Abstract
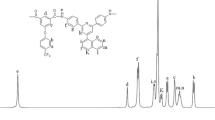
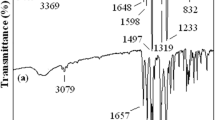
1,5-Bis(4-trimellitimido)naphthalene (II) was prepared by the condensation reaction of 1,5-naphthalenediamine and trimellitic anhydride. A series of aromatic poly(amide-imide)s (IV a–o) was synthesized by the direct polycondensation of the diimide-diacid (II) and various aromatic diamines (III a–o). The reaction utilized triphenyl phosphite and pyridine as condensing agents in the presence of calcium chloride in N-methyl-2-pyrrolidone (NMP). The inherent viscosities of the resulting poly(amide-imide)s were in the range of 0.55∼1.39 dL/g. These polymers were generally soluble in polar solvents, such as N,N-dimethylacetamide (DMAc), NMP, N,N-dimethylformamide (DMF). Flexible and tough poly(amide-imide) films were obtained by casting from a DMAc solution and had tensile strengths of 90∼145 MPa, elongations to break of 5∼13 %, and initial moduli of 2.29∼3.73 GPa. The glass transition temperatures of some poly(amide-imide)s were recorded in the range of 206∼218 °C, and most of the polymers did not show discernible glass transition on their DSC traces. The 10% weight loss temperatures were above 522 °C in nitrogen and above 474 °C in air atmosphere.
Similar content being viewed by others
References
D. Wilson, H. D. Stenzenberger and P. H. Hergenrother Eds., Polyimides, Blackie & Son, Glascow and London, 1990.
M. K. Ghosh and K. L. Mittal Eds., Polyimides: Fundamentals and Applications, Marcel Dekker, New York, 1996.
Standard Oil Co., U. S. Pat. 3,920,612 (1975).
Standard Oil Co., U. S. Pat. 4,048,114 (1977).
B. A. Zhubanov, S. A. Mashkevich and S. R. Rafikov, Vysokomol. Soedin., Ser. A, 14, 2201 (1972).
J. de Abajo, J. P. Gabarda and J. Fontan, Angew, Makromol. Chem., 71, 143 (1972).
S. Maiti and A. Ray, J. Appl. Polym. Sci., 27, 4345 (1982).
S. Maiti and A. Ray, J. Polym. Sci., Polym. Chem. Ed., 22, 999 (1983).
H. Shigeyoshi, Y. Tsunemasa, I. Masao and U. Moriya, Jpn. Pat. 72-30,312 (1972).
G. Bellmann and A. Groult, Fr. Demande 2,399,452 (1979).
M. Ghosh, Angew. Makromol. Chem., 172, 165 (1989).
T. Nishioka, I. Ishibashi, Y. Suzuki and Y. Hamada, Jpn. Pat. 70-35,072 (1970).
K. Tanaka, Jpn. Pat. 71-413 (1971).
J. de Abajo, A. G. Brunet, S. G. Babe and J. Fontan, An. Quim., 70, 908 (1974).
J. L. Nieto, J. G. de la Campa and J. de Abajo, Makromol. Chem., 183, 557 (1982).
Y. Segui, H. Benalla and J. Farenc, Proc. Int. Conf. Conduct. Breakdown Solid Dielectr., 1st, 124 (1989).
N. Yamazaki, M. Matsumoto and F. Higashi, J. Polym. Sci., Polym. Chem. Ed., 13, 1373 (1975).
C. P. Yang and S. H. Hsiao, Makromol. Chem., 190, 2119 (1989).
S. H. Hsiao and C. P. Yang, J. Polym. Sci., Polym. Chem. Ed., 28, 1449 (1990).
S. H. Hsiao and C. P. Yang, Makromol. Chem., 191, 155 (1990).
S. H. Hsiao and C. P. Yang, J. Polym. Sci., Polym. Chem. Ed., 29, 447 (1991).
C. P. Yang, S. H. Hsiao and J. H. Lin, J. Polym. Sci., Polym. Chem. Ed., 29, 1175 (1991).
C. P. Yang, S. H. Hsiao and Y. Y. Yen, J. Polym. Sci., Polym. Chem. Ed., 30, 1885 (1992).
C. P. Yang, S. H. Hsiao and J. H. Lin, J. Polym. Sci., Polym. Chem. Ed., 30, 1865 (1993).
C. P. Yang, J. M. Cheng and S. H. Hsiao, Makromol. Chem., 193, 455 (1992).
N. A. Adrova, M. M. Koton, A. M. Dubnova and N. P. Kuznetsov, Vysokomol. Soedin. B, 16, 504 (1974).
R. B. Stackman and A. B. Conciatori, U. S. Pat. 4,064,107 (1978).
Y. Imai, N. N. Malder and M. Kakimoto, J. Polym. Sci., Polym. Chem. Ed., 22, 2189 (1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yang, CP., Hsiao, SH. & Tsai, MR. Synthesis and properties of poly(amide-imide)s based on 1,5-bis(4-trimellitimido)naphthalene. J Polym Res 5, 23–29 (1998). https://doi.org/10.1007/s10965-006-0036-9
Issue Date:
DOI: https://doi.org/10.1007/s10965-006-0036-9