Abstract
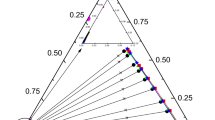
The liquid–liquid equilibrium (LLE) data for 1,3-propanediol + 1-nonanol + extractants (1,2-xylene, 1-chlorobutane, isopropyl ether and water) were measured at 298.2 K and 101.3 kPa. The separation efficiency of the extractant for the mixture of 1,3-propanediol and 1-nonanol was evaluated by the distribution constant (D) and selectivity (S). Meanwhile, the empirical equations of Bachman and Othmer-Tobias verified the consistency of the experimental data, and the squares of its linear correlation are all greater than 0.98. The thermodynamic models NRTL and UNIQUAC were used to correlate the experimental data, and different model parameters were obtained by regression. After comparing the calculated values of the model with the experimental data, it is found that the root means square deviation (RMSD%) and absolute mean deviation (AAD%) are less than 0.97% and 0.60% respectively, which indicate that both models have a reasonable correlation with the experimental data of the ternary system and can accurately predict the experimental data. Finally, the graphical user interface (GUI) was used to verify that the regression parameters have a good consistency.









Similar content being viewed by others
References
Ran, H.S., Wang, C.H., Yang, X.H., Tong, M.Y.: Application and market of 1,3-propanediol. Guangdong Chem. Ind 3 3, 29–32 (2006)
Wei, G.F., Gu, L.X., Dai, Z.B., Chang, H.: Research progress of poly (propylene terephthalate). Synth. Fiber China 34, 1–6 (2005)
Xie, Y.F.: Recycling of waste polyester. Chem. Ind. Times 19, 56–58 (2005)
Wu, X., Wu, J., Wang, S., Gao, J., Xu, D., Zhang, L., Wang, Y.: Liquid–liquid equilibrium for ternary systems (ethyl acetate/isopropyl acetate + 2,2-difluoroethanol + water) at 298.15 and 308.15 K. J. Chem. Eng. Data 66, 1399–1405 (2021)
Renon, H., Prausnitz, J.M.: Local compositions in thermodynamic excess functions for liquid mixtures. AIChE J. 14, 135–144 (1968)
Abrams, D.S., Prausnitz, J.M.: Statistical thermodynamics of liquid mixtures: a new expression for the excess gibbs energy of partly or completely miscible systems. AIChE J. 21, 116–128 (1975)
Stephenson, R., Stuart, J.: Mutual binary solubilities: water–alcohols and water–esters. J. Chem. Eng. Data 31, 56–70 (1986)
Zhang, L., Liao, Z., Zhu, C., Yu, Y., Li, Q.: Determination and correlation of liquid–liquid equilibria data for ternary system isopropyl acetate + isopropanol + water at different temperatures. J. Chem. Eng. Data 64, 972–978 (2019)
Dong, S., Sun, W., Jiang, Y., Jia, B.: Liquid–liquid equilibrium study for ternary systems of (water + furfuryl alcohol + solvents) at 298.2 K: measurement and thermodynamic modelling. J. Chem. Thermodyn. 148, 106136 (2020)
Esquivel, M.M., Bernardo-Gil, M.G.: Liquid–liquid equilibria for the systems: water/1-pentanol/acetic acid and water/1-hexanol/acetic acid. Fluid Phase Equilib 62, 97–107 (1991)
Shen, S., Chen, Y., Jiang, M.: Experimental measurement, correlation and COSMO-SAC prediction of liquid–liquid equilibrium for MIPK + dimethylphenols + water mixtures. J. Chem. Thermodyn. 140, 105900 (2020)
Zhu, Z., Xu, Y., Feng, T., Wang, N., Liu, K., Fan, H., Wang, L.: Measurement and correlation of liquid–liquid equilibria of three imidazolium ionic liquids with acetone and cyclohexane. J. Mol. Liq. 298, 111947 (2020)
Yan, H., Fan, W., Sun, S., Guo, H., Han, Y., Li, Q.: Liquid–liquid equilibria data and thermodynamic modeling for the ternary system of water, 2,2,3,3-tetrafluoro-1-propanol, and different solvents. J. Chem. Eng. Data 66, 2327–2332 (2021)
Reyes-Labarta, J.A.: Graphical user interface (GUI) for the representation of GM surfaces (using the NRTL model) and curves, including tie-lines and hessian matrix. RUA. (2015)
Guo, H.Y., Cao, T.Y., Zhao, Y., Liu, H., Li, J., Li, Q.S.: Liquid–liquid equilibrium measurement and thermodynamic modeling for ternary system of water + cyclohexanone + solvents (n-propyl acetate, methyl acetate) at different temperatures. J. Chem. Thermodyn. 166, 106672 (2022)
Lin, S., Yan, H., Jiang, Y., Guo, H.Y., Zhao, Y., Han, M., Li, Q.S.: Liquid–liquid equilibrium for the ternary system of water, 2,2,2-trifluoroethanol and different solvents at 303.2 K under 101.3 kPa. J. Chem. Thermodyn. 170, 106783 (2022)
Wang, Z., Fan, W., Xu, D., He, S., Huang, H., Gao, J., Wang, Y.: Liquid–liquid-phase equilibrium for quaternary systems (n-decane + 1-tetradecene + 1-methylnaphthalene + sulfolane/dimethyl sulfoxide) for separation of 1-methylnaphthalene from FCC diesel. J. Chem. Eng. Data 66, 2803–2811 (2021)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yu, Y., Yi, L., Geng, T. et al. Liquid–Liquid Equilibrium for Ternary Systems of 1,3-Propanediol, 1-Nonanol and Different Extractants at 298.2 K and 101.3 kPa. J Solution Chem 52, 187–200 (2023). https://doi.org/10.1007/s10953-022-01225-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10953-022-01225-4