmuir 22:8289–8292
Liao CS, Wang CF, Lin HC, Chou HY, Chang FC (2008) J Phys Chem C 112:16189–16191
Ishida H, Allen DJ (1996) J Polym Sci Polym Phys 34:1019–1030
Ishida H, Low HY (1997) Macromolecules 30:1099–1106
Liu X, Gu Y (2002) J Appl Polym Sci 84:1107–1113
Sudo A, Kudoh R, Nakayama H, Arima K, Endo T (2008) Macromolecules 41:9030–9034
Kim HJ, Brunovska Z, Ishida H (1999) Polymer 40:6565–6573
Shen SB, Ishida H (1996) J Appl Polym Sci 61:1595–1605
Shen SB, Ishida H (1999) J Polym Sci Polym Phys 37:3257–3268
Russell VM, Koenig JL, Low HY, Ishida H (1998) J Appl Polym Sci 70:1401–1411
Russell VM, Koenig JL, Low HY, Ishida H (1998) J Appl Polym Sci 70:1413–1425
Low HY, Ishida H (1999) J Polym Sci Polym Phys 37:647–659
Kumar KSS, Nair CPR, Radhakrishnan TS, Ninan KN (2007) Eur Polym J 43:2504–2514
Ishida H, Sanders DP (2000) Macromolecules 33:8149–8157
Ghetiya RM, Kundariya DS, Parsania PH, Patel VA (2008) Polym Plast Technol Eng 47:836–841
Cao HW, Xu RW, Liu H, Yu DS (2006) Des Monomers and Polym 19:369–382
Ning X, Ishida H (1994) J Polym Sci Polym Chem 32:1121–1129
Vyazovkin S, Sbirrazzuoli N (1996) Macromolecules 29:1867–1873
Sponton M, Larrechi MS, Ronda JC, Galia M, Cadiz V (2008) J Polym Sci Polym Chem 46:7162–7172
Dunker JP, Zarate EA, Ishida H (1996) J Phys Chem 100:13514–13520
Kim HD, Ishida H (2002) J Phys Chem A 106:3271–3280
Liu YL, Chou CI (2005) J Polym Sci Polym Chem 43:5267–5285
Low HY, Ishida H (1998) J Polym Sci Polym Phys 36:1935–1946
Low HY, Ishida H (1999) Polymer 40:4365–4376
Hemvichian K, Ishida H (2002) Polymer 2002:4391–4402
Hemvichian K, Laobuthee A, Chirachanchai S, Ishida H (2002) Polym Degrad Stab 76:1–15
Brunovska Z, Lyon R, Ishida H (2000) Therm Aca 357:195–203
Choi SW, Ohba S, Brunovska Z, Hemvichian K, Ishida H (2006) Polym Degrad Stab 91:1166–1178
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 20774060).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, P., Gu, Y. Synthesis and curing behavior of a benzoxazine based on phenolphthalein and its high performance polymer. J Polym Res 18, 1725–1733 (2011). https://doi.org/10.1007/s10965-011-9578-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10965-011-9578-6